��Ŀ����
��1��ԭ�ӽṹ��Ԫ�����ڱ�������������ϵ��������ѧ���ʽṹ֪ʶ������ش��������⣺
�پ��У�n-1��d10ns2���ӹ��͵�Ԫ��λ�����ڱ��� ���� �壮
���ճ������й㷺Ӧ�õIJ���֣��������������������˸�Ԫ�أ���Ԫ�ػ�̬ԭ��δ
�ɶԵ�����Ϊ ��
��2���ȵ���ԭ��--ԭ��������ͬ���۵���������ͬ�����Ӿ������ƵĻ�ѧ�����������ǵ���������������ģ�д�������ɶ��ԭ����ɵĺ�����N3-��������ͬ�����ӵĻ�ѧʽ �� ����Щ���ӿռ乹��Ϊ ��
��3���Ȼ�������SOCl2����һ�ֺ���Ҫ�Ļ�ѧ�Լ���������Ϊ�Ȼ�������ˮ�����Ȼ��������ӵ�VSEPR������ ������ԭ�Ӳ�ȡ �ӻ���ʽ��
��4���յ�����ɫ���ޡ��۸����������һЩ��ҵ�Ƿ���ΪʳƷ�ͻ�ױƷ�ȵ�Ⱦɫ��������Σ�����ǽ������յ��쳣���Т�4�����ͣ��յ����ŵķ��ӽṹ��ͼ1��ʾ��
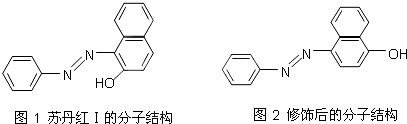
�յ������ˮ�е��ܽ�Ⱥ�С�������Ҵ������˰��ǻ�ȡ���ڶ�λ�γ�ͼ2��ʾ�Ľṹ��������ˮ�е��ܽ�Ȼ� �������С������ԭ���� ��
�پ��У�n-1��d10ns2���ӹ��͵�Ԫ��λ�����ڱ���
���ճ������й㷺Ӧ�õIJ���֣��������������������˸�Ԫ�أ���Ԫ�ػ�̬ԭ��δ
�ɶԵ�����Ϊ
��2���ȵ���ԭ��--ԭ��������ͬ���۵���������ͬ�����Ӿ������ƵĻ�ѧ�����������ǵ���������������ģ�д�������ɶ��ԭ����ɵĺ�����N3-��������ͬ�����ӵĻ�ѧʽ
��3���Ȼ�������SOCl2����һ�ֺ���Ҫ�Ļ�ѧ�Լ���������Ϊ�Ȼ�������ˮ�����Ȼ��������ӵ�VSEPR������
��4���յ�����ɫ���ޡ��۸����������һЩ��ҵ�Ƿ���ΪʳƷ�ͻ�ױƷ�ȵ�Ⱦɫ��������Σ�����ǽ������յ��쳣���Т�4�����ͣ��յ����ŵķ��ӽṹ��ͼ1��ʾ��

�յ������ˮ�е��ܽ�Ⱥ�С�������Ҵ������˰��ǻ�ȡ���ڶ�λ�γ�ͼ2��ʾ�Ľṹ��������ˮ�е��ܽ�Ȼ�
���㣺ԭ�Ӻ�������Ų�,�жϼ��ӻ����ӵĹ���,���ȵ���ԭ������Ӧ��,����Ĵ��ڶ��������ʵ�Ӱ��
ר�⣺
��������1���ٸ���Ԫ��ԭ�ӵ���Χ�����Ų����������ɽ�Ԫ�����ڱ��ֳ��������s����p����d����ds����f������n-1��dȫ���������ĵ�����s�����Ϊds��Ԫ�أ�
�ڸ��ݸ�Ԫ�صĻ�̬ԭ�ӵ����Ų�ʽ��������
��2�������������е�����=ԭ�ӵ�������+������������к��еĵ�������������ԭ�ӵĵ������ĺ������1��N����7�����ӣ�N3-��22�����ӣ�N3-��CO2Ϊ�ȵ����壬������̼Ϊֱ���ͷ��ӣ��ʿ���ΪN3-�Ĺ���Ϊֱ���ͣ�
