��Ŀ����
7������ʯ�ǹ�ҵ��������Ҫԭ��֮һ������Ҫ�ɷ�Ϊ����������������в�����Ԫ�غ���Ԫ�أ������ʲ���H2SO4��Ӧ����ij�о���ѧϰС���ij����ʯ������������Ļ�ѧʽ����̽����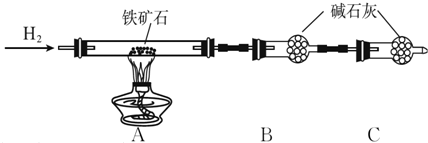
��1��������ʯ�к������IJⶨ
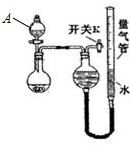
�ٰ���ͼ��װ���������װ�õ������ԣ�
�ڽ�5.0 g����ʯ����Ӳ�ʲ������У�װ��B��C�е�ҩƷ��ͼ��ʾ���г�������ʡ�ԣ���
�۴���˵����ܿڴ����ϵػ���ͨ��H2����Cװ�ó��ڴ�H2�鴿��ȼA���ƾ���
�ܳ�ַ�Ӧ�����ƾ��ƣ��ٳ���ͨ����������ȫ��ȴ��
��1��װ��C������Ϊ��ֹ�����е�ˮ������CO2����B�У�Ӱ��ⶨ�����
��2����ķ�Ӧ��װ��B����1.35 g��������ʯ�����İٷֺ���Ϊ24%��
������ʯ�к������IJⶨ
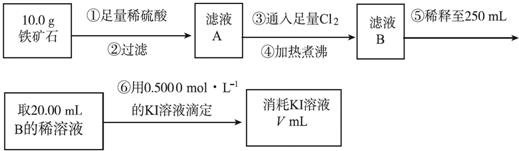
��1�����������е������Ǹ�����Һ���ܽ�Ĺ�����Cl2��
��2����������õ��IJ����������ձ�������������ͷ�ιܡ�250mL����ƿ��
��3�������йز���IJ�����˵����ȷ����df��
a����Ϊ��ˮΪ��ɫ�����Եζ������в����ָʾ��
b���ζ������п����õ�����Һ��Ϊָʾ��
c���ζ���������ˮϴ�Ӻ����ֱ��װҺ
d����ƿ����Ҫ�ô���Һ��ϴ
e���ζ������У��۾�ע�ӵζ�����Һ��仯
f���ζ�������30s����Һ���ָ�ԭ������ɫ�ٶ���
��4�����ζ�����������0.5000mol?L-1KI��Һ20.00mL��������ʯ�����İٷֺ���Ϊ70%��
���ɢ���������������ʯ������������Ļ�ѧʽΪFe5O6��
���� ��1��B�еļ�ʯ���������û���Ӧ���ɵ�ˮ�ģ�Ϊ�˷�ֹ�����ɷֶ�ʵ���Ӱ�죬Ҫ��һ��װ�����տ����е�ˮ�Լ�������̼��
��2����Ӧ��װ��B����1.35g������������������Ӧ���������������ֵ�����Ը��ݲ����������㣻
��1����п��Խ�ˮ�е�������ߣ�
��2������ϡ��Һ�������һ���������Һ��ѡ����������ش�
��3�����ݵζ������Լ��ζ������е�ʵ��������֪ʶ���ش��жϣ�
��4������Ԫ���غ�ͻ�ѧ��Ӧ����ʽ���м��㣻
������Ԫ��������������Ԫ�������������������������Ļ�ѧʽ��
��� �⣺��1����ʵ���У���������������Ӧ���ɽ�������ˮ�����ݹ��������ı仯���������ĺ�����B���ĸ�������������ղ�����ˮ����������Cװ��Ҫ��ֹ��ֹ�����е�ˮ������CO2����B�У�Ӱ��ⶨ�����
�ʴ�Ϊ����ֹ�����е�ˮ������CO2����B�У�Ӱ��ⶨ�����
��2����ķ�Ӧ��װ��B����1.35g�����ݷ�Ӧ��ʵ�ʣ����ӵ���ˮ�����������������غ㶨�ɣ���������ʯ�����İٷֺ����ǣ�$\frac{\frac{1.35}{18}��16}{5.0}$��100%=24%��
�ʴ�Ϊ��24%��
��1��������ʯ�м������ᣬ����Ӧ���������������Һ�������ڹ�����������Һ�������������������к���Խ����������ܽ�ȣ�������Һ���ܽ�Ĺ�����Cl2��
�ʴ�Ϊ��������Һ���ܽ�Ĺ�����Cl2��
��2����ԭ��Һϡ�͵�250mL����Ҫʹ�õIJ������������У��ձ�������������ͷ�ιܡ�250mL����ƿ����ȱ��250mL����ƿ��
�ʴ�Ϊ��250mL����ƿ��
��3��a����ˮΪ��ɫ������������Ҳ�ǻ�ɫ��Һ���ζ����������ָʾ������a����
b���ζ������У����������Ժ͵����ӷ�����Ӧ�����������Ӻ͵ⵥ�ʣ��ⵥ������������Һ��ʾ��ɫ������ȷ���Ƿ�ﵽ�ζ��յ㣬��b����
c���ζ���������ˮϴ�Ӻ�����ñ�Һ��ϴ����c����
d����ƿ����Ҫ�ô���Һ��ϴ����d��ȷ��
e���ζ������У��۾�ע����ƿ����ɫ�ı仯����e����
f���ζ�������30s����Һ���ָ�ԭ������ɫ�ٶ�������f��ȷ��
�ʴ�Ϊ��df��
