��Ŀ����
18���ס�����λͬѧ�ֱ��ò�ͬ�ķ�������100mL��3.6mol•L-1��ϡ���ᣮ��1��������18mol•L-1��Ũ����������Һ����Ҫ�õ�Ũ��������Ϊ20mL��
��2����ͬѧ����ȡŨ����С�ĵص���ʢ������ˮ���ձ��У�������ȣ�����ȴ�����º�ת�Ƶ�100mL����ƿ�У���������ˮ���ձ�������ϴ��2��3�Σ�ÿ��ϴ��ҺҲת�Ƶ�����ƿ�У�Ȼ��С�ĵ�������ƿ����ˮ���̶��߶��ݣ�����ƿ�����������µߵ�ҡ�ȣ�
�ٽ���Һת�Ƶ�����ƿ�е���ȷ�����ǽ���������������ƿ�̶������£�ʹ��Һ�ز����������ص�������ƿ�У�
��ϴ�Ӳ����У���ϴ���ձ����ϴ��ҺҲע������ƿ�У���Ŀ����ʹ������ȫת�Ƶ�����ƿ�У�
�۶��ݵ���ȷ�����Ǽ�ˮ��������ƿ�̶���1��2cmʱ�����ý�ͷ�ιܵμ�ˮ��Һ����̶������У�
���ý�ͷ�ι�������ƿ�м�ˮʱ����С��Һ�泬���˿̶ȣ������ķ�����D������ĸ��ţ���
A����������Һ�壬ʹ��Һ����̶�������
B��С�ļ�������ƿ����������ʹ��Һ����̶�������
C�����������һ������Ũ����
D����������
��3����ͬѧ����100mL��Ͳ��ȡŨ���ᣬ��������С�ĵؼ�������ˮ��������ȣ�����ȴ�����º��ټ���ˮ��100mL�̶��ߣ���������ȣ�����Ϊ�˷��Ƿ���ȷ��������ȷ��ָ�����д���֮��������ȷ����������Ͳ������Һ�����ܽ�ˮ���뵽Ũ�����У�
���� ��1������Ũ��Һ��ϡ��ǰ�����ʵ����ʵ���������㣻
��2���ٸ��ݰѲ���������������������ƿ�̶������ϻ����ʲôӰ���Լ������������÷�����
�ڴ�ϴ��Һ����������ƿ�ܷ���������з�����
�۸���Һ�����״������
�ܸ���ABCѡ���ܷ���������з�����
��3��������Ͳ�����á���ˮ�����������к�Σ�շ�����
��� �⣺��1������C1V1=C2V2�ã�V1=$\frac{{C}_{2}{V}_{2}}{{C}_{1}}$=$\frac{0.1��3.6}{18}$=20.0ml���ʴ�Ϊ20.0mL��
��2����������������������ƿ�̶������ϣ���ʹ������Һ�����ڿ̶������϶����¶���ʱ��ƫ���������ƿ����ϸ��Ϊ������Һ��������Ӧ�ò������������ʴ�Ϊ������������������ƿ�̶������£�ʹ��Һ�ز����������ص�������ƿ�У�
��ϴ��Һ�к��в������ʣ�����ϴ��Һ��������ƿ�У��ᵼ����ҺŨ�Ƚ��ͣ��ʴ�Ϊ��ʹ������ȫת�Ƶ�����ƿ�У�
�ۼ�ˮ����̶���1��2cmʱ���ý�ͷ�ιܵμ�ˮ��������ˮ�������Һ���ǰ��εģ�ֻ��Һ����Ͷ���̶�������ʱ��������Һ����ʵ������������Ũ����ƫ��ʴ�Ϊ����ˮ����̶���1��2cmʱ�����ý�ͷ�ιܵμ�ˮ��Һ����̶������У�
���ý�ͷ�ι�������ƿ�м�ˮʱ����С��Һ�泬���˿̶ȣ����۲�ȡA��B��C�ĺ��ַ�ʽ���������ó���Ҫ����Һ��ֻ���������ã��ʴ�Ϊ��D��
��3����Ͳֻ����ȡҺ�岻��������Һ�������ˮ����Ũ�����л����Һ��ɽ����ʴ�Ϊ������ȷ����������Ͳ������Һ�����ܽ�ˮ���뵽Ũ�����У�
���� ���Ũ��ƫ��ƫС��������������C=n/V�����ж������ʸı仹����Һ�ı䣬�Ӷ��ж�Ũ��ƫ����ƫС��

| A�� | ʹ������ƿǰ�������Ƿ�©ˮ | |
| B�� | ������ˮע������ƿ�У�Һ����̶�����1-2cmʱ�����ý�ͷ�ιܵμ���Һ����̶������� | |
| C�� | ������Һʱ������Ͳ��ȡ������ֱ�ӵ�������ƿ�У�������������ˮ���̶��� | |
| D�� | ���ݺ�Ǻ�ƿ�����������µߵ���ҡ�� |
| A�� | ��ʪ���pH��ֽ��ϡ��Һ��pHֵ���ⶨֵƫС | |
| B�� | ������ƿ������Һ������ʱ���ӿ̶��ߣ�������ҺŨ��ƫ�� | |
| C�� | ���������Һ�ζ�δ֪Ũ�ȵ�NaOH��Һʱ�����ζ�ǰ�ζ����������ݣ��յ����ʱ�����ݣ�������NaOH��Һ��Ũ��ƫ�� | |
| D�� | �ⶨ�кͷ�Ӧ�ķ�Ӧ��ʱ��������������У������¶�ֵƫ�� |
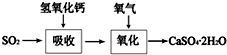 ��ҵ�ϳ����������ķ�������ˮ�����������ȣ�����������ã������ŷţ��������Ⱦ���������ѧ���գ��ɱ��Ϊ����
��ҵ�ϳ����������ķ�������ˮ�����������ȣ�����������ã������ŷţ��������Ⱦ���������ѧ���գ��ɱ��Ϊ����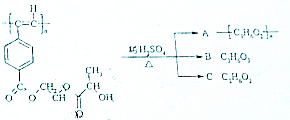 ����Ȳ����ۺ���ĺϳ�ʹ�߷��Ӳ��Ͻ����ˡ��ϳɽ����������ϵ���ѧʱ������ͼ�Ǿ���Ȳ���������M�Ľṹʽ��M��ϡ���������µ�ˮ��ʾ��ͼ��
����Ȳ����ۺ���ĺϳ�ʹ�߷��Ӳ��Ͻ����ˡ��ϳɽ����������ϵ���ѧʱ������ͼ�Ǿ���Ȳ���������M�Ľṹʽ��M��ϡ���������µ�ˮ��ʾ��ͼ��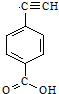 ��
��