��Ŀ����
2���⺣��Ϊ����������Ӱ��������������Ӱ����Ӧ�ĸ�Σ���صIJ��ˣ��������Ի�����AΪԭ�Ϻϳɵ⺣����·�ߣ�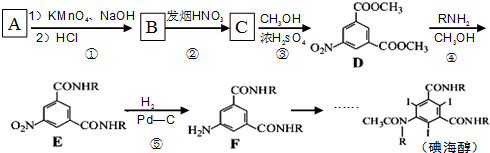
����R-Ϊ-CH2CH��OH��CH2OH
��ش��������⣺
��1������D�к��еĹ�����Ϊ������������
��2��A�DZ���ͬϵ���Է�����Ϊ106����A�Ľṹ��ʽ��
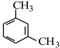 ��
����3��д��A�IJ�������������һ��ȡ�����������գ�
��4����Ӧ�ڵĻ�ѧ����ʽ
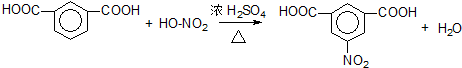 ��
����5����Ӧ�١����У�����������Ӧ���Ǣ٣�����ţ���
��6��д����ͬʱ��������������D������ͬ���칹��Ľṹ��ʽ��
 ��
��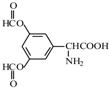 ��
����������3��ȡ�����������ϵ�һ±������2�֣�
���ܷ���������Ӧ��ˮ�����֮һ����FeCl3��Һ������ɫ��Ӧ��
����1����-������Ľṹ�����磺CH3CH��NH2��COOH���ڦ�-�����ᣩ
��7����д���ԡ�
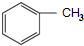 ���ͣ�CH3CO��2OΪԭ���Ʊ�Ⱦ���м��塡
���ͣ�CH3CO��2OΪԭ���Ʊ�Ⱦ���м��塡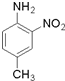 ���ĺϳ�·����
���ĺϳ�·������ͼ�����Լ����ã����ϳ�·������ͼʾ����
��֪����RNH2+��CH3CO��2O��CH3CONHR+CH3COOH
��CH3CONHR+NaOH��RNH2+CH3COONa��
��
 ���������ԣ��ױ�����
���������ԣ��ױ����� �ĺϳ�·������ͼ��
�ĺϳ�·������ͼ��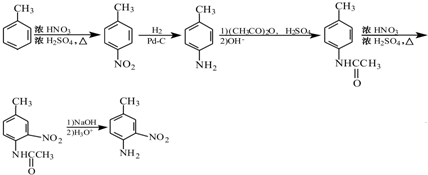 ��
��
���� ����D�Ľṹ��ʽ����C��״�����������Ӧ����D����CΪ ��B����������Ӧ����C������BΪ
��B����������Ӧ����C������BΪ ��A�����������������B����A�DZ���ͬϵ���������Է�������Ϊ106�����Ի�����A����ʽΪC8H10���Ƶ�A�Ľṹ��ʽΪ��
��A�����������������B����A�DZ���ͬϵ���������Է�������Ϊ106�����Ի�����A����ʽΪC8H10���Ƶ�A�Ľṹ��ʽΪ�� ��
��
��7���� �ͣ�CH3CO��2OΪԭ���Ʊ�Ⱦ���м���
�ͣ�CH3CO��2OΪԭ���Ʊ�Ⱦ���м��� ������Ҫ�����λ�������ٻ�ԭ�ɰ�����������
������Ҫ�����λ�������ٻ�ԭ�ɰ����������� �������ԣ��ױ����������������λ����ʱҪ�����������������ȼ�λ�������룬�ڽ�������ԭ��
�������ԣ��ױ����������������λ����ʱҪ�����������������ȼ�λ�������룬�ڽ�������ԭ��
��� �⣺����D�Ľṹ��ʽ����C��״�����������Ӧ����D����CΪ ��B����������Ӧ����C������BΪ
��B����������Ӧ����C������BΪ ��A�����������������B����A�DZ���ͬϵ���������Է���������106�����Ի�����A����ʽΪC8H10���Ƶ�A�Ľṹ��ʽΪ��
��A�����������������B����A�DZ���ͬϵ���������Է���������106�����Ի�����A����ʽΪC8H10���Ƶ�A�Ľṹ��ʽΪ�� ��
��
��1��D�й�����Ϊ�������������ʴ�Ϊ��������������
��2����������ķ�����֪A�Ľṹ��ʽΪ ���ʴ�Ϊ��
���ʴ�Ϊ�� ��
��
��3������ͬϵ���ڹ��յ����������������������ϵ�ȡ�����ʴ�Ϊ�����գ�
��4���÷�Ӧ����ʽΪ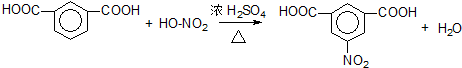 ��
��
�ʴ�Ϊ��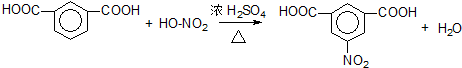 ��
��
��5�����ݣ�1���ķ�����Ӧ��Ϊ��������Ϊ��������ȡ����������������ȡ������ΪD�е�-OCH3��-NHRȡ������������������ԭ��Ӧ��
�ʴ�Ϊ���٣�
