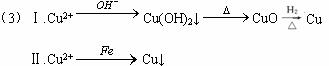��Ŀ����
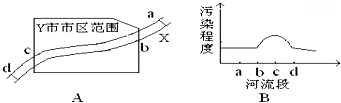
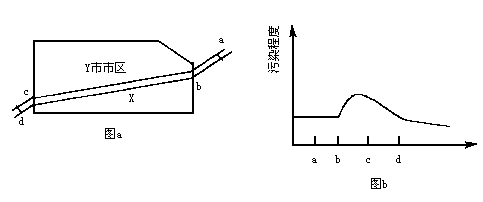
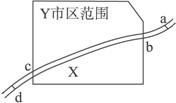
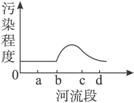
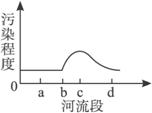
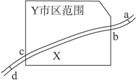
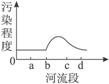
��ͼ1��ʾ��X������ij��ҵ����Y�У�����a�������Σ�d�������Ρ�����2006��10���л����ּ��վ��X��ˮ�ʼ���������Ƴɼ�ͼ��ͼ2��ʾ�������ͼ����������⡣
��1�����X����Ⱦ����Ҫ��ȾԴ����ֲܷ���___________________��
A.ab�� B.bc�� C.cd�� D.bd��
��2���о����X����Ⱦ�����ֿ��ܵ���ȾԴ��__________��__________��__________��
ͼ1 ͼ2
��3����c��ˮ�����м�⣬�������к���Cu2+���ؽ������ӡ���Ҫ��ͭ���ӱ�Ϊ����ͭ�����о����ֻ��յķ�����
������:__________________________________________;
������________________________________________��
��4��d������Ⱦ�̶����Խ��ͣ��������ѧ����ѧ������ѧ�����������ԭ��_________________________________________________��
(1)B
(2)��ҵ��ˮ��������������ˮ����������(�������ּ���)
��3����.Cu2+![]() OH-Cu(OH)2��
OH-Cu(OH)2��![]() CuO
CuO![]() Cu
Cu
��.Cu2+![]() Cu��
Cu��
(4)������������ѧת��������ֽ��
����:
��1����ͼ2��������Ⱦ�̶ȴ�b�㿪ʼ���������X����Ⱦ����Ҫ��ȾԴ�ֲ���bc�Ρ�
��2��bc�κӰ�����Y������Χ�����X����Ⱦ�Ŀ�����ȾԴ�ǹ�ҵ��ˮ��������ˮ��������������ҵ����������ȡ�
��3����Ҫ����ˮ��Cu2+��Ϊ����Cu�����Լ�NaOH��Һ�����ˣ��õ�Cu(OH)2���������������ȷֽ��CuO������H2��ԭ�õ�����Cu����������۽����û����õ�����Cu��
��4��d����Ⱦ�̶����Խ��ͣ������Dz�����������������ѧת��������ֽ�ȷ�����������

 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д