��Ŀ����
9��������Ӧ���л��ϳ���һ����Ҫ�ķ�Ӧ���練Ӧ�٣�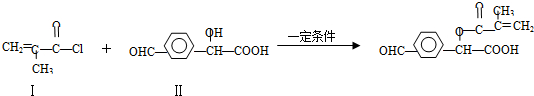
���������ɻ������ϳɣ�
��1�����������Br2�ӳɵIJ���Ľṹ��ʽΪCH2BrCBr��CH3��COCl������������ɻ�����I�ķ�Ӧ����Ϊȡ����Ӧ��
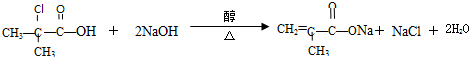
��2����֪�������ĺ˴Ź����������������շ壬�����֮��ΪΪ6��1�������Ľṹ��ʽΪ��CH3��2CClCOOH���������������NaOH����Һ���ȷ�Ӧ�Ļ�ѧ����ʽΪ��
 ��
����3��ͬʱ�������������Ģ��ͬ���칹�干��9�֣�
�ٱ�����ֻ��2��ȡ������
�ڿɷ���ˮ�ⷴӦ����ˮ�������ʹFeCl3��Һ��ɫ
������NaHCO3��Ӧ��������
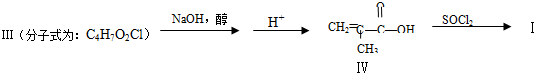
��4�����������
 �������Ļ��������һ��������Ҳ�ܷ������Ʒ�Ӧ�ٵķ�Ӧ�������л��������������Ľṹ��ʽΪ
�������Ļ��������һ��������Ҳ�ܷ������Ʒ�Ӧ�ٵķ�Ӧ�������л��������������Ľṹ��ʽΪ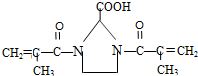 ��
��
���� ��1�����ݻ������Ľṹ��֪�����е�̼̼˫�����巢���ӳɷ�Ӧ���Ƚϻ�������ͻ�����I�Ľṹ��ʽ��֪�����е��ǻ�����ԭ��ȡ����
��2������������������ƵĴ���Һ�з�����ȥ��Ӧ���ữ�â�����ĺ˴Ź����������������շ壬�����֮��ΪΪ6��1����Ϣ��Ľṹ��֪��Ľṹ��ʽ��
��3�������������ٱ�����ֻ��2��ȡ�������ڿɷ���ˮ�ⷴӦ����ˮ�������ʹFeCl3��Һ��ɫ��˵��ˮ������з��ǻ���������NaHCO3��Ӧ�������ݣ�˵�����Ȼ�����Ϣ�Ľṹ��ʽ��д���ͬ���칹�壻
��4����������� �е��뵪��������ԭ�ӱ���������� CH2=C��CH3��CO-����ȡ�����ݴ��ƶϢ��Ľṹ��
�е��뵪��������ԭ�ӱ���������� CH2=C��CH3��CO-����ȡ�����ݴ��ƶϢ��Ľṹ��
��� �⣺��1�����ݻ������Ľṹ��֪�����е�̼̼˫�����巢���ӳɷ�Ӧ�õ��Ľṹ��ʽΪCH2BrCBr��CH3��COCl���Ƚϻ�������ͻ�����I�Ľṹ��ʽ��֪�����е��ǻ�����ԭ��ȡ�������Ի���������ɻ�����I�ķ�Ӧ����Ϊȡ����Ӧ��
�ʴ�Ϊ��CH2BrCBr��CH3��COCl��ȡ����Ӧ��
��2������������������ƵĴ���Һ�з�����ȥ��Ӧ���ữ�â�����ĺ˴Ź����������������շ壬�����֮��ΪΪ6��1����Ϣ��Ľṹ��֪��Ľṹ��ʽΪ ��CH3��2CClCOOH����Ӧ����ʽΪ ��
��
�ʴ�Ϊ����CH3��2CClCOOH�� ��
��
��3�������������ٱ�����ֻ��2��ȡ�������ڿɷ���ˮ�ⷴӦ����ˮ�������ʹFeCl3��Һ��ɫ��˵��ˮ������з��ǻ���������NaHCO3��Ӧ�������ݣ�˵�����Ȼ�����Ϣ�Ľṹ��ʽ��֪���ͬ���칹��Ϊ����������-CH3��-OOCCOOH����-OOCH��-CH2COOH����CH3COO-��-COOH��ÿ�ֶ����ڼ�����֣����Թ���9�֣�
�ʴ�Ϊ��9��
