��Ŀ����
12��������J��ij�ֽ�����ʹҩ����AΪԭ�ϵĹ�ҵ�ϳ�·����ͼ��ʾ��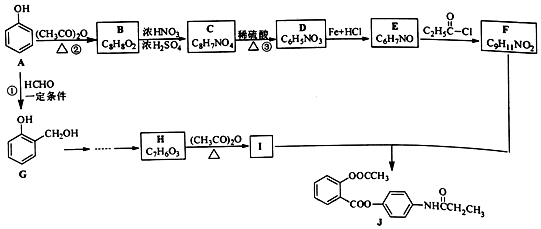
��֪
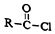 +R��-NH2��
+R��-NH2��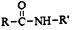 +HCl
+HClR-NO2$\stackrel{Fe/HCl}{��}$R-NH2
��ش�
��1��д��ͬʱ��������������F������ͬ���칹��Ľṹ��ʽ
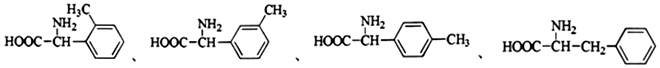 ��
������a-�������a̼���뺬����ԭ�ӣ�
��1H-NMR�ױ��������к��б�����
��2������˵������ȷ����D
A��������C���������������
B��������E�ȿ�����NaOH��Ӧ�ֿ�����HC1��Ӧ
C��������F����FeCl3������ɫ��Ӧ
D��������J�ķ���ʽΪC18H15NO5
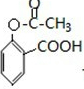
��3��д��I�Ľṹ��ʽ
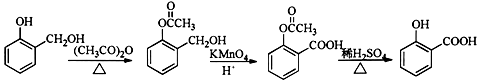
 ���ϳɹ�������Ʋ���ں͢۵�Ŀ���DZ������ǻ���������
���ϳɹ�������Ʋ���ں͢۵�Ŀ���DZ������ǻ�����������4�����G��H�ĺϳ�·�ߣ�������ͼ��ʾ�����Լ���ѡ��
 ��
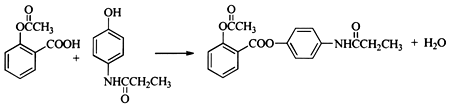
����5��д��I+F��J�Ļ�ѧ����ʽ
 ��
��
���� �ɷ�Ӧ�ٵ��������֪��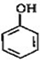 ��HCHO�����ӳɷ�Ӧ����
��HCHO�����ӳɷ�Ӧ����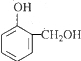 ������B�ķ���ʽ��֪����Ӧ��Ϊ
������B�ķ���ʽ��֪����Ӧ��Ϊ ������������ȡ������BΪ
������������ȡ������BΪ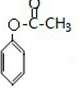 �����ݷ�Ӧ������֪��B����������Ӧ����CΪ
�����ݷ�Ӧ������֪��B����������Ӧ����CΪ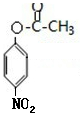 ��Cˮ������DΪ
��Cˮ������DΪ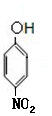 ������������Ϣ��֪��D������ԭ��Ӧ����EΪ
������������Ϣ��֪��D������ԭ��Ӧ����EΪ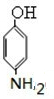 ��E������Ϣ�е�ȡ����Ӧ����FΪ
��E������Ϣ�е�ȡ����Ӧ����FΪ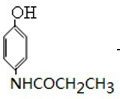 ������J��F�Ľṹ��ʽ��֪IΪ
������J��F�Ľṹ��ʽ��֪IΪ �����Կ��Է��Ƶ�HΪ
�����Կ��Է��Ƶ�HΪ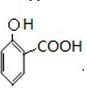 ����
���� ������������ȡ�����ɷ��������ø�������������ǻ�����������������ˮ��ɵ�
������������ȡ�����ɷ��������ø�������������ǻ�����������������ˮ��ɵ� ���ݴ˴��⣮
���ݴ˴��⣮
��� �⣺��1��FΪ ��������������a-�������a̼���뺬����ԭ�ӣ������а������Ȼ�����1H-NMR�ױ��������к��б�������ͬʱ����������F������ͬ���칹��Ľṹ��ʽΪ
��������������a-�������a̼���뺬����ԭ�ӣ������а������Ȼ�����1H-NMR�ױ��������к��б�������ͬʱ����������F������ͬ���칹��Ľṹ��ʽΪ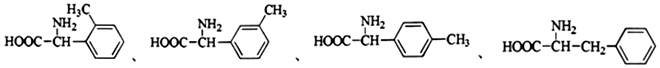 ��
��
�ʴ�Ϊ��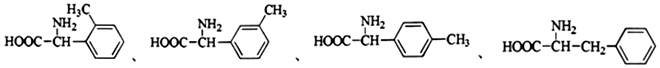 ��
��
��2��A��CΪ ��������C�к��������������֣���A��ȷ��
��������C�к��������������֣���A��ȷ��
B��EΪ ��E�ȿ�����NaOH��Ӧ�ֿ�����HC1��Ӧ����B��ȷ��
��E�ȿ�����NaOH��Ӧ�ֿ�����HC1��Ӧ����B��ȷ��
C��FΪ ���з��ǻ�������F����FeCl3������ɫ��Ӧ����C��ȷ��
���з��ǻ�������F����FeCl3������ɫ��Ӧ����C��ȷ��
D����J�Ľṹ��ʽ��֪��J�ķ���ʽΪC18H17NO5����D����
��ѡD��
��3����������ķ�����֪��I�Ľṹ��ʽΪ ���ϳɹ�������Ʋ���ں͢۵�Ŀ���DZ������ǻ�����������
���ϳɹ�������Ʋ���ں͢۵�Ŀ���DZ������ǻ�����������
�ʴ�Ϊ�� ���������ǻ���������
���������ǻ���������
��4���� ������������ȡ�����ɷ��������ø�������������ǻ�����������������ˮ��ɵ�
������������ȡ�����ɷ��������ø�������������ǻ�����������������ˮ��ɵ� ����Ӧ�ĺϳ�·��Ϊ
����Ӧ�ĺϳ�·��Ϊ ��
��
�ʴ�Ϊ�� ��
��
��5��I+F��J�Ļ�ѧ����ʽΪ ��
��
�ʴ�Ϊ�� ��
��
���� ���⿼���л���ĺϳɣ���ȷ�ϳ�·���и����ʵ��ƶ��ǽ����Ĺؼ���ע��̼���Ǽܡ������ŵı仯����Ӧ�����뷴Ӧ���͵Ĺ�ϵ���ɽ����Ŀ�Ѷ��еȣ�

�٦�=��17V+22400��/��22.4+22.4V�� �ڦ�=17c/��1000�ѣ�
��C=1000�Ѧ�/35 �ܦ�=17V/��17V+22400���ݣ�c=1000V��/��17V+22400��
| A�� | �٢ڢܢ� | B�� | �ڢܢ� | C�� | �ۢܢ� | D�� | �٢ڢ� |
| A�� | ��������ֱ�ӷ�Ӧ���ܵõ�Na2O2 | |
| B�� | Na2O2��ˮ���ҷ�Ӧ�ų�O2����ÿ����1 mol O2ת��2 mol���� | |
| C�� | Na2O2��CO2���Է�Ӧ�ų�O2����˿�������DZˮͧ�Ĺ����� | |
| D�� | Na2O2�ڿ����г�ʱ�䱩¶��������Na2CO3 |
| A�� | C3H8��C5H12һ����Ϊͬϵ�� | |
| B�� | 1mol�������������Ľ����Ʒ�Ӧ����������2.5molH | |
| C�� | ���ȼ���ֻ��һ�ֽṹ֤���������������幹�� | |
| D�� | ��֬��ʳ���в���������ߵ�Ӫ������ |
| A�� | NH4HCO3��Һ�����KOHŨ��Һ��NH4++OH-�TNH3��+H2O | |
| B�� | ��Ba��OH��2��Һ�еμ�NaHSO4��Һ��ǡ��Ϊ���ԣ�Ba2++OH-+H++SO42-�TBaSO4��+H2O | |
| C�� | ϡ�����������м��Ӧ��3Fe+8H++2NO3-�T3Fe2++2NO��+4H2O | |
| D�� | Fe��OH��3�����������Fe��OH��3+3H+�TFe3++3H2O |
| A�� | H2O | B�� | ҺNH3 | C�� | Һ̬HF | D�� | ��H2SO4 |
| A�� | �����£�100 mL 1 mol•L-1 Na2CO3��Һ�к��е�������������0.1NA | |
| B�� | ���³�ѹ�£�28g14CO��N2�Ļ����������������Ϊ14NA | |
| C�� | ��NA��NH3��������1Lˮ�еõ�1 mol•L-1�İ�ˮ | |
| D�� | 1molCl2�ֱ�����������������������Һ��Ӧ��ת�Ƶ�������Ϊ2NA |

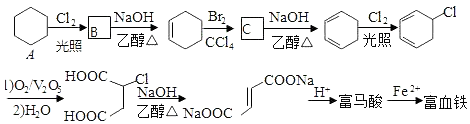
 ��
�� �����к��еĹ���������Ϊ���Ȼ���̼̼˫����
�����к��еĹ���������Ϊ���Ȼ���̼̼˫����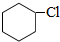 +NaOH$��_{��}^{�Ҵ�}$
+NaOH$��_{��}^{�Ҵ�}$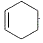 +NaCl+H2O��
+NaCl+H2O��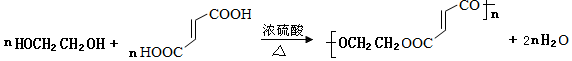 ��
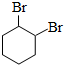
��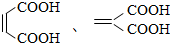 ��д ���ṹ��ʽ����
��д ���ṹ��ʽ����