��Ŀ����
 ���ǵ����ϼ�Ϊ�ḻ��Ԫ�أ�
���ǵ����ϼ�Ϊ�ḻ��Ԫ�أ���1���������Fe��SCN��2+�У��ṩ�չ�����ܹ¶Ե��ӵ������������ĵ����Ų�ʽΪ
��2��N��N�ļ���Ϊ942kJ?mol-1��N-N�����ļ���Ϊ247kJ?mol-1������˵��N2�е�
��3����CH3��3NH+��AlCl-4���γ����ӻ�����û����ﳣ����ΪҺ�壬��ӷ���һ����л��ܼ�
���������
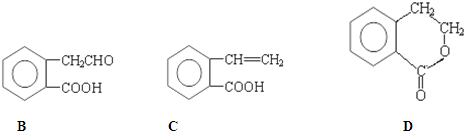
��4��X+�����е������ó���K��L��M�������Ӳ㣬����N3-�γɵľ���ṹ��ͼ��ʾ��X��Ԫ�ط�����
���㣺ԭ�Ӻ�������Ų�,�����ļ���
ר�⣺ԭ�������ṹר��,��ѧ���뾧��ṹ
��������1���������Fe��SCN��2+�У��ṩ�չ�����ܹ¶Ե��ӵ�����������Fe3+�������Ӻ�����23�����ӣ��������13�����ӣ�3d�ܼ�����5�����ӣ����ݹ���ԭ����д�����ӵ����Ų�ʽ��������������غϵķ���Ϊ�Ǽ��Է��ӣ�
��2��N��N�ļ���Ϊ942kJ?mol-1��N-N�����ļ���Ϊ247kJ?mol-1�����������Цм�����=
kJ/mol=347.5kJ/mol������ԽСԽ�ȶ���
��3�����Ӿ�����۷е�ϵ͡����Ӿ�����۷е�ϸߣ��۷е�ϵ͵�Һ���ӷ���
��4��X+�����е������ó���K��L��M�������Ӳ㣬������Ӻ��������=2+8+18=28������Xԭ�Ӻ��������Ϊ29��ΪCuԪ�أ���X+Ϊ���IJ����������и�ķ���ȷ����λ����
��2��N��N�ļ���Ϊ942kJ?mol-1��N-N�����ļ���Ϊ247kJ?mol-1�����������Цм�����=
| 942-247 |
| 2 |
��3�����Ӿ�����۷е�ϵ͡����Ӿ�����۷е�ϸߣ��۷е�ϵ͵�Һ���ӷ���
��4��X+�����е������ó���K��L��M�������Ӳ㣬������Ӻ��������=2+8+18=28������Xԭ�Ӻ��������Ϊ29��ΪCuԪ�أ���X+Ϊ���IJ����������и�ķ���ȷ����λ����
���
�⣺��1���������Fe��SCN��2+�У��ṩ�չ�����ܹ¶Ե��ӵ�����������Fe3+�������Ӻ�����23�����ӣ��������13�����ӣ�3d�ܼ�����5�����ӣ����ݹ���ԭ��֪�����ӵ����Ų�ʽΪ1s22s22p63s23p63d5��[Ar]3d5��SCN-�����Խṹ��HSCN����ΪV�νṹ����÷�������������IJ��غϣ�����Ϊ���Է��ӣ�
�ʴ�Ϊ��1s22s22p63s23p63d5��[Ar]3d5�����Է��ӣ�
��2��N��N�ļ���Ϊ942kJ?mol-1��N-N�����ļ���Ϊ247kJ?mol-1�����������Цм�����=
kJ/mol=347.5kJ/mol������ԽСԽ�ȶ�������N2�еĦҼ��Ȧм��ȶ����ʴ�Ϊ���ң��У�
��3�����Ӿ�����۷е�ϵ͡����Ӿ�����۷е�ϸߣ��۷е�ϵ͵�Һ���ӷ������ԣ�CH3��3NH+��AlCl-4�γɵ����ӻ�����ӷ���һ����л��ܼ�С���ʴ�Ϊ��С��
��4��X+�����е������ó���K��L��M�������Ӳ㣬������Ӻ��������=2+8+18=28������Xԭ�Ӻ��������Ϊ29��ΪCuԪ�أ���X+Ϊ���IJ����������и�ķ���֪������λ����6���ʴ�Ϊ��Cu��6��
�ʴ�Ϊ��1s22s22p63s23p63d5��[Ar]3d5�����Է��ӣ�
��2��N��N�ļ���Ϊ942kJ?mol-1��N-N�����ļ���Ϊ247kJ?mol-1�����������Цм�����=
| 942-247 |