��3�����ݼ۵��ӶԻ�������ȷ�����Ŀռ乹�ͣ�SOCl2��Sԭ�ӳ�2��S-Cl����1��S=O���ӻ��������3����Sԭ�Ӳ�ȡSP2�ӻ���������״Ϊƽ�������Σ�
��4�����ݷ���������ͷ��Ӽ���������ʷ�����
�ڸ��ݸ�Ԫ�صĻ�̬ԭ�ӵ����Ų�ʽ��������
��2�������������е�����=ԭ�ӵ�������+������������к��еĵ�������������ԭ�ӵĵ������ĺ������1��N����7�����ӣ�N3-��22�����ӣ�N3-��CO2Ϊ�ȵ����壬������̼Ϊֱ���ͷ��ӣ��ʿ���ΪN3-�Ĺ���Ϊֱ���ͣ�
��3�����ݼ۵��ӶԻ�������ȷ�����Ŀռ乹�ͣ�SOCl2��Sԭ�ӳ�2��S-Cl����1��S=O���ӻ��������3����Sԭ�Ӳ�ȡSP2�ӻ���������״Ϊƽ�������Σ�
��4�����ݷ���������ͷ��Ӽ���������ʷ�����
���
�⣺��1����������s���ӣ�Ϊs��Ԫ�أ�������A����A��HeԪ�أ�������p������ӣ�Ϊp��Ԫ�أ���ҪΪ�����0��Ԫ�أ�������d������ӣ�Ϊd��Ԫ�أ�������B������B�͢����壨�ϵ����ϵ���⣩��Ϊ����Ԫ�أ���n-1��dȫ���������ĵ�������s����ϣ�Ϊds��Ԫ�أ�������B����BԪ�أ�Ϊ���ɽ�������n-1��d10ns2���ӹ��͵�Ԫ�أ���n-1��dȫ���������2����������s����ϣ�Ϊds��Ԫ�أ�
�ʴ�Ϊ��ds����B��
�ڸ�Ԫ�صĻ�̬ԭ�ӵ����Ų�ʽΪ��1s22s22p63s23p63d24s44p2����Ԫ�ػ�̬ԭ��δ�ɶԵ�����Ϊ6���ʴ�Ϊ��6��
��2��1��N����7�����ӣ���N3-��22�����ӣ�����22�����ӵĶ�ԭ��������N2O��CO2��CNO-��BeF2��CaH2��C3H4�ȣ�N3-��CO2Ϊ�ȵ����壬CO2Ϊֱ���ͷ��ӣ��ȵ�������������������ƵĽṹ���ʿ���ΪN3-�Ĺ���Ϊֱ���ͣ�
�ʴ�Ϊ��CO2��N2O��ֱ���ͣ�
��3�����ݼ۵��ӶԻ�������ȷ�����Ŀռ乹�ͣ�SOCl2��Sԭ�ӳ�2��S-Cl����1��S=O���ӻ��������3����Sԭ�Ӳ�ȡSP2�ӻ���������״Ϊƽ�������Σ�
�ʴ�Ϊ��ƽ�������Σ�SP2��
��4����Ϊ�յ�������γɷ����������ʹ��ˮ�е��ܽ�Ⱥ�С�������Ҵ��������κ�Ľṹ���γɷ��Ӽ��������ˮ�����γ�����������ڵ�������ˮ�е��ܽ�ȣ��ʴ�Ϊ�������յ�������γɷ����������ʹ��ˮ�е��ܽ�Ⱥ�С�������κ�Ľṹ���γɷ��Ӽ��������ˮ�����γ�����������ڵ�������ˮ�е��ܽ�ȣ�
�ʴ�Ϊ��ds����B��
�ڸ�Ԫ�صĻ�̬ԭ�ӵ����Ų�ʽΪ��1s22s22p63s23p63d24s44p2����Ԫ�ػ�̬ԭ��δ�ɶԵ�����Ϊ6���ʴ�Ϊ��6��
