��Ŀ����
11��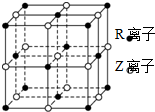 ԭ������С��36��X��Y��Z��W��R����Ԫ�أ�ԭ������������������Ԫ���У���RΪ����Ԫ�أ���ԭ������Ϊ27��X�۵����Ų�ʽΪnsnnpn��Ԫ��Z��̬ԭ��s����������p����������ȣ�W����������Ԫ���ܲ�����ͬ����δ�ɶԵ�����ĿΪ1����
ԭ������С��36��X��Y��Z��W��R����Ԫ�أ�ԭ������������������Ԫ���У���RΪ����Ԫ�أ���ԭ������Ϊ27��X�۵����Ų�ʽΪnsnnpn��Ԫ��Z��̬ԭ��s����������p����������ȣ�W����������Ԫ���ܲ�����ͬ����δ�ɶԵ�����ĿΪ1���������жϳ���Ԫ�ػش��������⣺
��1����̬Rԭ�Ӻ�������Ų�ʽΪ1s22s22p63s23p63d74s2��[Ar]3d74s2��X��Y��Z����ԭ�ӵ�һ�������ɴ�С��˳��ΪN��O��C���þ���Ԫ�ط��ű�ʾ����
��2��1mol�����[R��XY��6]4-�к��ЦҼ�����ĿΪ12mol��12��6.02��1023��
��3��XZW2��һ�ָ߶������ʣ��ṹ���ȩ���ƣ�Xԭ���ӻ���ʽΪsp2����е���ڼ�ȩ����Ҫԭ������ΪCOCl2����Է��������ȼ�ȩ�ʷ��»����ȼ�ȩ������е�ȼ�ȩ�ߣ�
��4��YW3Ϊ��ɫ����״�����д̼�����ζ��Һ�壬�÷��ӵ����幹��Ϊ�����Σ���������������
��5��ij����ľ����ṹ��ͼ��ʾ����û�����Ļ�ѧʽΪCoO��R��Z�þ���Ԫ�ط��ű�ʾ����
���� ԭ������С��36��X��Y��Z��W��R����Ԫ�أ�ԭ������������������Ԫ���У���RΪ����Ԫ�أ���ԭ������Ϊ27����RΪCoԪ�أ�X�۵����Ų�ʽΪnsnnpn��n=2����X�ĵ����Ų�ʽΪ2s22p2��XΪCԪ�أ�Ԫ��Z��̬ԭ��s����������p����������ȣ���Z�ĵ����Ų�ʽֻ��Ϊ1s22s22p2��ΪOԪ�أ����Y��ԭ��������֪YΪNԪ�أ�W����������Ԫ���ܲ�����ͬ����δ�ɶԵ�����ĿΪ1����Yֻ��λ�ڵ������ڣ�ΪClԪ�أ��ݴ˽��н��
��� �⣺ԭ������С��36��X��Y��Z��W��R����Ԫ�أ�ԭ������������������Ԫ���У���RΪ����Ԫ�أ���ԭ������Ϊ27����RΪCoԪ�أ�X�۵����Ų�ʽΪnsnnpn��n=2����X�ĵ����Ų�ʽΪ2s22p2��XΪCԪ�أ�Ԫ��Z��̬ԭ��s����������p����������ȣ���Z�ĵ����Ų�ʽֻ��Ϊ1s22s22p2��ΪOԪ�أ����Y��ԭ��������֪YΪNԪ�أ�W����������Ԫ���ܲ�����ͬ����δ�ɶԵ�����ĿΪ1����Yֻ��λ�ڵ������ڣ�ΪClԪ�أ�
��1��R��ԭ������Ϊ27�����̬ԭ�Ӻ����������Ϊ27�����������Ų�ʽΪ��1s22s22p63s23p63d74s2��[Ar]3d74s2��
ͬһ����Ԫ�أ�Ԫ�صĵ�һ����������ԭ�����������������IIA�塢��VA��Ԫ�ص�һ�����ܴ���������Ԫ�أ�C��N��OԪ�ش���ͬһ������ԭ������������N���ڵ�VA�壬���Ե�һ�����ܴ�СΪ��N��O��C��
�ʴ�Ϊ��1s22s22p63s23p63d74s2��[Ar]3d74s2��N��O��C��
��2��[R��XY��6]4-Ϊ[Co��CN��6]4-��[Co��CN��6]4-��CN-��Co֮����6����λ������ÿ��CN-�ڲ���һ�����ۼ�������1mol��������к��ЦҼ�����ĿΪ12NA����12��6.02��1023����
