��Ŀ����
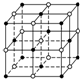 �����ϵ�Ԫ�ش�����ǽ�����Na��Mg��Al�dz������������Ԫ�أ�Fe��Cu�dz����Ĺ��ɽ���Ԫ�أ������ڻ�ѧ��ռ�м�����Ҫ�ĵ�λ��
�����ϵ�Ԫ�ش�����ǽ�����Na��Mg��Al�dz������������Ԫ�أ�Fe��Cu�dz����Ĺ��ɽ���Ԫ�أ������ڻ�ѧ��ռ�м�����Ҫ�ĵ�λ����1����Na��Mg��Al����Ԫ�ص�һ�������ɴ�С��˳��Ϊ
�ڽ�����ԭ�ӵĶѻ���ʽ����������������λ��Ϊ
��2��ѡ�����α���42ҳʵ��2-2����ʢ������ͭˮ��Һ���Թ�����백ˮ�������γ�����������Ӱ�ˮ���������ܽ⣬�õ�����ɫ������Һ�������뼫�Խ�С���ܼ������Ҵ���������������ɫ�ľ��壮
������ͭ��Һ�г�
��д�����Ľṹ��ʽ��������������λ��
����д���������ܽ�õ�����ɫ������Һ�����ӷ���ʽ
�۽ṹ�ⶨ��ʵ��֤���������ڰ�ˮ��Һ�л����ھ����У�����ɫ�������ڴ���ij�����ӣ�1mol�����Ӻ���
��3������ʢ���κκ�Fe3+����Һ���Թ��еμ�1�����軯����Һ�����Թ۲쵽��ʵ������Ϊ
��д������Na2O2���Ȼ�����Һ��Ӧ�����ӷ�Ӧ����ʽ
���㣺λ�ýṹ���ʵ����ϵӦ��,�����ijɼ����
ר�⣺Ԫ����������Ԫ�����ڱ�ר��
��������1����Na��Mg��Alͬ���ڣ�ͬ�����������Ԫ�صĵ�һ�����ܳ��������ƣ���Mg�����3s�ܼ�����2�����ӣ�Ϊȫ��ȷ��״̬�������ϵͣ���һ������Ҳ����ͬ��������Ԫ�أ��ʵ�һ������Na��Al��Mg������Ϊ���������ѻ����ƾ�����ÿ�������ӵ���λ��Ϊ8��
��2����ͭ������ˮ�������ˮ��ͭ��������ɫ��ͭ�����ṩ4���չ����
���������ӷ�Ӧ����ʽ��д������д���ɣ�
�����ݽṹʽ���㺬�еĹ��ۼ�����
��3������������KSCN����Ϸ�Ӧ���Ժ�ɫ��
�ڹ�����������ˮ��Ӧ���������������������������������������������������ɫ������
��2����ͭ������ˮ�������ˮ��ͭ��������ɫ��ͭ�����ṩ4���չ����
���������ӷ�Ӧ����ʽ��д������д���ɣ�
�����ݽṹʽ���㺬�еĹ��ۼ�����
��3������������KSCN����Ϸ�Ӧ���Ժ�ɫ��
�ڹ�����������ˮ��Ӧ���������������������������������������������������ɫ������
���
�⣺��1����Na��Mg��Alͬ���ڣ�ͬ�����������Ԫ�صĵ�һ�����ܳ��������ƣ���Mg�����3s�ܼ�����2�����ӣ�Ϊȫ��ȷ��״̬�������ϵͣ���һ������Ҳ����ͬ��������Ԫ�أ��ʵ�һ������Na��Al��Mg���ʴ�Ϊ��Mg��Al��Na��
����Ϊ���������ѻ������ƾ�����ÿ�������ӵ���λ��Ϊ8���Ȼ��ƾ����������Ӻ������ӵ���λ������6���ʴ�Ϊ��8��6��
��2��������ͭ��Һ����Ϊ����ˮ��ͭ���Ӷ�ʹ����Һ������ɫ��ˮ��ͭ����Ϊ��[Cu��H2O��4]2+������Cu2+��4��OH-�γ���λ������ṹʽΪ��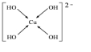 ���ʴ�Ϊ��������
���ʴ�Ϊ�������� ��
��
�ڰ�ˮ������ͭ��Ӧ����������ͭ��ɫ����������ˮ����ʱ����ˮ��������ͭ��Ӧ���ɿ����Ե�ͭ�����������������ܽ�õ�����ɫ������Һ���漰�����ӷ���ʽΪ��Cu2++2NH3?H2O�TCu��OH��2��+2NH4+��Cu��OH��2+4NH3�T[Cu��NH3��4]2++2OH-���ʴ�Ϊ��Cu��OH��2+4 NH3?H2O=[Cu��NH3��4]2++2OH-+4H2O��
������ɫ��Һ�е�������Ϊ�İ���ͭ�����ӣ�[Cu��NH3��4]2+��1mol[Cu��NH3��4]2+�д���4+12=16mol�Ҽ����ʴ�Ϊ��16��
��3����Fe3+��KSCN����Fe��SCN��3������������Ժ�ɫ���ʴ�Ϊ����Һ��ΪѪ��ɫ��
