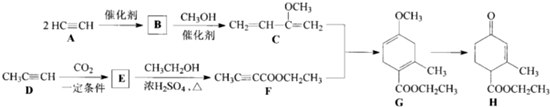
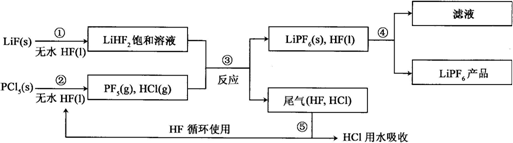
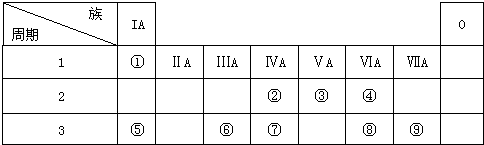
��Ŀ����
 ��ͼΪʵ����ijŨ�����Լ�ƿ�ϵı�ǩ���Ը����й����ݻش��������⣺
��ͼΪʵ����ijŨ�����Լ�ƿ�ϵı�ǩ���Ը����й����ݻش��������⣺��1����Ũ��������ʵ���Ũ��Ϊ
��2��ȡ����������ĸ�������Һʱ�������������в�����ȡ����Ķ��ٶ��仯����
A����Һ��HCl�����ʵ��� B����Һ��Ũ��
C����Һ��Cl-����Ŀ D����Һ���ܶ�
��3��ijѧ����������Ũ���������ˮ����500mL���ʵ���Ũ��Ϊ0.3mol/Lϡ���ᣮ
�ٸ�ѧ����Ҫ��ȡ
�ڿɹ�ѡ�õ������У��ٽ�ͷ�ιܣ�����ƿ�����ձ�����ҩ�ף�����Ͳ����������ƽ���߲�����������ϡ����ʱ����ȱ�ٵ�������
������ʱ������ȷ�IJ���˳���ǣ���ĸ��ʾ��ÿ����ĸֻ����һ�Σ�
A����30mLˮϴ���ձ�2-3�Σ�ϴ��Һ��ע������ƿ����
B������Ͳȷ��ȡ����Ũ���������������ر���ע��ʢ������ˮ��Լ30mL�����ձ��У��ò���������������ʹ���Ͼ���
C��������ȴ�������ز�����ע��500mL������ƿ��
D��������ƿ�ǽ����ߵ�ҡ��
E�����ý�ͷ�ιܼ�ˮ��ʹ��Һ��Һ��ǡ����̶�������
F������������ƿ��С�ļ�ˮ��ֱ��Һ��ӽ��̶���1-2cm��
�������ƹ����У�����ʵ������������Ƶ�ϡ��������ʵ���Ũ���к�Ӱ�죿
���ں������ƫ�ߡ���ƫ�͡�����Ӱ�족����
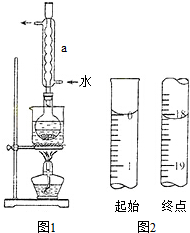
��1������ʱ���ӹ۲찼Һ��
��2����Һע������ƿǰû�лָ������¾ͽ��ж���
��3������ƿ������ϴ�Ӻ������������ˮ
��4�����ù����ձ���������δϴ��
���㣺����һ�����ʵ���Ũ�ȵ���Һ
ר�⣺���ʵ���Ũ�Ⱥ��ܽ��ר��
��������1���������ʵ���Ũ�ȵļ��㹫ʽc=
���м��㣻
��2�����ݸ����ļ��㹫ʽ�Ƿ�������й��жϣ�
��3���ٸ�������500mL���ʵ���Ũ��Ϊ0.3mol/Lϡ������Ҫ���Ȼ�������ʵ��������Ũ����������
�ڸ�������һ�����ʵ���Ũ�ȵ���Һ�IJ���ѡ��������
�۸���������Һʱ����������Ϊ����ȡ���ܽ⡢��Һ��ϴ�ӡ����ݡ��Ȳ�����������
�ܸ���c=
�����жϣ����nƫС��Vƫ����cƫС�����nƫ���VƫС����cƫ�ݴ˷�����
| 1000�Ѧ� |
| M |
��2�����ݸ����ļ��㹫ʽ�Ƿ�������й��жϣ�
��3���ٸ�������500mL���ʵ���Ũ��Ϊ0.3mol/Lϡ������Ҫ���Ȼ�������ʵ��������Ũ����������
�ڸ�������һ�����ʵ���Ũ�ȵ���Һ�IJ���ѡ��������
�۸���������Һʱ����������Ϊ����ȡ���ܽ⡢��Һ��ϴ�ӡ����ݡ��Ȳ�����������
�ܸ���c=
| n |
| V |
���
�⣺��1������ͼʾ�����ݣ��������Ũ��Ϊ��c=
=
mol/L=12mol/L��
�ʴ�Ϊ��12��
��2��A��n=c?V��������Һ��HCl�����ʵ�������Һ����йأ�
B����Һ��Ũ���Ǿ�һ�ȶ��ģ�����ȡ��Һ������أ�
C��N=n?NA=c?V?NA����������Һ����йأ�
D����Һ���ܶ��Ǿ�һ�ģ���������ȡ��Һ������أ�
��ѡ��BD��
��3����c1V1=c2V2��
12mol/L��V1=0.300mol?L-1��0.5L��
����V1=0.0125L=12.5mL
�ʴ�Ϊ��12.5��
������һ�����ʵ�����Ũ�ȵ���Һ��Ҫ�������У��ձ�����Ͳ�������������ж�����Ҫ��ͷ�ιܡ�����500mL��Һ��Ҫѡ��500mL����ƿ��
