��Ŀ����
18��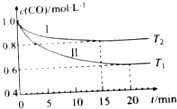 Ϊ�˼���CO�Դ�������Ⱦ��ij����С����һ�����Ϊ1L���ݻ�������ܱ������г��������CO��H2O��g�����о���CO��H2O��g����Ӧת��Ϊ��ɫ��ԴH2����ͬ�¶��£�CO��Ũ��[c��CO��]��ʱ��[t]�仯��������ͼ��ʾ��
Ϊ�˼���CO�Դ�������Ⱦ��ij����С����һ�����Ϊ1L���ݻ�������ܱ������г��������CO��H2O��g�����о���CO��H2O��g����Ӧת��Ϊ��ɫ��ԴH2����ͬ�¶��£�CO��Ũ��[c��CO��]��ʱ��[t]�仯��������ͼ��ʾ����1��CO��g��+H2O��g��?CO2��g��+H2��g����H��0���������������=����
��2��T1ʱ��0��20min��v��H2��=0.02mol/��L•min��
��3��T1��ʱ���������������ܱ������а������1��2����CO��ˮ�����Ļ��������з�Ӧ����˵���÷�Ӧ�Ѵﵽƽ��״̬����C������ĸ��ţ���
A��CO2��H2�����ʵ���֮�ȱ��ֲ���
B��CO��H2O�����ʵ���֮�ȱ��ֲ���
C��v����CO��=v����H2��
D���������ܶȱ��ֲ���
��4��T3��ʱ���÷�Ӧ�Ļ�ѧƽ�ⳣ��K=4���������м���1.00 mol CO��1.00 mol H2O��g������Ӧ���ﵽƽ�⣬��T3��ʱCO��ת����Ϊ66.7%����Ӧ�ﵽƽ��������¶ȣ���ʱƽ�ⳣ������С���������䡱��С����
��5����CO��H2O��Ӧת��Ϊ��ɫ��ԴH2�У�Ϊ�����CO��ת���ʣ��ɲ��õĴ�ʩ��BD������ĸ��ţ���
A������CO��Ũ��B������H2O��g����Ũ��C��ʹ�ô���D�������¶�E����С���������
���� ��1��ͼʾ��Ϣ���¶ȸߣ����ʿ죬�õ�T2��T1���¶�Խ�ߣ�c��CO��Խ��˵�������¶ȣ���ѧ��Ӧ�����ƶ���
��2��0��20min��v��H2��=$\frac{��c}{��t}$�������ʱ�ʾ������֮�ȵ���ϵ��֮�ȣ��ݴ˼���ش�
��3����ѧƽ��״̬��ֱ�ӱ�־�������ʹ�ϵ������Ӧ�������淴Ӧ������ȣ��ڷ�Ӧ��ϵ�и����ʵİٷֺ������ֲ��䣮
��ӱ�־���ٻ���������ѹǿ��������������ʵ�������ʱ��ĸı���ı䣨m+n��p+q�����ڸ����ʵ�Ũ�ȡ����ʵ�������ʱ��ĸı���ı䣻�۸�����������������ķ�ѹ����ʱ��ĸı���ı䣻
��4����������ʽ�Լ���ѧƽ���Ӱ������֪ʶ���ش�
��5��Ϊ�����CO��ת���ʣ��ɲ��õĴ�ʩ��ʹ��ƽ�������ƶ��Ĵ�ʩ�������Ǹı��¶ȡ�ѹǿ�����أ�����ƽ���ƶ�ԭ�����ش�
��� �⣺��1��ͼʾ��Ϣ���¶ȸߣ����ʿ죬�õ�T2��T1���¶�Խ�ߣ�c��CO��Խ��˵�������¶ȣ���ѧ��Ӧ�����ƶ������Է�Ӧ�Ƿ��ȵģ��ʴ�Ϊ������
��2��0��20min��v��CO��=$\frac{��c}{��t}$=$\frac{1.0-0.6}{20}$mol/��L•min��=0.02mol/��L•min�������ݸ����ʱ�ʾ������֮�ȵ���ϵ��֮�ȣ�����0��20min��v��H2��=0.02mol/��L•min�����ʴ�Ϊ��0.02mol/��L•min����
��3��A��CO2��H2�����ʵ���֮�ȱ��ֲ��䣬����˵�����淴Ӧ������ȣ�����ƽ�����������A����
B��CO��H2O�����ʵ���֮����һ��ʼ�ձ��ֲ����ֵ�����Դ�״̬��һ��ƽ�⣬��B����
C��v����CO��=v����H2����˵�����淴Ӧ������ȣ���״̬��ƽ��״̬����C��ȷ��
D���������ܶȵ�����������������ı�ֵ������ֵʼ����һ����ֵ�����Ը�״̬��һ����ƽ��״̬����D����
��ѡC��
��4��T3��ʱ���÷�Ӧ�Ļ�ѧƽ�ⳣ��K=4���������м���1.00 mol CO��1.00 mol H2O��g������Ӧ���ﵽƽ�⣬��T3��ʱCO�ı仯����x��
CO��g��+H2O��g��?CO2��g��+H2��g��
��ʼŨ�ȣ�1 1 0 0
�仯Ũ�ȣ�x x x x
ƽ��Ũ�ȣ�1-x 1-x x x
$\frac{{x}^{2}}{��1-x��^{2}}$=4�����x=0.67mol/L������CO��ת����Ϊ$\frac{0.67}{1}$��100%=67%����Ӧ�Ƿ��ȷ�Ӧ�����Է�Ӧ�ﵽƽ��������¶ȣ���ʱƽ�ⳣ������С���ʴ�Ϊ��66.7%����С��
