��Ŀ����
N2O����ҽѧ�ϵ�һ��������������һ�ֳ������������塣��ȡN2O�ķ����кܶ࣬����
��NH4NO3===N2O����2H2O
��K2SO3��2NO===K2SO4��N2O��
��2NH3��2O2===N2O����3H2O
��2NaNO3����NH4��2SO4===2N2O����Na2SO4��4H2O�ȡ�����˵����ȷ����
A����Ӧ��������識������������ǻ�ԭ����H2O����������
B����Ӧ����K2SO3�ǻ�ԭ����N2O����������
C����Ӧ����ÿ����1 mol N2O��ת��8 mol����
D����Ӧ����NaNO3�ǻ�ԭ����N2O���������������ǻ�ԭ����
��ϰ��ϵ�д�
�����Ŀ
18�������й����ӵļ��飬��ȷ���ǣ�������
| A�� | ij��������ɫ��Ӧ�ʻ�ɫ��˵����������һ������Na+ | |
| B�� | ��ij��Һ�м������������ݲ�����˵��һ����CO32- | |
| C�� | ��ij��Һ�м����Ȼ�����Һ���а�ɫ����������˵��һ����SO42- | |
| D�� | ��ij��Һ�м�����������Һ���а�ɫ����������˵��һ����Cl- |


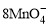 ��5Cu2S��44H��= 10Cu2����5SO2����8Mn2����22H2O
��5Cu2S��44H��= 10Cu2����5SO2����8Mn2����22H2O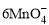 ��5CuS��28H�� = 5Cu2����5SO2����6Mn2����14H2O
��5CuS��28H�� = 5Cu2����5SO2����6Mn2����14H2O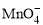 ��______Fe2����______H�� =______Mn2����______Fe3����___
��______Fe2����______H�� =______Mn2����______Fe3����___ ___H2O
___H2O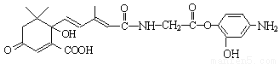 ��NaOH��Һ��Ӧ���������5molNaOH
��NaOH��Һ��Ӧ���������5molNaOH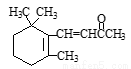 ����1molHBr�����ӳɷ�Ӧ�ɵõ�3�ֲ�ͬ����
����1molHBr�����ӳɷ�Ӧ�ɵõ�3�ֲ�ͬ���� ===Cu2����NO2����H2O
===Cu2����NO2����H2O 2 OH����H2����Cl2��
2 OH����H2����Cl2��