| 2 |
��3�����Ӿ�����۷е�ϵ͡����Ӿ�����۷е�ϸߣ��۷е�ϵ͵�Һ���ӷ������ԣ�CH3��3NH+��AlCl-4�γɵ����ӻ�����ӷ���һ����л��ܼ�С���ʴ�Ϊ��С��
��4��X+�����е������ó���K��L��M�������Ӳ㣬������Ӻ��������=2+8+18=28������Xԭ�Ӻ��������Ϊ29��ΪCuԪ�أ���X+Ϊ���IJ����������и�ķ���֪������λ����6���ʴ�Ϊ��Cu��6��
���������⿼�����ʽṹ�����ʣ��漰ԭ�ӽṹ�����ܡ������Ų�ʽ����д�������ļ����֪ʶ�㣬�ۺ��Խ�ǿ����ȷԭ�ӽṹ�������ļ��㷽����֪ʶ�㼴�ɽ���ѵ�����λ���ļ��㷽����ע��ԭ��ʧ����˳��ԭ��ʧ����ʱ���Ǹ���������С�����Ǵ����ʧ���ӣ�Ϊ�״��㣮

��ϰ��ϵ�д�
�����Ŀ
����˵������ȷ���ǣ�������
| A��һ������������1L������ͨ��46gNO2����NO2 �����ʵ���Ũ��һ��Ϊ1mol/L |
| B�������22.4L�ļ�������20NAԭ�� |
| C��1mol��������ˮת��1mol e- |
| D��1molNa2O2������ˮ��Ӧ��ת��1mol e-������0.5molO2 |
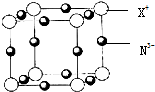
���й��ڵ�ⱥ��ʳ��ˮ��ȡ������˵����ȷ���ǣ�������

| A��ͨ��һ��ʱ��������������ĵ缫��Χ����Һ��ʹ��̪��� |
| B�������������ĵ缫������������ |
| C���븺�������ĵ缫������������ |
| D��Ϊ���������֮�䷢����Ӧ������ʯ��Ĥ�����ӽ���Ĥ�����۷ָ��������� |
 ��ͼ����MgCl2��AlCl3�����Һ�У��Ⱥ�����Լ�A��Bʱ���ó������ʵ���y��mol�� ���Լ����V��mL����Ĺ�ϵͼ����ʼ�μ�6mL�Լ�A������μ�A���ɣ�֮��ĵ��Լ�B�����½�����ȷ���ǣ�������
��ͼ����MgCl2��AlCl3�����Һ�У��Ⱥ�����Լ�A��Bʱ���ó������ʵ���y��mol�� ���Լ����V��mL����Ĺ�ϵͼ����ʼ�μ�6mL�Լ�A������μ�A���ɣ�֮��ĵ��Լ�B�����½�����ȷ���ǣ�������| A��A������NaOH��B���������ᣬ��2cA=cB |
| B��ԭ���Һ�У�c��Al3+����c��Mg2+����c��Cl-��=1��2��7 |
| C��A������Ba��OH��2��B���������ᣬ��cA=2cB |
| D����A��B��ΪһԪǿ���һԪǿ���μ�7mL�Լ�A��ĵ��Լ�B |
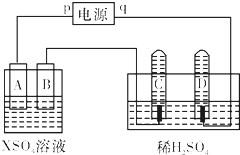 ��ͼ��p��qΪֱ����Դ��������A��+2�۽�������X�Ƴɣ�B��C��DΪ���缫����ͨ��Դ������X������B����ͬʱC��D���Ͼ��������ݣ��Իش�
��ͼ��p��qΪֱ����Դ��������A��+2�۽�������X�Ƴɣ�B��C��DΪ���缫����ͨ��Դ������X������B����ͬʱC��D���Ͼ��������ݣ��Իش�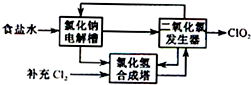 �������ȣ�ClO2����һ�ָ�Ч�����ס���ȫ��ɱ������������
�������ȣ�ClO2����һ�ָ�Ч�����ס���ȫ��ɱ������������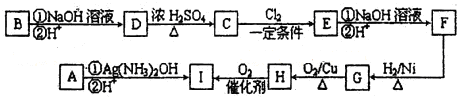
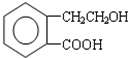 ������ͨ����ͬ��ѧ��Ӧ�ֱ��Ƶ�B��C��D �������ʣ�
������ͨ����ͬ��ѧ��Ӧ�ֱ��Ƶ�B��C��D �������ʣ