��Ŀ����
��ѧ����������Ĺ�ϵԽ��Խ���У�
��1�����и���ʳ���е����ʺ�����ḻ���� ������ţ���ͬ����
��2������ȱ����Ԫ���������״���״�Ԫ�������ڱ��е���Ϣ��ͼ��ʾ��
������˵����ȷ���� ��
�ٵ�ʳ���еĵ���ָ�ⵥ��
�ڵ��ԭ��������53
�������߲ˡ��Ϲ��к��ḻ�ĵ�Ԫ��
�ܵ�Ԫ�ص����ԭ��������126.9��
��3�����������е���Ʒ�����л��ϳɲ����Ƴɵ��� ��
��4�����Ǵ����·�ͨ��������ά֯�ɵģ���������ά��������ë�����ڵȣ������շ��ɳ���������������ά���������պ�����ս���ë��ζ����ά�� ��
���� ����ë �۵���
��5����ѧ��Ԥ�⡰���ܡ�����21���������������Դ������ȼ�յĻ�ѧ����ʽΪ ��
��1�����и���ʳ���е����ʺ�����ḻ����

��2������ȱ����Ԫ���������״���״�Ԫ�������ڱ��е���Ϣ��ͼ��ʾ��

������˵����ȷ����
�ٵ�ʳ���еĵ���ָ�ⵥ��
�ڵ��ԭ��������53
�������߲ˡ��Ϲ��к��ḻ�ĵ�Ԫ��
�ܵ�Ԫ�ص����ԭ��������126.9��
��3�����������е���Ʒ�����л��ϳɲ����Ƴɵ���

��4�����Ǵ����·�ͨ��������ά֯�ɵģ���������ά��������ë�����ڵȣ������շ��ɳ���������������ά���������պ�����ս���ë��ζ����ά��
���� ����ë �۵���
��5����ѧ��Ԥ�⡰���ܡ�����21���������������Դ������ȼ�յĻ�ѧ����ʽΪ
���㣺�����ᡢ�����ʵĽṹ�������ص�,���ǡ���ѿ�Ǽ��,��Ԫ�ض����彡������Ҫ����
ר�⣺
��������1�����ݵ������ǻ�����������Ҫԭ�ϣ���ϳ�����ʳ��ɷַ������
��2������������Ԫ����ɻ�ԭ�ӹ��ɵģ�
�ڢܸ���ͼʾ�и������ִ����ĺ�����
���߲˺Ϲ�����Ҫ��ά���أ�
��3�������л��ϳɲ��ϵĶ�������жϣ�
��4������������ë�����ڵijɷּ����ʽ��з�����
��5�����ݻ�ѧ����ʽ����дҪ����У�
��2������������Ԫ����ɻ�ԭ�ӹ��ɵģ�
�ڢܸ���ͼʾ�и������ִ����ĺ�����
���߲˺Ϲ�����Ҫ��ά���أ�
��3�������л��ϳɲ��ϵĶ�������жϣ�
��4������������ë�����ڵijɷּ����ʽ��з�����
��5�����ݻ�ѧ����ʽ����дҪ����У�
���
�⣺��1���������ǹ���ϸ���Ļ������ʣ��ǻ�����������Ҫԭ�ϣ��٢ڸ���ά���أ�����Ҫ�ɷ������࣬�ܸ��������ʣ��ʴ�Ϊ���ܣ�
��2���ٴ���������Ԫ����ɵģ�����˵����ԭ�ӹ��ɵģ��ӵĵ��ǵ�����еĵ�Ԫ�أ�
��53�������ǵ��ԭ��������
���߲ˡ��Ϲ��и���ά���أ�������Ҫ���ں���Ʒ�У�
��126.9��ʾ���ǵ�����ԭ��������
��ѡ���ڢܣ�
��3�����л��߷��ӻ������ƳɵIJ��Ͼ����л��ϳɲ��ϣ������ϳ���ά���ϳ������ϣ�
�����ںϳ��������ɽ����Ƴɵģ��������������Ȼ�л����ϣ����������ϣ��ʴ�Ϊ���٢ܣ�
��4���������������ս���ëζ����ë��Ҫ�ɷ��ǵ����ʣ��ʴ�Ϊ���ڣ�
��5������ȼ������ˮ����д������ʽ���ʴ�Ϊ��2H2+O2
2H2O��
��2���ٴ���������Ԫ����ɵģ�����˵����ԭ�ӹ��ɵģ��ӵĵ��ǵ�����еĵ�Ԫ�أ�
��53�������ǵ��ԭ��������
���߲ˡ��Ϲ��и���ά���أ�������Ҫ���ں���Ʒ�У�
��126.9��ʾ���ǵ�����ԭ��������
��ѡ���ڢܣ�
��3�����л��߷��ӻ������ƳɵIJ��Ͼ����л��ϳɲ��ϣ������ϳ���ά���ϳ������ϣ�
�����ںϳ��������ɽ����Ƴɵģ��������������Ȼ�л����ϣ����������ϣ��ʴ�Ϊ���٢ܣ�
��4���������������ս���ëζ����ë��Ҫ�ɷ��ǵ����ʣ��ʴ�Ϊ���ڣ�
��5������ȼ������ˮ����д������ʽ���ʴ�Ϊ��2H2+O2
| ||
���������⿼���л�������ʣ��Ƚϻ��������ضԻ���֪ʶ�Ĺ��̣�ע��Ի���֪ʶ���������գ�

