��Ŀ����
��һ�������Ͻ𣬽����������������У��ټ����������������Һ���ڿ����о��������ɫ������������Ϊֹ���������˳������������أ��õ����������������ԭ�Ͻ��������ͬ����
��1���Ͻ�������������������Һ�к��е��������� �� �� ��
��2�������˳������������أ��õ���������Ļ�ѧʽΪ ��
��3���������˳������õ���Һ�д��ڵ��������� �� �� ��
��4���Ͻ���������������Ϊ ��
��1���Ͻ�������������������Һ�к��е���������
��2�������˳������������أ��õ���������Ļ�ѧʽΪ
��3���������˳������õ���Һ�д��ڵ���������
��4���Ͻ���������������Ϊ
���㣺���Ļ�ѧ����,���Ļ�ѧ����,�йػ���ﷴӦ�ļ���
ר�⣺Ԫ�ؼ��仯����
��������1�����������Ͻ������ᷴӦ�IJ������жϣ�
��2�������Ͻ��ĩ�������������У��������NaOH ��Һ������Ϊ������������ϴ�ӡ�������գ��õ�����ɫ��ĩΪ��������
��3����������������ǿ�Ӧ����ƫ�����Ƽ��������ƹ������жϣ�
��4�������Ͻ��ĩ�������������У��������NaOH ��Һ������Ϊ������������ϴ�ӡ�������գ��õ�����ɫ��ĩΪ���������������������ںϽ��������������ɿ�֪����������Ԫ�ص��������ںϽ��������������Դ������
��2�������Ͻ��ĩ�������������У��������NaOH ��Һ������Ϊ������������ϴ�ӡ�������գ��õ�����ɫ��ĩΪ��������
��3����������������ǿ�Ӧ����ƫ�����Ƽ��������ƹ������жϣ�
��4�������Ͻ��ĩ�������������У��������NaOH ��Һ������Ϊ������������ϴ�ӡ�������գ��õ�����ɫ��ĩΪ���������������������ںϽ��������������ɿ�֪����������Ԫ�ص��������ںϽ��������������Դ������
���
�⣺��1�������Ͻ𣬽��������������ᣬ������Ӧ�����Ȼ��������Ȼ��������������������������Һ�к��е�������ΪFe2+��Al3+��H+ �ʴ�Ϊ��Fe2+��Al3+��H+��
��2�������Ͻ��ĩ�������������У��������NaOH ��Һ������Ϊ������������ϴ�ӡ�������գ��õ�����ɫ��ĩΪ���������ʴ�Ϊ��Fe2O3��
��3����Ϊ����������ǿ�Ӧ����ƫ�����ƣ�������������ֹ������������õ���Һ�д��ڵ�������Cl-��OH-��AlO2-���ʴ�Ϊ��Cl-��OH-��AlO2-��
��4���������Ͻ��ĩ�������������У��������NaOH ��Һ������Ϊ������������ϴ�ӡ�������գ��õ�����ɫ��ĩΪ���������������������ںϽ��������������ɿ�֪����������Ԫ�ص��������ںϽ���������������Ͻ���Al����������������������������������=
��100%=30%��
�ʴ�Ϊ��30%��
��2�������Ͻ��ĩ�������������У��������NaOH ��Һ������Ϊ������������ϴ�ӡ�������գ��õ�����ɫ��ĩΪ���������ʴ�Ϊ��Fe2O3��
��3����Ϊ����������ǿ�Ӧ����ƫ�����ƣ�������������ֹ������������õ���Һ�д��ڵ�������Cl-��OH-��AlO2-���ʴ�Ϊ��Cl-��OH-��AlO2-��
��4���������Ͻ��ĩ�������������У��������NaOH ��Һ������Ϊ������������ϴ�ӡ�������գ��õ�����ɫ��ĩΪ���������������������ںϽ��������������ɿ�֪����������Ԫ�ص��������ںϽ���������������Ͻ���Al����������������������������������=
| 16��3 |
| 160 |
�ʴ�Ϊ��30%��
���������⿼��������йط�Ӧ���̼�����㣬������ѧ���ķ��������ͼ��������Ŀ��飬��ȷ�����ķ�Ӧ�ҳ��Ͻ���������Ĺ�ϵ�ǽ����Ĺؼ���ע����Ԫ�ص��غ㣬�ѶȲ���

��ϰ��ϵ�д�
 ��������һ���þ�ϵ�д�
��������һ���þ�ϵ�д� Сѧ��10����Ӧ����ϵ�д�
Сѧ��10����Ӧ����ϵ�д�
�����Ŀ
���ҹ����л����еĴ�����Ⱦ����Ҫ�ǣ�������
| A��O2��Cl2��HCl������ |
| B��HCl��SO2��N2���۳� |
| C��NH3��CO��CO2���� |
| D��SO2��NO2��CO���̳� |
25��ʱ��ˮ�ĵ���ﵽƽ�⣺H2O?H++OH-��H��0������������ȷ���ǣ�������
| A����ˮ�м���ϡ��ˮ��ƽ�������ƶ���c��OH-������ |
| B����ˮ�м������������������ƣ�c��H+������Kw��� |
| C����ˮ�м����Ȼ�����Һ��c��H+�����䣬n��H+����� |
| D����ˮ���ȣ�Kw����pH���� |
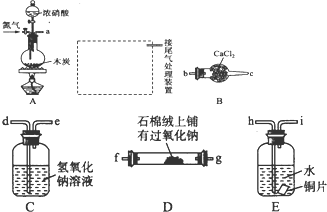
 ij������Ϊ̽������ɫ�����������壨FeC2O4?2H2O���Ļ�ѧ���ʣ�������һϵ��ʵ��̽����
ij������Ϊ̽������ɫ�����������壨FeC2O4?2H2O���Ļ�ѧ���ʣ�������һϵ��ʵ��̽����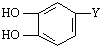 ������ȡ����-Y�в���֧��������̼̼˫����������������ת����ϵ��
������ȡ����-Y�в���֧��������̼̼˫����������������ת����ϵ��
 ����д��D�����ڷ��㴼������2��ͬ���칹����
����д��D�����ڷ��㴼������2��ͬ���칹����