��Ŀ����
̼���⡢��3��Ԫ����ɵ��л���A����Է�������Ϊ������51�����������������Ϊ9.8%��������ԭ�Ӹ���Ϊ����5����A��2����ͬ�ĺ��������ţ�һ�������£�A��������Ӧ����B��B���ӵĽṹ����Ϊ1��̼ԭ��������2����������2���ṹ��ͬ�Ļ��ţ�
��1��A�ķ���ʽ�� ��B�Ľṹ��ʽ ��
��2��д��������A������ͬ�����š�������֧����ͬ���칹��Ľṹ��ʽ�� �� ��
��3��A������һ������ͬ���칹�壬���칹��������������ˮ�⣬����������Է���������ͬ�Ļ��������һ�ֵķ�������2�������˷�Ӧ�Ļ�ѧ����ʽ�� ��
��1��A�ķ���ʽ��
��2��д��������A������ͬ�����š�������֧����ͬ���칹��Ľṹ��ʽ��
��3��A������һ������ͬ���칹�壬���칹��������������ˮ�⣬����������Է���������ͬ�Ļ��������һ�ֵķ�������2�������˷�Ӧ�Ļ�ѧ����ʽ��
���㣺�л�����ƶ�
ר�⣺�л���Ļ�ѧ���ʼ��ƶ�
�������л���A��Է�������Ϊ������51�������л���A����Է�������=51��2=102���������������Ϊ9.8%��������к��е�N��H��=
=10����������ԭ�Ӹ���Ϊ��������5������N��O��=
=5��N��C��=
=5�������л���A�ķ���ʽΪC5H10O2��һ�������£�A��һ��������Ӧ����B��B���ӵĽṹ����Ϊһ��̼ԭ�������������������������ṹ��ͬ�Ļ��ţ���BΪ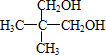 ����ӦΪ
����ӦΪ ��
��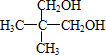 ����AΪ
����AΪ ���ݴ˽��
���ݴ˽��
| 102��9.8% |
| 1 |
| 10 |
| 2 |
| 102-10-16��2 |
| 12 |
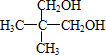 ����ӦΪ
����ӦΪ ��
��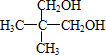 ����AΪ
����AΪ ���ݴ˽��
���ݴ˽�����
�⣺�л���A��Է�������Ϊ������51�������л���A����Է�������=51��2=102���������������Ϊ9.8%��������к��е�N��H��=
=10����������ԭ�Ӹ���Ϊ��������5������N��O��=
=5��N��C��=
=5�������л���A�ķ���ʽΪC5H10O2��һ�������£�A��һ��������Ӧ����B��B���ӵĽṹ����Ϊһ��̼ԭ�������������������������ṹ��ͬ�Ļ��ţ���BΪ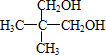 ����ӦΪ
����ӦΪ ��
��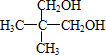 ����AΪ
����AΪ ��
��
��1��������������֪���л���A�ķ���ʽΪC5H10O2��BΪ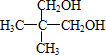 ��
��
�ʴ�Ϊ��C5H10O2�� ��
��
��2���� ������ͬ�����š�������֧����ͬ���칹��Ľṹ��ʽ�У�
������ͬ�����š�������֧����ͬ���칹��Ľṹ��ʽ�У�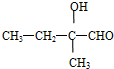 ��
��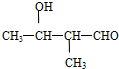 �ȣ�
�ȣ�
�ʴ�Ϊ�� ��
�� ��
��
��3��A�� ��������һ������ͬ���칹�壬���칹��������������ˮ�⣬����������Է���������ͬ�Ļ������Ϊ���������������һ�ֵķ�������2��������Ϊ2-������A��ͬ���칹��ΪCH3COOCH��CH3��2���˷�Ӧ�Ļ�ѧ����ʽ��CH3COOCH��CH3��2+H2O