��5��A������CO��Ũ�ȣ�CO��ת���ʼ�С����A����
B������H2O��g����Ũ�ȣ�ƽ�������ƶ���һ����̼��ת���ʼ�С����B��ȷ��
C��ʹ�ô�������������ת���ʵı仯����C����
D�������¶ȣ���Ӧ������У�CO��ת�������ӣ���D��ȷ��
E����С���������ѹǿ���ӣ�ƽ�ⲻһ����ת���ʲ��䣬��E����
��ѡBD��
���� ������Ҫ�����˻�ѧƽ�ⳣ������ѧƽ��ͼ��ƽ��״̬���жϵȣ���Ŀ�Ѷ��еȣ����ض�ѧ�������Ŀ��飮

| A�� | 1mol�κ����ʶ�����Լ6.02��1023��ԭ�� | |
| B�� | 0.012kg12C����Լ6.02��1023��̼ԭ�� | |
| C�� | 1����ԭ�ӵ�ʵ������ԼΪ$\frac{6.02��1{0}^{23}}{32}$g | |
| D�� | 3molOH-������Ϊ51g |
| A�� | v��A��=0.5mol•L-1•min-1 | B�� | v��B��=0.3mol•L-1•s-1 | ||
| C�� | v��C��=0.1mol•L-1•s-1 | D�� | v��D��=0.4mol•L-1•min-1 |
| A�� | �Ƹ���Һ���� | B�� | ����ɫ��ζ��������� | ||
| C�� | ˮ��Һ��Ϊ��ɫ | D�� | ˮ��Һ��Ϊ��ɫ |
WO3 ��s��+3H2 ��g�� $\stackrel{����}{?}$W ��s��+3H2O ��g��
��ش��������⣺
��1��������Ӧ�Ļ�ѧƽ�ⳣ������ʽΪ$\frac{{c}^{3}��{H}_{2}O��}{{c}^{3}��{H}_{2}��}$��
��2��ij�¶��·�Ӧ��ƽ��ʱ��H2��ˮ�����������Ϊ2��3����H2��ƽ��ת����Ϊ60%�����¶ȵ����ߣ�H2��ˮ����������ȼ�С����÷�ӦΪ��Ӧ���ȣ�����ȡ����ȡ�����
��3�������ܷ�Ӧ���̴��·�Ϊ�����Σ�������Ҫ�ɷ����¶ȵĹ�ϵ���±���ʾ��
| �¶� | 25�桫550�桫600�桫700�� |
| ��Ҫ�ɷ� | WO3 W2O5 WO2 W |
��4����֪���¶ȹ���ʱ��WO2 ��s��ת��ΪWO2��g����
WO2 ��s��+2H2 ��g���TW ��s��+2H2O ��g������H=+66.0kJ•mol-1
WO2 ��g��+2H2��g���TW ��s��+2H2O ��g������H=-137.9kJ•mol-1
��WO2 ��s���TWO2 ��g�� �ġ�H=+203.9 kJ•mol-1��
��5����˿�ƹ��е�W��ʹ�ù����л����ӷ���ʹ��˿��ϸ������I2���ӳ��ƹܵ�ʹ���������乤��ԭ��Ϊ��W ��s��+2I2 ��g��$?_{Լ3000��}^{1400��}$WI4 ��g��������˵����ȷ����ab��
a���ƹ��ڵ�I2��ѭ��ʹ��
b��WI4�ڵ�˿�Ϸֽ⣬������W�ֳ����ڵ�˿��
c��WI4�ڵƹܱ��Ϸֽ⣬ʹ�ƹܵ������ӳ�
d���¶�����ʱ��WI4�ķֽ����ʼӿ죬W��I2�Ļ������ʼ�����
| A�� | �����ӵİ뾶��Z��Y | |
| B�� | ��̬�⻯���ȶ��ԣ�Z��Y��X | |
| C�� | �������ˮ��������ԣ�Z��Y | |
| D�� | Ԫ��W��X��Z������ߺ���ͻ��ϼ۵Ĵ����ͷֱ�Ϊ0��2��6 |
| A�� | 92.37% | B�� | 9.2% | C�� | 50% | D�� | 25% |
| ���� | �� | �� | �� |
| ��Ӧ���Ͷ���� | 3molH2��2molN2 | 6molH2��4molN2 | 2molNH3 |
| �ﵽƽ���ʱ��/min | 5 | 8 | |
| ƽ��ʱN2��Ũ��/mol•L-1 | c1 | 1.5 | |
| NH3��������� | W1 | W3 | |
| ���������ܶ�/g•L-1 | ��1 | ��2 |
| A�� | �������з�Ӧ�ӿ�ʼ����ƽ��ķ�Ӧ����Ϊv��H2��=1.5mol•L-1•min-1 | |
| B�� | 2c1��1.5 | |
| C�� | 2��1=��2 | |
| D�� | �ڸ��¶��¼������з�Ӧ��ƽ�ⳣ��K=$\frac{��2-2{c}_{1}��^{2}}{{c}_{1}��3{c}_{1}-1.5��^{3}}$ |
| A�� | Ũ����ϡ��ʱ��Ӧ��ˮ��������������Ũ�����У��������ò��������� | |
| B�� | Ũ����ֽ⣬���ӷ������Կ��Գ��ڷ����ڿ����� | |
| C�� | Ũ������������ԣ�ϡ������������ | |
| D�� | �������������۳����������Ũ���� |