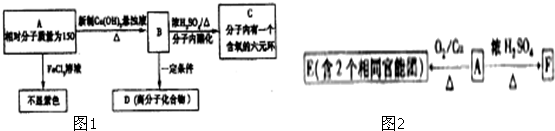
��Ŀ����
�״���Դ�ḻ���۸�������������淽�㣬��һ����Ҫ�Ļ���ԭ�ϣ�������Ҫ����;��Ӧ��ǰ����
��1����һ���¶��£���1molCO��3molH2�������Ϊ5L�ܱպ��������У�
������ӦCO��g��+2H2��g��?CH3OH��g����H��0���ﵽƽ�����CO�� ת����Ϊ25%���������ڵ�ѹǿ����ʼѹǿ֮��Ϊ ���÷�Ӧ�Ļ�ѧƽ�ⳣ�� ��
��2����ҵ�����ü״��Ʊ������ij��÷��������֣�
�ټ״��������������÷��е�һ����Ҫ��ӦΪCH3OH��g��??CO��g��+2��2��g�����˷�Ӧ���Է����е�ԭ���� ��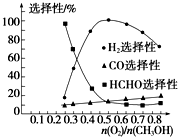
�ڼ״���������������һ���¶�����Ag/CeO2ZnOΪ����ʱԭ���������Է�Ӧ��ѡ���ԣ�ѡ����Խ��ʾ���ɵĸ�����Խ�ࣩӰ���ϵ��ͼ��ʾ����
=0.25ʱ��CH3OH��O2��������Ҫ��Ӧ����ʽΪ �����Ʊ�H2ʱ��ÿ���
= ��
��1����һ���¶��£���1molCO��3molH2�������Ϊ5L�ܱպ��������У�
������ӦCO��g��+2H2��g��?CH3OH��g����H��0���ﵽƽ�����CO�� ת����Ϊ25%���������ڵ�ѹǿ����ʼѹǿ֮��Ϊ
��2����ҵ�����ü״��Ʊ������ij��÷��������֣�
�ټ״��������������÷��е�һ����Ҫ��ӦΪCH3OH��g��??CO��g��+2��2��g�����˷�Ӧ���Է����е�ԭ����

�ڼ״���������������һ���¶�����Ag/CeO2ZnOΪ����ʱԭ���������Է�Ӧ��ѡ���ԣ�ѡ����Խ��ʾ���ɵĸ�����Խ�ࣩӰ���ϵ��ͼ��ʾ����
| n(O2) |
| n(CH3OH) |
| n(O2) |
| n(CH3OH) |
���㣺��ѧƽ��ļ���,��Ӧ�Ⱥ��ʱ�,��ѧƽ���Ӱ������
ר�⣺��ѧƽ��ר��
��������1�����ݻ�ѧƽ������ʽ��ʽ���㣬����ѹǿ֮�ȵ����������ʵ���֮�ȣ�ƽ�ⳣ��=
��
��2���ٷ�ӦΪCH3OH��g��?CO��g��+2H2��g�����˷�Ӧ���Է����е�ԭ���ǡ�H-T��S��0����Ӧ�������ӵķ�Ӧ��
�ڵ�n��O2��/n��CH3OH��=0.25ʱ��CH3OH��O2��������Ҫ��ӦΪ�״��Ĵ��������ɼ�ȩ����Ӧ�ķ���ʽΪ��2CH3OH+O2
2HCHO+2H2O������ͼ������Ʊ�H2ʱ��ÿ���
��
| ������ƽ��Ũ���ݴη��˻� |
| ��Ӧ��ƽ��Ũ���ݴη��˻� |
��2���ٷ�ӦΪCH3OH��g��?CO��g��+2H2��g�����˷�Ӧ���Է����е�ԭ���ǡ�H-T��S��0����Ӧ�������ӵķ�Ӧ��
�ڵ�n��O2��/n��CH3OH��=0.25ʱ��CH3OH��O2��������Ҫ��ӦΪ�״��Ĵ��������ɼ�ȩ����Ӧ�ķ���ʽΪ��2CH3OH+O2
| ���� |
| һ���¶� |
| n(O2) |
| n(CH3OH) |
���
�⣺��1�����ݻ�ѧƽ������ʽ��ʽ���㣬�ﵽƽ�����CO�� ת����Ϊ25%
CO��g��+2H2��g��?CH3OH��g��
��ʼ����mol�� 1 3 0
�仯����mol�� 0.25 0.5 0.25
ƽ������mol�� 0.75 2.5 0.25
����ѹǿ֮�ȵ����������ʵ���֮�ȣ������ڵ�ѹǿ����ʼѹǿ֮��=��0.75mol+2.5mol+0.25mol������1+3��=7��8��
ƽ�ⳣ��K=
=
=
��
�ʴ�Ϊ��7��8��
��
��2���ٷ�ӦΪCH3OH��g��?CO��g��+2H2��g�����˷�Ӧ���Է����е�ԭ���ǡ�H-T��S��0����Ӧ�������ӵķ�Ӧ��
�ʴ�Ϊ���÷�Ӧ��һ�������ķ�Ӧ�����S��0����
�ڵ�n��O2��/n��CH3OH��=0.25ʱ��CH3OH��O2��������Ҫ��ӦΪ�״��Ĵ��������ɼ�ȩ����Ӧ�ķ���ʽΪ��2CH3OH+O2
2HCHO+2H2O������ͼ�������ѡ����Խ��ʾ���ɵĸ�����Խ�࣬
=0.5�Ʊ�����ʱ������ѡ�����������������࣬�������Ʊ�H2ʱ��ÿ���
=0.5��
�ʴ�Ϊ��2CH3OH+O2
2HCHO+2H2O��0.5��
CO��g��+2H2��g��?CH3OH��g��
��ʼ����mol�� 1 3 0
�仯����mol�� 0.25 0.5 0.25
ƽ������mol�� 0.75 2.5 0.25
����ѹǿ֮�ȵ����������ʵ���֮�ȣ������ڵ�ѹǿ����ʼѹǿ֮��=��0.75mol+2.5mol+0.25mol������1+3��=7��8��
ƽ�ⳣ��K=
| ������ƽ��Ũ���ݴη��˻� |
| ��Ӧ��ƽ��Ũ���ݴη��˻� |
| ||||
|
| 4 |
| 3 |
�ʴ�Ϊ��7��8��
| 4 |
| 3 |
��2���ٷ�ӦΪCH3OH��g��?CO��g��+2H2��g�����˷�Ӧ���Է����е�ԭ���ǡ�H-T��S��0����Ӧ�������ӵķ�Ӧ��
�ʴ�Ϊ���÷�Ӧ��һ�������ķ�Ӧ�����S��0����
�ڵ�n��O2��/n��CH3OH��=0.25ʱ��CH3OH��O2��������Ҫ��ӦΪ�״��Ĵ��������ɼ�ȩ����Ӧ�ķ���ʽΪ��2CH3OH+O2
| ���� |
| һ���¶� |
| n(O2) |
| n(CH3OH) |
| n(O2) |
| n(CH3OH) |
�ʴ�Ϊ��2CH3OH+O2
| ���� |
| һ���¶� |
���������⿼���˻�ѧƽ������ʽ���������ͼ�������Ӧ�ã����ջ����ǹؼ�����Ŀ�Ѷ��еȣ�

��ϰ��ϵ�д�
 �ִʾ�ƪ��ͬ�����Ĵ��ϵ�д�
�ִʾ�ƪ��ͬ�����Ĵ��ϵ�д�
�����Ŀ
ͬ��ͬѹ�£���������CO��CO2������Ƚϣ�������������ȷ���ǣ�������
| A������̼ԭ������Ϊ1��1 |
| B�������Ϊ11��7 |
| C���ܶȱ�Ϊ7��11 |
| D����������Ϊ7��11 |
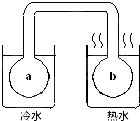 ��ͼ��ʾ���ܷ�˫����ϵ�г��к���ɫ����X���������ձ���ˮԡ����²����a��������ɫ��b���������ɫ�����������Һ��������X�����ǣ�������
��ͼ��ʾ���ܷ�˫����ϵ�г��к���ɫ����X���������ձ���ˮԡ����²����a��������ɫ��b���������ɫ�����������Һ��������X�����ǣ�������| A��NO2��g�� |
| B��Br2��g�� |
| C��NO��g�� |
| D��N2��g�� |
���и��������з�����һ����ͬ���ǣ�������
| A��11.2LH2��0.5molCO2 |
| B��18mLH2O��4�棩��2��105Pa��27��32gO2 |
| C��1molCO��88gCO2 |
| D��27�棬22.4LCl2�ͺ�4mol��ԭ�ӵ�H2SO4 |
��NA��ʾ�����ӵ�������ֵ������������ȷ���ǣ�������
| A���ڱ�״���£�11.2L ��������NA����ԭ�� |
| B��25�棬1.01��105Pa��64gSO2�к��е�ԭ����Ϊ3NA |
| C��11.2L CO���еķ�����Ϊ0.5NA |
| D��1mol�����Ӻ��еĵ�������1mol�������еĵ�����֮��Ϊ16��7 |
��a��b��c��d�ĸ������缫���йص�ʵ��װ�ü�����ʵ�����������ɴ˿��ж������ֽ����Ļ��˳���ǣ�������
| ʵ�� װ�� |  |  |  |  |
| ����ʵ ������ | a��������С��b���������� | b�������������c���ޱ仯 | d���ܽ⣬c����������� | ������a������d�� |
| A��a��b��c��d |
| B��b��c��d��a |
| C��d��a��b��c |
| D��a��b��d��c |