��Ŀ����
(6��)(1)����ʵ����ƻ������������__________��
a��ʵ�������屽ʱѡ�õ��Լ���������ˮ��������м��
b������������Һʱ������ˮ��μӵ� AgNO3��Һ�У��ӱ���ֱ������ǡ����ȫ��ʧ��
c����10����������뵽1mL10%���ռ���Һ�У�����Ƭ�̺��ٵμ�2��2%����������Һ���Լ���ˮ�����ɵ������ӡ�
d��ʵ��������Ȳʱ��Ϊ�˼�����Ӧ���ʣ����ñ���ʳ��ˮ��������ˮ��
(2)��ˮ����ѧ��ѧʵ���г��õ�һ���Լ�������ˮ�ɼ�������и������ʣ���д��������Ӧ�Ļ�ѧ����ʽ��
�ٱ������Ҵ�
����ϩ������
(6��)(1)bd
(2) 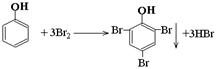 CH2=CH2+Br2®CH2BrCH2Br
CH2=CH2+Br2®CH2BrCH2Br
����

��ϰ��ϵ�д�
 һ����ʦȨ����ҵ��ϵ�д�
һ����ʦȨ����ҵ��ϵ�д�
�����Ŀ