�ʴ�Ϊ��12mol��12��6.02��1023��
��3��XZW2ΪCOCl2�����ȩ�ṹ���ƣ���������к���1��̼̼˫����2��C-Cl������������ԭ��C����sp2�ӻ���
����COCl2����Է��������ȼ�ȩ���»����ȼ�ȩ��������е���ڼ�ȩ��
�ʴ�Ϊ��sp2����ΪCOCl2����Է��������ȼ�ȩ�ʷ��»����ȼ�ȩ������е�ȼ�ȩ�ߣ�
��4��NCl3�ķ�����Nԭ���γ�3��N-Cl��������1�Թµ��Ӷԣ��������幹��Ϊ�����Σ�
�ʴ�Ϊ�������Σ�
��5��RΪCo��ZΪO�����ݾ�̯����֪�������������������λ�����Ϻ����ģ���Ŀ=12��$\frac{1}{4}$+1=4��������λ�ڶ��㡢���ģ���Ŀ=8��$\frac{1}{8}$+6��$\frac{1}{2}$=4�����������ӡ������Ӹ�������1��1���仯ѧʽΪ��CoO��
�ʴ�Ϊ��CoO��
���� ���⿼����λ�á��ṹ�����ʹ�ϵ���ۺ�Ӧ�ã���Ŀ�Ѷ��еȣ��ƶ�Ԫ��Ϊ���ؼ���ע����������ԭ�ӽṹ��Ԫ�������ɡ�Ԫ�����ڱ��Ĺ�ϵ����5��Ϊ�״��㣬ע����ȷ��̯���ھ��������е�Ӧ�ã�

 ���Ž�������С״Ԫϵ�д�
���Ž�������С״Ԫϵ�д�| A�� | 3A+B=C | B�� | 2A+2B=3C | C�� | 4A+6B=9C | D�� | 9A+6B=4C |
| A�� | Ԫ��Hֻ��${\;}_1^1$H��${\;}_1^2$H���ֺ��� | |
| B�� | Cl2��NH4Cl����Ԫ�����İ뾶��r��Cl����r��Cl-�� | |
| C�� | ��ҵ�ϳ��ø���ĸ�ƿ����Һ�ȣ�����Ϊ�����������κ������¶�����Ӧ | |
| D�� | �÷�Ӧ���������������������� |
| A�� | 1 mol FeBr2������������Ӧʱ��������ת�Ƶĵ�����ԼΪ3��6.02��1023 | |
| B�� | �����£�pH��Ϊ3��������Һ����HCl��Һ ��H2SO4��Һ��CH3COOH��Һ���������ʵ����ʵ���Ũ�ȴ�С˳��Ϊ��=�ڣ��� | |
| C�� | �������NaCl��AlCl3��ȡNa��Al | |
| D�� | һ�������·�ӦCr2O72-��aq��+H2O��l��?2CrO42-��aq��+2H+��aq���ﵽƽ��μ�����Ũ���ᣬ���´�ƽ��ǰ��2v����Cr2O72-����v����CrO42-�� |
| A�� | H2SO4���ᣬ��ΪH2SO4�к�����Ԫ�� | |
| B�� | �����ǽ��壬��Ϊ�����еķ�ɢ������ֱ����1��100 nm֮�� | |
| C�� | Na��Cl2��ȼ������������ԭ��Ӧ����Ϊ��Ӧ���е��ӵ�ʧ | |
| D�� | Na��H2O�ķ�Ӧ�������ӷ�Ӧ����Ϊ��Ӧ��û�����Ӳμ� |
| A�� | ��Ӧ�١��ھ�Ϊ������ԭ��Ӧ | |
| B�� | ��Ӧ�������������뻹ԭ��������ʵ���֮��Ϊ1��1 | |
| C�� | ����β��N0x�����չ����У���ת�Ƶ��ӵ����ʵ���Ϊ1mol | |
| D�� | ����β����NO2��N0�������Ϊ6��1�� |

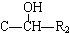 ��R������������ԭ�ӣ���ͬ����
��R������������ԭ�ӣ���ͬ����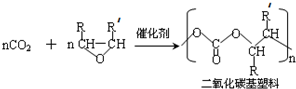
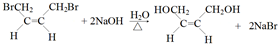 ����Ӧ�ܵĻ�ѧ��Ӧ����ʽ
����Ӧ�ܵĻ�ѧ��Ӧ����ʽ ����Ӧ�ݵĻ�ѧ��Ӧ����ʽ
����Ӧ�ݵĻ�ѧ��Ӧ����ʽ