��ϰ��ϵ�д�
�����Ŀ
�û����������ȡH2SO4������H2SO4������ȡ���ʣ�NH4��2SO4�����պ�FeS280.2%�Ļ�����75.0t������������79.2t��NH4��2SO4����֪NH3��������Ϊ92.6%��H2SO4��������Ϊ89.8%�������������ȡH2SO4ʱ����ʧ��Ϊ��������
| A��23.8% |
| B��33.4% |
| C��35.6% |
| D��63.8% |
ʵ�����в�ͬ���Լ��в�ͬ�ı��淽�������б��淽���в���ȷ���� ��������
| A����������������Һ������������������� |
| B�������Ʊ�����װ��ú�͵IJ��������ƿ�� |
| C���廯�����屣������ɫ�������Ĺ��ƿ�� |
| D������Ũ�������ô���Ƥ���ӵ���ɫϸ��ƿ |
һ����Na2O2��һ������CO2��Ӧ��Ĺ�������18.4g��ǡ����400mL 1mol?L-1��ϡ������ȫ��Ӧ������˵���������ǣ�NA��ʾ����٤����������������
| A����Ӧ�е���ת����Ϊ0.2NA |
| B��һ������CO2Ϊ0.1NA |
| C��18.4 g��������Ϊ0.1mol Na2CO3��0.1molNa2O2�Ļ���� |
| D��һ������Na2O2Ϊ0.1mol |
�����ж���֬������Ӧ������ɵ������ǣ�������
| A����ӦҺʹ��ɫʯ����ֽ���� |
| B����Ӧ���ã���ӦҺ���ֲ� |
| C����Ӧ���ã���ӦҺ��Ϊ���� |
| D����ӦҺʹ��ɫʯ����ֽ��� |
0.1mol?L-1��ϡ������0.2mol?L-1��������Һ�������ȫ��Ӧ��÷�Ӧ�����Һ��c��CH3COO-����c��Cl-�����������ж��д�����ǣ�������
| A���¶�Ϊ298Kʱ��pH��7 |
| B��c��CH3COOH��+c��CH3COO-��=0.1 mol?L-1 |
| C��c��CH3COOH����c��CH3COO-�� |
| D��c��CH3COO-��+c��OH-��=c��Na+��+c��H+�� |
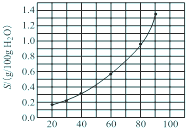 ��������AgBrO3�����ܽ�����¶ȱ仯������ͼ��ʾ������˵��������ǣ�������
��������AgBrO3�����ܽ�����¶ȱ仯������ͼ��ʾ������˵��������ǣ�������| A���¶�����ʱ�������ܽ��ٶȼӿ� |
| B�����������ܽ��Ƿ��ȹ��� |
| C��60��ʱ����������KspԼ����6��10-4 |
| D������������������ˮ |
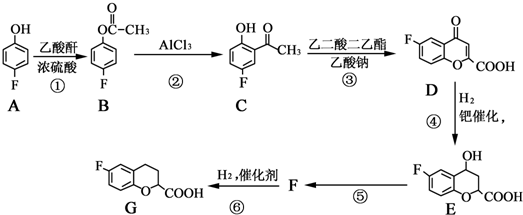
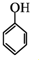 ��������Ϊԭ���Ʊ�
��������Ϊԭ���Ʊ�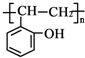 �ĺϳ�·
�ĺϳ�· �ͼ۵ij��������ա����������Ƕ�δ����Դ���輰�۸�����й�ע�����Ҵ�ʹ����Ѱ��ʯ�͵����Ʒ��
�ͼ۵ij��������ա����������Ƕ�δ����Դ���輰�۸�����й�ע�����Ҵ�ʹ����Ѱ��ʯ�͵����Ʒ��