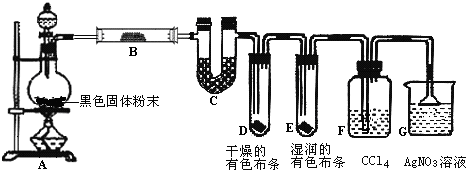
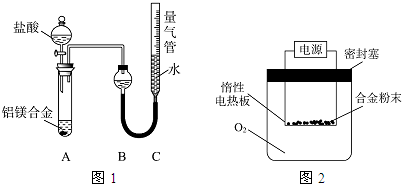
��Ŀ����
��25mL 0.1mol?L-1��NaOH��Һ����μ���0.2mol?L-1��CH3COOH��Һ����ҺpH�仯������ͼ1��ʾ��
��1��B����Һ�����ԣ����˾ݴ���Ϊ����B��ʱNaOH��Һ��CH3COOH��Һǡ����ȫ��Ӧ�����ֿ����Ƿ���ȷ�� ����ǡ������������ȷ�������ǡ����ȫ��Ӧ�ĵ����� ���AB������BC����CD���������ڣ�
��2�����ڸõζ�ʵ�飬������ѡ����ѡ����ǡ����һ�ѡ�õζ�����ͼ2��ʾ�� ��
��3����C�㣬��Һ������Ũ���ɴ�С��˳��Ϊ
��4����D��ʱ����Һ��c��CH3COO-��+c��CH3COOH�� 2c��Na+�������������������=������

��1��B����Һ�����ԣ����˾ݴ���Ϊ����B��ʱNaOH��Һ��CH3COOH��Һǡ����ȫ��Ӧ�����ֿ����Ƿ���ȷ��
��2�����ڸõζ�ʵ�飬������ѡ����ѡ����ǡ����һ�ѡ�õζ�����ͼ2��ʾ��
| ��ƿ�е���Һ | �ζ����е���Һ | ѡ��ָʾ�� | ѡ�õζ��� | |
| A | �� | �� | ʯ�� | �� |
| B | �� | �� | ���� | �� |
| C | �� | �� | ��̪ | �� |
| D | �� | �� | ��̪ | �� |
��4����D��ʱ����Һ��c��CH3COO-��+c��CH3COOH��

���㣺�к͵ζ�,����Ũ�ȴ�С�ıȽ�
ר�⣺
��������1��������ǡ����ȫ��Ӧ�������˴����ƣ���ҺӦ����ʾ���Է�����
��2��25mL 0.1mol?L-1��NaOH��Һ����μ���0.2mol?L-1��CH3COOH��Һ����ƿ��ʢ������������Һ���ζ�����ʢ�Ŵ�����Һ��ָʾ��Ӧ��ѡ�÷�̪��
��3��C������������Һ��ʾ���ԣ����ݵ���غ��ж���Һ�и�����Ũ�ȴ�С��
��4�����������غ���з�����
��2��25mL 0.1mol?L-1��NaOH��Һ����μ���0.2mol?L-1��CH3COOH��Һ����ƿ��ʢ������������Һ���ζ�����ʢ�Ŵ�����Һ��ָʾ��Ӧ��ѡ�÷�̪��
��3��C������������Һ��ʾ���ԣ����ݵ���غ��ж���Һ�и�����Ũ�ȴ�С��
��4�����������غ���з�����
���
�⣺��1��NaOH��Һ��CH3COOH��Һǡ����ȫ��Ӧʱ�������˴����ƣ����������ˮ�⣬��Һ��ʾ���ԣ���Һ��pH����7������ǡ����ȫ��Ӧʱ��Һ��pH����7������Ӧ����AB�Σ�
�ʴ�Ϊ����AB��
��2�����������Ϣ����ƿ��Ӧ��ʢ������������Һ���ζ�����ʢ�Ŵ��ᣬ����ǡ�÷�Ӧʱ��Һ��ʾ���ԣ�Ӧ��ʹ�÷�̪��Ϊָʾ��������C��ȷ��
��ѡ��C��
��3����C�㣬��Һ��pH��7��������Ũ�ȴ�������������Ũ�ȣ����ݵ���غ㣺CH3COO-+OH-=Na++H+��֪����Һ�У�c��CH3COO-����c��Na+����c��H+����c��OH-����
�ʴ�Ϊ��c��CH3COO-����c��Na+����c��H+����c��OH-����
��4��D��ʱ��������Ϊ25mL��n��CH3COO-��+n��CH3COOH��=0.2mol?L-1��0.025L=0.005mol��
n��Na+��=0.1mol?L-1��0.025L=0.0025mol������n��CH3COO-��+n��CH3COOH��=2n��Na+����
������Һ�������ͬ������c��CH3COO-��+c��CH3COOH��=2c��Na+����
�ʴ�Ϊ��=��
�ʴ�Ϊ����AB��
��2�����������Ϣ����ƿ��Ӧ��ʢ������������Һ���ζ�����ʢ�Ŵ��ᣬ����ǡ�÷�Ӧʱ��Һ��ʾ���ԣ�Ӧ��ʹ�÷�̪��Ϊָʾ��������C��ȷ��
��ѡ��C��
��3����C�㣬��Һ��pH��7��������Ũ�ȴ�������������Ũ�ȣ����ݵ���غ㣺CH3COO-+OH-=Na++H+��֪����Һ�У�c��CH3COO-����c��Na+����c��H+����c��OH-����
�ʴ�Ϊ��c��CH3COO-����c��Na+����c��H+����c��OH-����
��4��D��ʱ��������Ϊ25mL��n��CH3COO-��+n��CH3COOH��=0.2mol?L-1��0.025L=0.005mol��
n��Na+��=0.1mol?L-1��0.025L=0.0025mol������n��CH3COO-��+n��CH3COOH��=2n��Na+����
������Һ�������ͬ������c��CH3COO-��+c��CH3COOH��=2c��Na+����
�ʴ�Ϊ��=��
���������⿼�����к͵ζ�����Ŀ�ѶȲ���ע�������к͵ζ�������ָʾ����ѡ�÷������Ƚ�����Ũ�ȴ�Сʱ��ע�����غ�������غ��Ӧ�ã�������������ѧ���ķ������������������Ӧ����ѧ֪ʶ��������

��ϰ��ϵ�д�
�����Ŀ
����˵������ȷ���ǣ�������
| A�����ȵ���ʧȥ�ᾧˮ���õ���ɫ����ˮ����ͭ����һ���̽з绯 |
| B������ʯ�Na2CO3?10H2O�����ú��п�״��ɷ�ĩ״����һ�仯�Ƿ绯�Ľ�� |
| C����ɫ����ˮ����ͭ��ĩ��ˮ�γ���ɫ������ͭ�Ǹ����壬��һ���̽г��� |
| D���ӱ�����ȡ����Ʒ������ܿ����ˮ�飬��������г��� |
�ÿ������������Ʊ�������������ɫ�ϳ�·��֮һ��ͼ��ʾ��
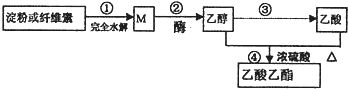
����˵����ȷ���ǣ�������

����˵����ȷ���ǣ�������
| A�������£�M����������Һ������Ӧ |
| B���ܵķ�Ӧ��������������Ӧ��������ȡ����Ӧ |
| C���ñ���̼�����Һ�ܼ����Ҵ���������������� |
| D�����ۺ���ά�صĻ�ѧʽ��Ϊ��C6H10O5��n������Ϊͬ���칹�� |