��6����������3��ȡ�����������ϵ�һ±������2�֣����ܷ���������Ӧ��ˮ�����֮һ����FeCl3��Һ������ɫ��Ӧ��˵�����з���������1����-������Ľṹ�������������D������ͬ���칹��Ľṹ��ʽΪ ��
�� ��
��
�ʴ�Ϊ�� ��
�� ��
��
��7���� �ͣ�CH3CO��2OΪԭ���Ʊ�Ⱦ���м���
�ͣ�CH3CO��2OΪԭ���Ʊ�Ⱦ���м��� ������Ҫ�����λ�������ٻ�ԭ�ɰ�����������
������Ҫ�����λ�������ٻ�ԭ�ɰ����������� �������ԣ��ױ����������������λ����ʱҪ�����������������ȼ�λ�������룬�ڽ�������ԭ��������Ϊ��
�������ԣ��ױ����������������λ����ʱҪ�����������������ȼ�λ�������룬�ڽ�������ԭ��������Ϊ�� ��
��
�ʴ�Ϊ�� ��
��
���� ���⿼���л��ϳɺ��л��ƶϣ�Ϊ��Ƶ���㣬���ؿ���ѧ�������ƶϼ�֪ʶ�ۺ�Ӧ����������ȷ�����л�������ż������ʹ�ϵ���л���Ӧ���ͼ���Ӧ�����ǽⱾ��ؼ����ѵ��ǣ�7����ϳ�·����ƣ���Ҫѧ���������ջ���֪ʶ��������ã���Ŀ�Ѷ��еȣ�

a���ڻ��Һ�м���6mol•L-1NaOH��Һ����pH=8Ϊֹ��
b�����˺�õ�������������ˮ���ϴ�ӳ�����
c����ϴ�Ӻ�ij����м���2mol•L-1�����ᣬ������Һ��pH��4-6��������У����ȹ��ˣ���Һ��ΪZnSO4��Һ��
d����Һ�м���2mol•L-1�����ᣬʹ��pH=2��
��֪�����������������������ʽ��ʼ��������ȫ����ʱ��Һ��pH���±����ش��������⣺
| ������ | Fe��OH��3 | Zn��OH��2 |
| pH | 1.5-3.2 | 6.4-8.0 |
��2������d�м������ᣬʹ��pH=2��Ŀ��������Zn2+��ˮ�⣻ Ҫ�Ƶ�ZnSO4•7H2O�IJ���d��ȱ�ٵIJ����ǽ���Һ����Ũ������ȴ�ᾧ��
���õ���Ҫ�����������������ƾ��ơ���������
| A�� | ��ϩʹ��ˮ��ɫ���Ҵ�ʹ���������Һ��ɫ | |
| B�� | ��֬��������Ӧ����ϩ���Ҵ� | |
| C�� | ����ˮ�⣻�Ҵ����������������� | |
| D�� | �����屽�����������ƻ����� |

| A�� | ��b������������Ӧˮ����ΪH2bO4����a���⻯��Ļ�ѧʽΪaH3 | |
| B�� | ��b�ĵ��ʿ����뵼����ϣ���c�ĵ��ʲ�����Ϊ�뵼����� | |
| C�� | ��b�ĵ�����H2���ϣ���c�ĵ�����H2������ | |
| D�� | a��b֮�������γ����ӻ����� |
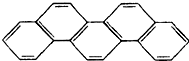 ��һ���л��������ư���ƥ���廷�죨��ͼ������ѧ�ҳ���Ϊ������ƥ�������������йء�����ƥ��������˵����ȷ���ǣ�������
��һ���л��������ư���ƥ���廷�죨��ͼ������ѧ�ҳ���Ϊ������ƥ�������������йء�����ƥ��������˵����ȷ���ǣ�������| A�� | ���л����������� | |
| B�� | �������������� | |
| C�� | �����ʷ�����ֻ���Ǽ��Լ� | |
| D�� | ��������ȫȼ�ղ���ˮ�����ʵ���С��CO2�����ʵ��� |
| A�� | ��ȥCuSO4��Һ�е�Fe��SO4��3����������CuO��ĩ����ֽ������ | |
| B�� | �ռС�մ���������������������θ����� | |
| C�� | ��ҵ�����У���������ͨ�����ʯ��ˮ�У���ȡƯ�� | |
| D�� | ���ܵ���ֱ����Դ�����������ɻ���ܵ���ʴ |
| A�� | O2ͨ���ữ��KI������Һ�� | |
| B�� | CO2ͨ��CaCl2��Һ�� | |
| C�� | 0.1mol•L-lϡH2SO4����0.1 mol•L-1Na2S2O3��Һ�� | |
| D�� | 0.2mol•L-1H2C2O4��Һ�����ữ��KMnO4��Һ�� |
| A�� | SO2���л�ԭ�ԣ�������Ư��ֽ�� | |
| B�� | ��������Һ�ʼ��ԣ�������ľ�ķ���� | |
| C�� | �������������ԣ�������ʴ�̲��� | |
| D�� | H2O2���л�ԭ�ԣ������������Ը��������Һ��Ӧ��ȡ�������� |
