��Ŀ����
7����һ��ɫ�����ĩ������̼���ơ��������ơ��������е�һ�ֻ���֣�ij��ȤС����Ը���Ʒ����̽������1��ȡ������Ʒ�ܽ���ˮ���ܽ����õIJ����������ձ�����������
��2�����Ҫ�������Ʒ�Ƿ��������ƣ�����ķ������£�ȡ������1������Һ�����������ᣬ��ַ�Ӧ�������������ټ�������BaCl2��Һ�����г�������˵����Ʒ����Na2SO4������������˵��˵����Ʒ��û��Na2SO4����д���Լ��������ۣ�
��3��ȡ����������Ʒ������������У�������ų�����Ҫ����ų��������ɣ����������Լ���ѡ����ʵ��Լ����һ�����ʵ������û���������ɣ���Ҫд��ʵ�鷽��������ͽ��ۣ�
���Լ���Ʒ����Һ������ʯ��ˮ������KMnO4��Һ����������Ũ��Һ������NaHCO3��Һ��
�������������ͨ��ʢ��Ʒ����Һ������KMnO4��Һ��Ʒ����Һ������ʯ��ˮ��ϴ��ƿ�����ɲ������գ�
���� һ��ɫ�����ĩ������̼���ơ��������ơ��������е�һ�ֻ���֣�����ˮ���ȼ����ᣬ��μ��Ȼ�����Һ�����ɰ�ɫ������֤�������ƵĴ��ڣ�������Ʒ������������У�������ų�������Ϊ������̼�������������Ʒ������������ȥ�������������ʯ��ˮ���������̼���Դ������
��� �⣺��1��ȡ������Ʒ�ܽ���ˮ�����ձ����ܽ⣬�����Ͻ��裬�ܽ����õIJ����������ձ������������ʴ�Ϊ���ձ�����������
��2���������Ʒ�Ƿ��������ƣ�����Ϊȡ������1������Һ�����������ᣬ��ַ�Ӧ�������������ټ�������BaCl2��Һ�����г�������˵����Ʒ����Na2SO4������������˵��˵����Ʒ��û��Na2SO4��
�ʴ�Ϊ�����������ᣬ��ַ�Ӧ�������������ټ�������BaCl2��Һ�����г�������˵����Ʒ����Na2SO4������������˵��˵����Ʒ��û��Na2SO4��
��3��������Ʒ������������У�������ų�������Ϊ������̼�������������Ʒ������������ȥ�������������ʯ��ˮ���������̼�������������ͨ��ʢ��Ʒ����Һ������KMnO4��Һ��Ʒ����Һ������ʯ��ˮ��ϴ��ƿ��Ʒ����Һ��ɫ��˵������������ԭ��������������ƣ���֮���������ʯ��ˮ����ǣ�˵����̼���ƣ���֮������
�ʴ�Ϊ��Ʒ����Һ������KMnO4��Һ��Ʒ����Һ������ʯ��ˮ��Ʒ����Һ��ɫ��˵������������ԭ��������������ƣ���֮���������ʯ��ˮ����ǣ�˵����̼���ƣ���֮������
���� ���⿼��������ɵ��ƶϣ�Ϊ��Ƶ���㣬���ճ������ӵļ��鷽��������ʱ��������۵Ĺ�ϵΪ���Ĺؼ������ط�����ʵ�������Ŀ��飬��Ŀ�ѶȲ���

 ȫ�ܲ����ĩС״Ԫϵ�д�
ȫ�ܲ����ĩС״Ԫϵ�д�| A�� | A��CH2=CH2 B��CH3CHO C��CH3CH2OH | |
| B�� | A��CH3CHO B��CH2=CH2C��CH3CH2OH | |
| C�� | A��CH��CH B��CH3CH2OH C��CH3CHO | |
