��Ŀ����
���к͵ζ����ⶨ�ռ�Ĵ��ȣ����ռ��в��������ᷴӦ�����ʣ��Ը���ʵ��ش�
��1��ȷ��ȡ�ռ���Ʒ5.0g������Ʒ���250mL�Ĵ���Һ��
��2��ȡ10.00mL����Һ���� ��ȡע����ƿ�У�����������
��3����0.2000mol/L��������Һ�ζ������ռ���Һ���ζ�ʱ������ת��ʽ�ζ��ܵIJ������������ֲ�ͣ��ҡ����ƿ������ע�� ��ֱ���ζ��յ㣮
��4���������вⶨ���ݣ������õ��������ݣ���������ռ���Һ��Ũ�ȣ� ��
��5�����������ⶨ���ݣ������õ��������ݣ������ռ�Ĵ��� ��
��1��ȷ��ȡ�ռ���Ʒ5.0g������Ʒ���250mL�Ĵ���Һ��
��2��ȡ10.00mL����Һ����
��3����0.2000mol/L��������Һ�ζ������ռ���Һ���ζ�ʱ������ת��ʽ�ζ��ܵIJ������������ֲ�ͣ��ҡ����ƿ������ע��
��4���������вⶨ���ݣ������õ��������ݣ���������ռ���Һ��Ũ�ȣ�
| �ζ����� | ����Һ���/mL | ���������/mL | |
| �ζ�ǰ������mL�� | �ζ��������mL�� | ||
| ��һ�� | 10.00 | 0.50 | 20.40 |
| �ڶ��� | 10.00 | 4.00 | 24.10 |
| ������ | 10.00 | 4.20 | 25.70 |
���㣺�к͵ζ�
ר�⣺ʵ����
��������2�����ݼ�����Һʢ���ڼ�ʽ�ζ����У�
��3�����ݵζ�ʱ������ע����ƿ����Һ��ɫ�ı仯��
��4���ȸ������ݷ�����ȥ���̫������ݣ��پ�c�����⣩=
�����c�����⣩��
��5����c�����⣩���m=cVM����250mL�ռ���Ʒ���������Ƶ��������ٸ�������������ʽ�����������Ƶ�����������
��3�����ݵζ�ʱ������ע����ƿ����Һ��ɫ�ı仯��
��4���ȸ������ݷ�����ȥ���̫������ݣ��پ�c�����⣩=
| c(��)��V(��) |
| V(����) |
��5����c�����⣩���m=cVM����250mL�ռ���Ʒ���������Ƶ��������ٸ�������������ʽ�����������Ƶ�����������
���
�⣺��2������Һ�Ǽ�����Һ��Ӧʢ���ڼ�ʽ�ζ����У��ʴ�Ϊ����ʽ�ζ��ܣ�
��3���ζ�ʱ������ע����ƿ����Һ��ɫ�ı仯���Ա�ȷ�ж��յ�ĵ���ʴ�Ϊ����ƿ����Һ��ɫ�ı仯��
��4����3��ʵ�����̫��V������=[��20.40-0.50��+��24.10-4.00��]mL��2=20.00mL��
c�����⣩=
=
=0.4000mol?L-1��
�ʴ�Ϊ��0.4000mol?L-1��
��5����m=cVM=0.4000mol?L-1��0.25L��40g/mol=4.00g���أ�NaOH��=
��100%=80%���ʴ�Ϊ��80%��
��3���ζ�ʱ������ע����ƿ����Һ��ɫ�ı仯���Ա�ȷ�ж��յ�ĵ���ʴ�Ϊ����ƿ����Һ��ɫ�ı仯��
��4����3��ʵ�����̫��V������=[��20.40-0.50��+��24.10-4.00��]mL��2=20.00mL��
c�����⣩=
| c(��)��V(��) |
| V(����) |
| 0.2000mol/L��20.00mL |
| 10.00mL |
�ʴ�Ϊ��0.4000mol?L-1��
��5����m=cVM=0.4000mol?L-1��0.25L��40g/mol=4.00g���أ�NaOH��=
| 4.00g |
| 5.0g |
������������Ҫ�������к͵ζ������Ļ������輰�ռ�Ĵ��ȵļ��㣬�Ѷ��еȣ�ע��֪ʶ�Ļ��ۣ�

��ϰ��ϵ�д�
 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д�
�����Ŀ
������ǯ��סһС��������ִ��������������ھƾ����ϼ������ۻ��������ۻ������������䣮���й�������ʵ������Ľ�����ȷ���ǣ�������
| A��������������Ӧ |
| B�����ڿ������ܺܿ��γ�����Ĥ |
| C�����������۵�������۵�� |
| D�����ᴦ��ʱ��������ܽ������� |
�����йذ����ӵ�������NA����˵��������ǣ�������
| A�����³�ѹ��28��N2������ԭ����ĿΪ2NA |
| B����״����0.5molH2O���е�ԭ����ĿΪ1.5NA |
| C���κ�������0.5mol�������ӵķ�����һ����0.5NA |
| D��49��H2SO4�к��е�ԭ����ĿΪ1.5NA |
̼����������������ǶԴ�����ò�ͬ���෨�ķ��࣬������ȷ���ǣ�������
��1���� ��2���������� ��3������ ��4��̼���Σ�
��1���� ��2���������� ��3������ ��4��̼���Σ�
| A����1����2����3�� |
| B����1����3����4�� |
| C����1����2����4�� |
| D����2����3����4�� |
����ʵ�������ȷ���ǣ�������
| A���ζ�ʱ�����ֿ��Ƶζ��ܻ����������ճ���ƿ���ߵα����۾�ע�ӵζ����е�Һ�� |
| B���ⶨij��ɫ��Һ��pHֵʱ���ø���Ľྻ������պȡ��Һ������pH��ֽ�ϣ�������ɫ���Ƚ� |
| C���ζ���ϴ��������ˮ��ϴ������ע���Һ���еζ� |
| D����ij����Һ�м����������Ʋ����ȣ���ʪ�����ɫʯ����ֽ��������죬˵��ԭ��Һ��NH4+ |

 ������������ͼ��
������������ͼ��
 ��-X��-YΪȡ�������Ƕ��ǻ�������������ͬ���칹�����ܷ���������Ӧ����-X�Ľṹ��ʽ������
��-X��-YΪȡ�������Ƕ��ǻ�������������ͬ���칹�����ܷ���������Ӧ����-X�Ľṹ��ʽ������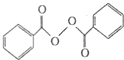 ����ȥ���������������������Ŀǰ�ѱ����ã�
����ȥ���������������������Ŀǰ�ѱ����ã�
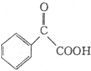 �����̣�ʾ�����£�
�����̣�ʾ�����£