��Ŀ����
12���������ƣ�NaClO2����һ��ǿ������Ư�����㷺���ڷ�֯��ӡȾ��ʳƷ��ҵ�����ڼ��Ի������ȶ����ڣ�ijͬѧ�������Ϻ��������NaClO2����Ҫ�������£�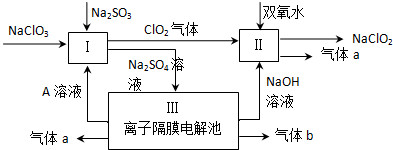
��1�����з�����Ӧ�Ļ�ԭ����Na2SO3���ѧʽ����
��2�����з�Ӧ�����ӷ���ʽ��2ClO2+H2O2+2OH-=2ClO2-+O2��+2H2O��
��3��A�Ļ�ѧʽ��H2SO4������b�Ļ�ѧʽ��H2��װ�â��з�����Ӧ�Ļ�ѧ����ʽ��2H2O$\frac{\underline{\;���\;}}{\;}$2H2��+O2����
��4��ClO2��һ�ָ�Чˮ�������������������ƺ�ϡ����Ϊԭ���Ʊ���5NaClO2+4HCl=5NaCl+4ClO2��+2H2O��
�ٸ÷�Ӧ���������ͻ�ԭ�������ʵ���֮����1��4��
���о�����������Ӧ��ʼʱ����Ũ��Խ�������������Cl2�ĺ���Խ������������ԭ��Ӧ���ɷ�����ԭ����ClO2-�������ԣ���Cl-�Ļ�ԭ�ԣ�����Һ�����Ժ�Ũ�ȵ��������ǿ��
���� ��������Һͨ�����Ӹ�Ĥ�����У�ClO2��˫��ˮ��II�з���������ԭ��Ӧ����NaClO2������a���÷�Ӧ��ClԪ�ػ��ϼ���+4�۱�Ϊ+3�ۣ���OԪ�ػ��ϼ���-1�۱�Ϊ0�ۣ��������ɵ�����a��O2�����ӷ�Ӧ����ʽΪ2 ClO2+H2O2+2OH-=2 ClO2-+O2��+2H2O��
III�з�����⣬����ͼ֪�������������ƣ���������������ͬʱ������������NaOH���������ɵ�����b��H2�����������������ӷŵ���������������a��O2��ͬʱ�������ᣬ����A��Һ��������������£�I��NaClO2��Na2SO3����������ԭ��Ӧ�����ӷ�Ӧ����ʽΪ2ClO3-+2H++SO32-=2ClO2��+SO42-+H2O��
��1��ͬһ��ѧ��Ӧ�У�ʧ���ӻ��ϼ۽��͵ķ�Ӧ���ǻ�ԭ����
��2�����������£�ClO2��H2O2����������ԭ��Ӧ����������ClO2-��ˮ��
��3��ͨ�����Ϸ���֪�����е��ˮ�������Ӹ�Ĥ�����A�����ᣬ�����������������ɣ�����������������
��4��5NaClO2+4HCl=5NaCl+4ClO2��+2H2O�У�5molNaClO2��1mol����������4mol����ԭ�����÷�Ӧ�����ԡ�Ũ��Ӱ�����ӵ����ʣ�
��� �⣺��1��I�з�Ӧ����ʽΪ2ClO3-+2H++SO32-=2ClO2��+SO42-+H2O��SԪ�صĻ��ϼ����ߣ���ԭ����Na2SO3���ʴ�Ϊ��Na2SO3��
��2������Ϊ���������£�ClO2��H2O2����������ԭ��Ӧ����������ClO2-��ˮ�����ӷ���ʽΪ2ClO2+H2O2+2OH-=2ClO2-+O2��+2H2O��
�ʴ�Ϊ��2ClO2+H2O2+2OH-=2ClO2-+O2��+2H2O��
��3��ͨ�����Ϸ���֪�������������Һ�����������������ӷŵ�����������ͬʱ�������ᣬ����A��H2SO4��������������������ͬʱ������������NaOH���������ɵ�����b��H2��װ�â��з�����Ӧ�Ļ�ѧ����ʽ��2H2O$\frac{\underline{\;���\;}}{\;}$2H2��+O2����
�ʴ�Ϊ��H2SO4��H2��2H2O$\frac{\underline{\;���\;}}{\;}$2H2��+O2����
��4����5NaClO2+4HCl=5NaCl+4ClO2��+2H2O�У�5molNaClO2��1mol����������4mol����ԭ�����÷�Ӧ���������ͻ�ԭ�������ʵ���֮����1��4���ʴ�Ϊ��1��4��
���о�����������Ӧ��ʼʱ����Ũ��Խ�������������Cl2�ĺ���Խ������������ԭ��Ӧ���ɷ�����ԭ����ClO2-�������ԣ���Cl-�Ļ�ԭ�ԣ�����Һ�����Ժ�Ũ�ȵ��������ǿ�����Cl-�������õ�Cl2��
�ʴ�Ϊ��ClO2-�������ԣ���Cl-�Ļ�ԭ�ԣ�����Һ�����Ժ�Ũ�ȵ��������ǿ��
���� ���⿼�����ʵ��Ʊ�ʵ�鷽����Ϊ��Ƶ���㣬��ȷI��II��III�з����ķ�Ӧ�����ʵ������ǽⱾ��ؼ���ע�ⳣ�����ʵ����ʼ����ӷ���ʽ����д�����ط�����ʵ�������Ŀ��飬�ۺ��Խ�ǿ����Ŀ�Ѷ��еȣ�

 ����5��2���ϵ�д�
����5��2���ϵ�д�| A�� | ����ͬһ����ѧ��Ӧ��һ����ɺͷּ�����ɣ��䷴Ӧ���ʱ䲻��ͬ | |
| B�� | ���ij��ѧ��Ӧ�ġ�H�͡�S��С��0����Ӧһ�����Է����� | |
| C�� | ��ѧ��Ӧ�����У��ϼ����ȣ��ɼ����� | |
| D�� | ��Ӧ������ʴ��ڷ�Ӧ���������ʱ����H��0 |
| A�� | Mg2+��Cl-��Al3+��SO42- | B�� | Ba2+��Na+��SO42-��MnO4- | ||
| C�� | K+��Cl-��HCO3-��CH3COO- | D�� | Cu2+��NH4+��NO3-��K+ |
| A�� | CuCl2 | B�� | FeCl2 | C�� | MgCl2 | D�� | Fe3O4 |
| A�� | ͬ��Ԫ�ص������ﲻ�����γɲ�ͬ���͵ľ��� | |
| B�� | SiO2������״�ṹ����С�Ļ��ϣ�Si��Oԭ����֮��Ϊ1��2 | |
| C�� | ����������ˮ���۵�ߣ����岻���磬�ۻ�ʱ����ľ���Ϊ���Ӿ��� | |
| D�� | ͬ���칹���У�ȼ������ֵԽ������Խ�ȶ� |
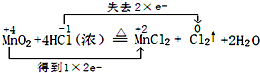 ��
��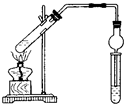 ��ͼ��ʾʵ��װ����ȡ�����������ش��������⣺
��ͼ��ʾʵ��װ����ȡ�����������ش��������⣺
 ��ͼ��ʾ���ڴ��Թ������3mL�Ҵ���2mL�����ᣬ�ٻ�������2mLŨ���ᣬ�ӱ�������һ֧�Թ��м��뱥��̼������Һ�������շ�Ӧ�����
��ͼ��ʾ���ڴ��Թ������3mL�Ҵ���2mL�����ᣬ�ٻ�������2mLŨ���ᣬ�ӱ�������һ֧�Թ��м��뱥��̼������Һ�������շ�Ӧ�����