| D�� | A��CH3CH2OH B��CH3-CH3C��CH��CH |
| A�� | �Ƶ��۵�ϵ� | B�� | �Ƶ��ܶȽ�С | ||
| C�� | �Ƶ�ʧ����������ǿ | D�� | �Ƶĵ����� |
| A�� | ��ˮ�� | B�� | ��ˮ�� | C�� | ǿ������ | D�� | ���� |
| A�� | �����£�0.1 mol/L Na2S��Һ�д��ڣ�c��OH-��=c��H+��+c��HS-��+c��H2S�� | |
| B�� | �����£�pHΪ1��0.1 mol/L HA��Һ��0.1 mol/L NaOH��Һǡ����ȫ��Ӧʱ����Һ��һ�����ڣ�c ��Na+��=c��A-����c��OH-��=c��H+�� | |
| C�� | �����£�pH=7��CH3COONa��CH3COOH�����Һ��c��Na+��=0.1 mol/L��c��Na+��=c��CH3COOH����c��CH3COO-����c��H+��=c��OH-�� | |
| D�� | �����£���0.1 mol/L CH3COOH��Һ��ˮϡ�ͣ�����Һ��pH��3.0����4.0ʱ����Һ��Kac��H+��ֵ��С��ԭ����$\frac{1}{10}$ |
| A�� | ��״���£�11.2L��O2��NO�Ļ���ﺬ�еķ�����ԼΪ0.5��6.02��1023 | |
| B�� | 1mol���ǻ���1 mol������������������������Ϊ9 NA | |
| C�� | ���³�ѹ��42g ��ϩ�Ͷ�ϩ��������У�̼�������ĿΪ6NA | |
| D�� | 6.4g SO2��3.2g������Ӧ����SO3��ת�Ƶ�����Ϊ0.2NA |
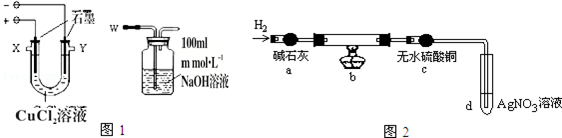
��1��������W��Ӧ�������X���ӣ��X����Y������
��2��ʵ���������̼���ϳ��˸����к�ɫ���ʣ���������������ɫ���ʣ�����������ʾ��
| �������Ƽ���ѧʽ | �Ȼ���ͭCuCl | ��ʽ�Ȼ�ͭCu2��OH��3Cl |
| ���� | ��ɫ���塢����ˮ | ��ɫ���塢����ˮ |
�ں�ɫ���ʿ�����Cu������Cu2O�����߶�����
ʵ���Ϊ̽������̼���ϸ��ŵĺ�ɫ����ɫ���ʣ����������ʵ�飺ȡ������̼����ϴ�ӡ������������ΪW1g�����������ͼ2��ʾװ��b�У�����ʵ�飮
ʵ���У�̼���ϵİ�ɫ������ȫ��Ϊ��ɫ����ˮ����ͭ����ɫ��d�г��ְ�ɫ������ʵ�����ʱ������ͨH2ֱ��̼����ȴ����������ΪW2g��
��3����ˮ����ͭ�������Ǽ����ɫ����������Cu2O��
��4��װ��b�з�����Ӧ�Ļ�ѧ����ʽ��2CuCl+H2=2Cu+2HCl��
��5�����CuCl2��Һʱ�������ϲ�����ɫ���ʵ�ԭ��Ϊ���õ缫��Ӧʽ���ͣ�Cu2++e-+Cl-=CuCl���������ϲ�����ɫ���ʵ����ʵ�����$\frac{W{\;}_{1}-W{\;}_{2}}{35.5}$mol��
ʵ���ⶨƯ��Һ��NaClO��Ũ�ȣ�ȷ��ȡ20.00mLϴ��ƿ�ڻ����Һ������������H2O2��Һ��ҡ�ȣ��μ�2��3�η�̪��Һ���� n mol•L-1����ζ����յ㣬��������V mL��
��6���û�ѧ����ʽ��ʾ����H2O2��Һ������NaClO+H2O2=O2��+NaCl+H2O��
��7��������ϴ��ƿ�ڵĸ���Ӧ������仯��Ư��Һ��NaClO��Ũ��Ϊ$\frac{20m-nV}{40}$mol•L-1��