��4�����ݷ�Ӧ�ķ���ʽΪ2Fe3++2I-=2Fe2++I2��֪�����ĵĵ����������������ʵ�����ȣ�n��Fe3+��=n��KI��������0.4000mol•L-1��0.025L=c��Fe3+����0.02L�����c��Fe3+��=0.5mol•L-1��������Ԫ�صİٷֺ���Ϊ��$\frac{0.5mol/L��0.25L��56g/mol}{10g}$��100%=70%��
�ʴ�Ϊ��70%��
����������������70%����Ԫ�ص�����������24%������100g����ʯ�У���Ԫ�ص�������70g����Ԫ��������24g����Ԫ�غ���Ԫ�ص����ʵ�����Ϊ��$\frac{70}{56}$��$\frac{24}{16}$=5��6������������Ļ�ѧʽΪ��Fe5O6��
�ʴ�Ϊ��Fe5O6 ��
���� ���⿼����̽������ʯ����Ԫ�غ���Ԫ�صĺ����ķ����������Ǹ߿��еij������ͣ������е��Ѷȵ����⣬���ض�ѧ��������������ѵ��������������ѧ���淶�Ͻ���ʵ����ơ��������������������ۺ���ǿ�����ۺ�ʵ������ϵ���ܣ�Ҫ��ѧ���������桢ϸ�µ����⣬��ϵ��ѧ����֪ʶ�ͼ��ܣ�ȫ��ϸ�µ�˼�����ܵó���ȷ�Ľ��ۣ�

 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д�| A�� | 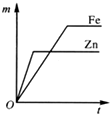 | B�� | 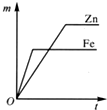 | C�� | 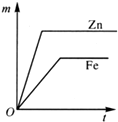 | D�� | 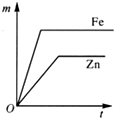 |
 þ������п�������г��������ֽ������������ϻ��������Ϣ��
þ������п�������г��������ֽ������������ϻ��������Ϣ����þ������п��������ɫ�Ľ���
��п��Zn��������NaOH��Һ��Ӧ����H2
��Zn��OH��2Ϊ��ɫ���壬������ˮ��������ǿ�NH3•H2O
��Zn2+���γ��������[Zn��NH3��4]2+�����������ǿ��ֽ�����Zn2+��NH4+��1����1����ͬѧȡþ���Ͻ���ж�����������ͼ��ʾװ�ý���ʵ�飬����������ݣ���������������ѻ���ɱ�״�������Ե���Һ����������������Ӱ�죩
| ��� | ��ĩ���� | �����ܵ�һ�ζ��� | �����ܵڶ��ζ��� |
| �� | 2.0 g | 10.0 mL | 346.2 mL |
| �� | 2.0 g | 10.0 mL | 335.0 mL |
| �� | 2.0 g | 10.0 mL | 345.8 mL |
�������Լ�����Ʒ��pH��ֽ��ϡ���ᡢNaOH��Һ����ˮ��
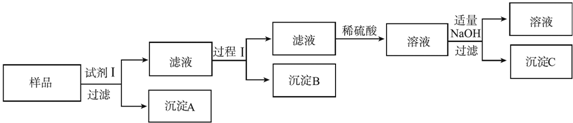
���Լ�����NaOH��Һ������B��Al��OH��3��
�ڹ��̢��ǣ�����Һ����μ���ϡ���ᣬֱ�����ɵij����պ��ܽ⣬�ټ���������ϡ��ˮ�����ˣ�
�۳���C�백ˮ��Ӧ�����ӷ���ʽΪZn��OH��2+4NH3=[Zn��NH3��4]2++2OH-����Zn��OH��2+4NH3•H2O=[Zn��NH3��4]2++2OH-+4H2O��
��A��B�õ������Ӻ�ͬʱ����ϡH2SO4��Һ�У�A��Ϊ������
��C��D�õ������Ӻ�ͬʱ����ϡH2SO4��Һ�У�������D�����ߡ�C��
��A��C������ͬʱ����ϡH2SO4��Һ�У�C���ϲ����������ݣ������������Ա仯��
��B��D�õ������Ӻ�ͬʱ����ϡH2SO4��Һ�У�D���Ϸ���������Ӧ��
�ݴˣ��ж����ֽ����Ļ��˳���ǣ�������
| A�� | A��B��C��D | B�� | A��C��D��B | C�� | C��A��B��D | D�� | B��D��C��A |
| A�� | Na | B�� | Mg | C�� | Al | D�� | Zn |
| A�� | CO2ͨ�����ʯ��ˮ�� | B�� | ϡ�����м���ͭƬ | ||
| C�� | ϡ�����м�����м | D�� | HCl��Һ����Na2CO3��Һ�� |
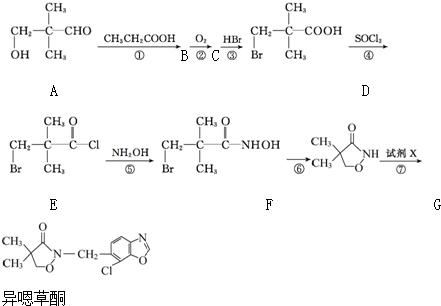
 ��
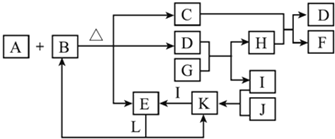
��
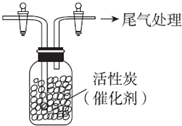 ijѧϰС������SO2���л�ԭ�ԣ��Ʋ�SO2�ܱ�Cl2��������SO2Cl2��
ijѧϰС������SO2���л�ԭ�ԣ��Ʋ�SO2�ܱ�Cl2��������SO2Cl2��