�ڹ���������ˮ��Ӧ�����������ƺ����������������������������������������������������ӷ�Ӧ����ʽΪ��6Na2O2+6H2O+4Fe3+�T4Fe��OH��3��+12Na++3O2�����ʴ�Ϊ��6Na2O2+6H2O+4Fe3+�T4Fe��OH��3��+12Na++3O2����
����Ϊ���������ѻ������ƾ�����ÿ�������ӵ���λ��Ϊ8���Ȼ��ƾ����������Ӻ������ӵ���λ������6���ʴ�Ϊ��8��6��
��2��������ͭ��Һ����Ϊ����ˮ��ͭ���Ӷ�ʹ����Һ������ɫ��ˮ��ͭ����Ϊ��[Cu��H2O��4]2+������Cu2+��4��OH-�γ���λ������ṹʽΪ��
 ���ʴ�Ϊ��������
���ʴ�Ϊ�������� ��
���ڰ�ˮ������ͭ��Ӧ����������ͭ��ɫ����������ˮ����ʱ����ˮ��������ͭ��Ӧ���ɿ����Ե�ͭ�����������������ܽ�õ�����ɫ������Һ���漰�����ӷ���ʽΪ��Cu2++2NH3?H2O�TCu��OH��2��+2NH4+��Cu��OH��2+4NH3�T[Cu��NH3��4]2++2OH-���ʴ�Ϊ��Cu��OH��2+4 NH3?H2O=[Cu��NH3��4]2++2OH-+4H2O��
������ɫ��Һ�е�������Ϊ�İ���ͭ�����ӣ�[Cu��NH3��4]2+��1mol[Cu��NH3��4]2+�д���4+12=16mol�Ҽ����ʴ�Ϊ��16��
��3����Fe3+��KSCN����Fe��SCN��3������������Ժ�ɫ���ʴ�Ϊ����Һ��ΪѪ��ɫ��
�ڹ���������ˮ��Ӧ�����������ƺ����������������������������������������������������ӷ�Ӧ����ʽΪ��6Na2O2+6H2O+4Fe3+�T4Fe��OH��3��+12Na++3O2�����ʴ�Ϊ��6Na2O2+6H2O+4Fe3+�T4Fe��OH��3��+12Na++3O2����
������������Ҫ������ǵ�һ�����ܴ�С�жϡ����Ӿ�������λ����ȷ�����������λ����֪ʶ�ȣ��ۺ��Խ�ǿ����һ�����Ѷȣ�

��ϰ��ϵ�д�
 ͬ����չ�Ķ�ϵ�д�
ͬ����չ�Ķ�ϵ�д�
�����Ŀ
��֪ʯī��ȼ����Ϊ��H1�����ʯ��ȼ����Ϊ��H2������˵����ȷ���ǣ�������
| A������ȼ�վ�Ϊ���ȷ�Ӧ������ʯīΪ��Ҫ����Դ |
| B������ȼ�շ�Ӧ���ȣ����ԡ�H1�͡�H2��ȡ��ֵ |
| C������ʯī�ͽ��ʯͬΪ̼Ԫ����ɣ����ԡ�H1=��H2 |
| D������ʯī�ͽ��ʯ���ڲ�ͬ�ĵ��ʣ����ԡ�H1�͡�H2���� |
���в���Ҫ�õ�����������ǣ�������
| A�����ά | B�������оƬ |
| C��ʯӢ�ӱ� | D����ͨ���� |
25��ʱ��������Һ������������������ǣ�������
| A��Fe2+��Mg2+��ClO-��Cl-�ܴ������� | ||
| B�������̪��Һ�Ժ�ɫ����Һ�У�K+��Na+��NO3-��SO42- | ||
C��������K+��Na+��NO3-��CO32-����Һ��c��H+��=
| ||
| D��C��Fe3+��=0.1 mol/L����Һ�У�NH4+��Al3+��I-��SO42- |
����ɫ��Һ���ܴ���������������ǣ�������
| A��Ca2+��Cl-��CO32- |
| B��K+��Na+��SO42- |
| C��HCO3-��Na+��OH- |
| D��K+��Fe3+��SCN- |
����ռ����Ƿ���д������ȷ�����ǣ�������
| A�����ȿ��Ƿ����������� |
| B������������Ƿ����������� |
| C���ܽ������Һ������� |
| D�����տ������Ƿ�Ϊ��ɫ |
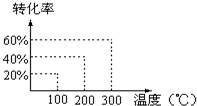 ��X��Y��1��2������Ȼ�Ϻ������ܱ������У���ѹ��3��107Pa���������·�Ӧ����g��+2Y��g��?2Z��g�����ﵽƽ��״̬ʱ����÷�Ӧ��������ʵ�����������������ʵ�����ȣ��й�������ͼ����Ӧ��Ӧ���¶��ǣ�������
��X��Y��1��2������Ȼ�Ϻ������ܱ������У���ѹ��3��107Pa���������·�Ӧ����g��+2Y��g��?2Z��g�����ﵽƽ��״̬ʱ����÷�Ӧ��������ʵ�����������������ʵ�����ȣ��й�������ͼ����Ӧ��Ӧ���¶��ǣ�������