��4����������� �е��뵪��������ԭ�ӱ���������� CH2=C��CH3��CO-����ȡ�������Ԣ��Ľṹ��ʽΪ
�е��뵪��������ԭ�ӱ���������� CH2=C��CH3��CO-����ȡ�������Ԣ��Ľṹ��ʽΪ ��
��
�ʴ�Ϊ�� ��
��
���� ���⿼���л���ĺϳɣ���Ŀ�ѶȲ������Ĺؼ��ǰ����л�������ŵ�ת�������ʣ�����ʱע�����⣬���������Ϣ��

 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д�| A�� | ������Ӧ��SO3+H2O+BaCl2�TBaSO4��+2HCl | |
| B�� | ������ɫ���� | |
| C�� | ���������� | |
| D�� | ����SO2ͨ��BaCl2��Һ�У�Ҳ�������SO3ͨ��BaCl2��Һ����ͬ������ |
| A�� | Fe2��SO4��3 | B�� | KNO3 | C�� | Na2SO4 | D�� | FeSO4 |
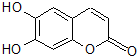 �в�ҩ��Ƥ�к��е���Ҷ����������ͼ�����п������ã���1mol�����ʷֱ���Ũ��ˮ��H2��ȫ��Ӧ�������ĵ�Br2��H2�����ʵ������ֱ�Ϊ��������
�в�ҩ��Ƥ�к��е���Ҷ����������ͼ�����п������ã���1mol�����ʷֱ���Ũ��ˮ��H2��ȫ��Ӧ�������ĵ�Br2��H2�����ʵ������ֱ�Ϊ��������| A�� | 2molBr2 3molH2 | B�� | 2molBr2 4molH2 | C�� | 3molBr2 4molH2 | D�� | 3molBr2 5molH2 |
| A�� | ԭ�Ӱ뾶��С��������Ϊ��X��Y��Z��W | |
| B�� | Z����������������ij�ᷴӦ��������ij�Ӧ�������������� | |
| C�� | Y������һ�������¿��Ը����������������û���Ӧ | |
| D�� | �����£�0.1 mol•L-1 W����̬�⻯���ˮ��Һ�������ӵ�Ũ��Ϊ0.2mol•L-1 |
| A�� | pH=1����ɫ��Һ��Na+��Cu2+��NO3-��SO42- | |
| B�� | ���ܽ�Al��OH��3�������Һ��K+��Na+��HCO3-��SO42- | |
| C�� | ˮ�������c��H+��=10-12mol/L����Һ��Ba2+��Na+��Cl-��NO3- | |
| D�� | ��ʹ����KI��ֽ��������Һ��K+��Fe2+��NO3-��Cl- |
| A�� | 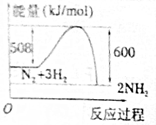 | B�� | 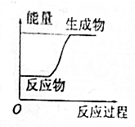 | ||
| C�� | 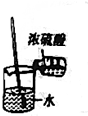 | D�� | 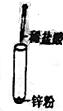 |
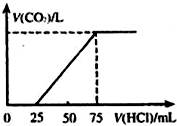 ��10mL NaOH��Һ��������ͨ��һ������CO2���õ���Na2CO3��NaHCO3�Ļ����Һ����������Һ����μ���0.1mol/L�����ᣬ�ӱ���ʹ���ַ�Ӧ������CO2������������״�����������������֮��Ĺ�ϵ��ͼ��ʾ��
��10mL NaOH��Һ��������ͨ��һ������CO2���õ���Na2CO3��NaHCO3�Ļ����Һ����������Һ����μ���0.1mol/L�����ᣬ�ӱ���ʹ���ַ�Ӧ������CO2������������״�����������������֮��Ĺ�ϵ��ͼ��ʾ��