��������һ������ͬ���칹�壬���칹��������������ˮ�⣬����������Է���������ͬ�Ļ������Ϊ���������������һ�ֵķ�������2��������Ϊ2-������A��ͬ���칹��ΪCH3COOCH��CH3��2���˷�Ӧ�Ļ�ѧ����ʽ��CH3COOCH��CH3��2+H2O
CH3COOH+��CH3��2CHOH��
�ʴ�Ϊ��CH3COOCH��CH3��2+H2O
CH3COOH+��CH3��2CHOH��
| 102��9.8% |
| 1 |
| 10 |
| 2 |
| 102-10-16��2 |
| 12 |
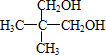 ����ӦΪ
����ӦΪ ��
��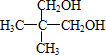 ����AΪ
����AΪ ��
����1��������������֪���л���A�ķ���ʽΪC5H10O2��BΪ
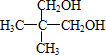 ��
���ʴ�Ϊ��C5H10O2��
 ��
����2����
 ������ͬ�����š�������֧����ͬ���칹��Ľṹ��ʽ�У�
������ͬ�����š�������֧����ͬ���칹��Ľṹ��ʽ�У� ��
�� �ȣ�
�ȣ��ʴ�Ϊ��
 ��
�� ��
����3��A��
 ��������һ������ͬ���칹�壬���칹��������������ˮ�⣬����������Է���������ͬ�Ļ������Ϊ���������������һ�ֵķ�������2��������Ϊ2-������A��ͬ���칹��ΪCH3COOCH��CH3��2���˷�Ӧ�Ļ�ѧ����ʽ��CH3COOCH��CH3��2+H2O
��������һ������ͬ���칹�壬���칹��������������ˮ�⣬����������Է���������ͬ�Ļ������Ϊ���������������һ�ֵķ�������2��������Ϊ2-������A��ͬ���칹��ΪCH3COOCH��CH3��2���˷�Ӧ�Ļ�ѧ����ʽ��CH3COOCH��CH3��2+H2O | ���� |
| �� |
�ʴ�Ϊ��CH3COOCH��CH3��2+H2O
| ���� |
| �� |
���������⿼���л������ʽ��ȷ�����Լ��л�������ŵĽṹ�����ʣ���Ŀ�Ѷ��еȣ������״���Ϊ��3����ע��ṹ��ʽ���ƶϣ�

��ϰ��ϵ�д�
 ��������һ���þ�ϵ�д�
��������һ���þ�ϵ�д� Сѧ��10����Ӧ����ϵ�д�
Сѧ��10����Ӧ����ϵ�д�
�����Ŀ
��1��д������Ũ���ᡢŨ������Һ��50��-60��ʱ��Ӧ�Ļ�ѧ����ʽ�� ��
��2��д����������������������Һ���ȵĻ�ѧ����ʽ�� ��
��2��д����������������������Һ���ȵĻ�ѧ����ʽ��
������ͨ��Ũ��ˮ�У��������������з�Ӧ��3Cl2+8NH3?H2O�T6NH4Cl+N2+8H2O���ڱ�״���£���1.12L Cl2��N2�Ļ�����壨90%Cl2��10%N2����Ϊ���������ͨ��Ũ��ˮ��ʵ�����ݳ��������Ϊ0.672L��������50%Cl2��50%N2�����˷�Ӧ�б�������NH3������Ϊ��������
| A��3.4 g |
| B��0.34 g |
| C��1.36 g |
| D��4.48 g |
���и�����������Һ�еķ�Ӧ�����ܷ�Ӧ�����Ķ��٣���ֻ����ͬһ�����ӷ���ʽ����ʾ���ǣ�������
| A��FeBr2��Cl2 |
| B��C6H5ONa��CO2 |
| C��HCl��Na2CO3 |
| D��Ca��HCO3��2��NaOH |

 ��
�� ��
�� ��
��
 ��ͼ��ʵ���������Լ���ǩ�ϵIJ������ݣ��ݴ��ж�
��ͼ��ʵ���������Լ���ǩ�ϵIJ������ݣ��ݴ��ж