�ʴ�Ϊ����ͷ�ι� 500ml����ƿ��
��������Һʱ����������Ϊ����ȡ���ܽ⡢��Һ��ϴ�ӡ����ݡ��Ȳ�������ȷ��˳��Ϊ��BCAFED��
�ʴ�Ϊ��BCAFED��
�ܣ�1������ʱ���ӹ۲찼Һ�棬�������Ƶ���Һ���ƫС��Ũ��ƫ�ߣ��ʴ�Ϊ��ƫ�ߣ�
��2����Һע������ƿǰû�лָ������¾ͽ��ж��ݣ���ȴ����Һ�����ƫС�����Ƶ���Һ��Ũ��ƫ�ߣ��ʴ�Ϊ��ƫ�ߣ�
��3�����ڶ���ʱ����Ҫ��������ˮ����������ƿ������ƿ������ϴ�Ӻ������������ˮ��Ӱ�����ƽ�����ʴ�Ϊ����Ӱ�죻
��4�����ù����ձ���������δϴ�ӣ��������Ƶ���Һ�����ʵ����ʵ���ƫС�������Ƶ���ҺŨ��ƫ�ͣ��ʴ�Ϊ��ƫ�ͣ�
| 1000�Ѧ� |
| M |
| 1000��1.2��36.5% |
| 36.5 |
�ʴ�Ϊ��12��
��2��A��n=c?V��������Һ��HCl�����ʵ�������Һ����йأ�
B����Һ��Ũ���Ǿ�һ�ȶ��ģ�����ȡ��Һ������أ�
C��N=n?NA=c?V?NA����������Һ����йأ�
D����Һ���ܶ��Ǿ�һ�ģ���������ȡ��Һ������أ�
��ѡ��BD��
��3����c1V1=c2V2��
12mol/L��V1=0.300mol?L-1��0.5L��
����V1=0.0125L=12.5mL
�ʴ�Ϊ��12.5��
������һ�����ʵ�����Ũ�ȵ���Һ��Ҫ�������У��ձ�����Ͳ�������������ж�����Ҫ��ͷ�ιܡ�����500mL��Һ��Ҫѡ��500mL����ƿ��
�ʴ�Ϊ����ͷ�ι� 500ml����ƿ��
��������Һʱ����������Ϊ����ȡ���ܽ⡢��Һ��ϴ�ӡ����ݡ��Ȳ�������ȷ��˳��Ϊ��BCAFED��
�ʴ�Ϊ��BCAFED��
�ܣ�1������ʱ���ӹ۲찼Һ�棬�������Ƶ���Һ���ƫС��Ũ��ƫ�ߣ��ʴ�Ϊ��ƫ�ߣ�
��2����Һע������ƿǰû�лָ������¾ͽ��ж��ݣ���ȴ����Һ�����ƫС�����Ƶ���Һ��Ũ��ƫ�ߣ��ʴ�Ϊ��ƫ�ߣ�
��3�����ڶ���ʱ����Ҫ��������ˮ����������ƿ������ƿ������ϴ�Ӻ������������ˮ��Ӱ�����ƽ�����ʴ�Ϊ����Ӱ�죻
��4�����ù����ձ���������δϴ�ӣ��������Ƶ���Һ�����ʵ����ʵ���ƫС�������Ƶ���ҺŨ��ƫ�ͣ��ʴ�Ϊ��ƫ�ͣ�
���������⿼�������ʵ���Ũ�ȵ��йؼ��㼰����һ�����ʵ���Ũ�ȵ���Һ���������ѶȲ���Ҫע������һ�����ʵ���Ũ����Һ���������������������c=
������Ӱ���жϣ������仯�����������Ӷ�ȷ��Ũ�ȵı仯��
| n |
| V |

��ϰ��ϵ�д�
�����Ŀ
�����£����и���������ָ����Һ��һ���ܴ���������ǣ�������
| A������Cu��Ӧ�����������Һ��Na+��Fe2+��Cl-��SO42- |
| B����������Ժ�ɫ����Һ��K+��Cu2+��NO3-��I- |
| C����ˮ�������C��H+��?C��OH-���T10-24��Һ��K+��Na+��NO3-��Cl- |
| D����ɫ��Һ��K+��Al3+��ClO-��SO42- |
����������Ԫ��X��Y��Z��W��ԭ��������������Xԭ�ӵ����������������ڲ��������2����Y�ǵؿ��к�����ߵ�Ԫ�أ�Z2+��Y2-������ͬ�ĵ��Ӳ�ṹ��W��Xͬ���壬����˵����ȷ���ǣ�������
| A��ԭ�Ӱ뾶�Ĵ�С˳��r��W����r��Z����r��Y����r��X�� |
| B��Y�ֱ���Z��W�γɵĻ������л�ѧ��������ͬ |
| C��X������������Ӧ��ˮ��������Ա�W���� |
| D��Y����̬���⻯������ȶ��Ա�W��ǿ |
 Na2S2O3����Ҫ�Ļ���ԭ�ϣ�������ˮ�������Ի���Ի������ȶ���
Na2S2O3����Ҫ�Ļ���ԭ�ϣ�������ˮ�������Ի���Ի������ȶ���