��2��1��N����7�����ӣ���N3-��22�����ӣ�����22�����ӵĶ�ԭ��������N2O��CO2��CNO-��BeF2��CaH2��C3H4�ȣ�N3-��CO2Ϊ�ȵ����壬CO2Ϊֱ���ͷ��ӣ��ȵ�������������������ƵĽṹ���ʿ���ΪN3-�Ĺ���Ϊֱ���ͣ�
�ʴ�Ϊ��CO2��N2O��ֱ���ͣ�
��3�����ݼ۵��ӶԻ�������ȷ�����Ŀռ乹�ͣ�SOCl2��Sԭ�ӳ�2��S-Cl����1��S=O���ӻ��������3����Sԭ�Ӳ�ȡSP2�ӻ���������״Ϊƽ�������Σ�
�ʴ�Ϊ��ƽ�������Σ�SP2��
��4����Ϊ�յ�������γɷ����������ʹ��ˮ�е��ܽ�Ⱥ�С�������Ҵ��������κ�Ľṹ���γɷ��Ӽ��������ˮ�����γ�����������ڵ�������ˮ�е��ܽ�ȣ��ʴ�Ϊ�������յ�������γɷ����������ʹ��ˮ�е��ܽ�Ⱥ�С�������κ�Ľṹ���γɷ��Ӽ��������ˮ�����γ�����������ڵ�������ˮ�е��ܽ�ȣ�
������������һ���ṹ��ѧ֪ʶ���ۺ���Ŀ������ѧ�������ͽ��������������ۺ��Խ�ǿ���ѶȽϴ�

��ϰ��ϵ�д�
 ��ѧʵ����ϵ�д�
��ѧʵ����ϵ�д�
�����Ŀ
��Ũ������Ը���������У�������
| A������ | B������ | C���⻯�� | D������ |
������������ʵ���Ũ�Ȣ�NaHCO3����Na2CO3������Һ����������ȷ���ǣ�������
| A����Һ��C��H+���ڣ��� |
| B����Һ�������ӵ����ʵ���Ũ��֮�ͣ��ڣ��� |
| C������Һ�У�c��HCO3-����c��H2CO3�� |
| D������Һ�У�c��CO32-����c��H2CO3�� |
�������ʵ���Һ���������м������ɲ����գ����õ������ʹ�����ǣ�������
| A���Ȼ��� | B������þ |
| C���������� | D��̼������ |
24mL 0.05mol/L��Na2SO3�ܹ���ǡ����20mL0.02mol/L��K2Cr2O7��Һ��ȫ��Ӧ���������Ʊ�����ΪNa2SO4�����Ԫ���ڻ�ԭ�����еĻ��ϼ�Ϊ��������
| A��+6 | B��+3 | C��+2 | D��0 |
�⻯泥�NH4H�����Ȼ�淋Ľṹ���ƣ���֪����ˮ��Ӧ���������ɣ����й����⻯���������ȷ���ǣ�������
| A�������ӻ�����������Ӽ����ۼ� |
B������ʽ��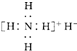 |
| C����ˮ��Ӧʱ���ǻ�ԭ��Ӧ |
| D������Ͷ��������ˮ�У�������������� |
������ʵ���õ绯ѧԭ�����͵��ǣ�������
| A��������Ʒ���ñ���䰵 |
| B��Զ���ֵ�β��װ��һ��������п�� |
| C�����������£��ڿ����������ױ���ʴ |
| D����п������������ʱ���������е���������ͭ |
