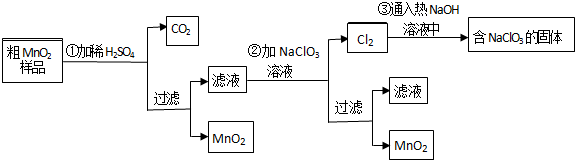
��Ŀ����
6��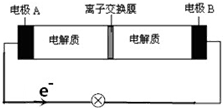 ��ͼװ����һ�ֿɳ����ʾ��ͼ��װ�õ����ӽ���Ĥֻ����Na+ͨ������֪�䡢�ŵ�Ļ�ѧ����ʽΪ2Na2S2+NaBr3$?_{���}^{�ŵ�}$Na2S4+3NaBr������˵����ȷ���ǣ�������
��ͼװ����һ�ֿɳ����ʾ��ͼ��װ�õ����ӽ���Ĥֻ����Na+ͨ������֪�䡢�ŵ�Ļ�ѧ����ʽΪ2Na2S2+NaBr3$?_{���}^{�ŵ�}$Na2S4+3NaBr������˵����ȷ���ǣ�������| A�� | ���ʱ��A��Ӧ��ֱ����Դ���������� | |
| B�� | �ŵ�ʱ������0.1molNa+ͨ�����ӽ���Ĥʱ��B������0.3molNaBr���� | |
| C�� | �ŵ�ʱ�������Ӵ�����ͨ�����ӽ���Ĥ | |
| D�� | �ŵ�ʱ��������ӦʽΪ3NaBr-2e-�TNaBr3+2Na+ |
���� A�����ʱ��A�������������������Դ����������
B���ŵ�ʱ������0.1molNa+ͨ�����ӽ���Ĥʱ��B�缫�ϵõ�0.1mol���ӣ�
C���ŵ�ʱ����������������������ƶ���
D���ŵ�ʱ��������ӦʽΪ2S22--2e-�TS42-��������ӦʽΪBr3-+2e-�T3Br-��
��� �⣺A�����ʱ��A�������������������Դ��������������A���Դ������������A����
B���ŵ�ʱ������0.1molNa+ͨ�����ӽ���Ĥʱ��B�缫�ϵõ�0.1mol���ӣ�����Br3-+2e-�T3Br-֪����0.15molNaBr���ɣ���B����
C���ŵ�ʱ����������������������ƶ�����Na+������ͨ�����ӽ���Ĥ����C��ȷ��
D���ŵ�ʱ��������ӦʽΪ2S22--2e-�TS42-��������ӦʽΪBr3-+2e-�T3Br-����D����
��ѡC��
���� ���⿼�黯ѧ��Դ���͵�أ�Ϊ��Ƶ���㣬����ԭ��غ͵���ԭ���������ƶ������ǽⱾ��ؼ����ѵ��ǵ缫��Ӧʽ����д��Bѡ����㣬��Ŀ�ѶȲ���

��ϰ��ϵ�д�
 �ƸԹھ��ο���ϵ�д�
�ƸԹھ��ο���ϵ�д�
�����Ŀ
2�������Ϊ2L���ܱ������г���2mol SO2��һ������O2��������Ӧ2SO2+O2?2SO3������Ӧ���е�4minʱ����ô�ʱSO2Ϊ0.4mol����ô��Ӧ���е�2minʱ���ܱ�������SO2���ʵ����ǣ�������
| A�� | ��1.2 mol | B�� | 1.2 mol | C�� | ����1.6 mol | D�� | 1.6 mol |
1�� ij������ȤС�����ⶨijNaOH��Һ��Ũ�ȣ�������������£�
ij������ȤС�����ⶨijNaOH��Һ��Ũ�ȣ�������������£�
�ٽ���ʽ�ζ���������ˮϴ�����ô�����Һ��ϴ����ע�������Һ�����ڵζ��ܵļ��첿�ֳ�����Һ����ʹҺ�洦�ڡ�0���̶����µ�λ�ã����¶���������ƿ������ˮϴ�����ô�����Һ��ϴ��ƿ2��3�Σ��Ӽ�ʽ�ζ����з�
��25.00mL������Һ����ƿ�У�
�ڽ���ʽ�ζ���������ˮϴ��������������ע��0.1000mol/L�����ᣬ���ڵζ��ܵļ��첿�ֳ�����Һ����ʹҺ�洦�ڡ�0���̶����µ�λ�ã����¶�����
������ƿ�е����̪��ָʾ�������еζ����ζ���ָʾ���պñ�ɫ���Ҳ������ϱ�ɫΪֹ�����������������ΪV1mL��
���ظ����Ϲ��̣����ڵζ�����������ƿ����5mL������ˮ�����������������ΪV2mL���Իش��������⣺
��1����ƿ�е���Һ�Ӻ�ɫ��Ϊ�� ɫʱ��ֹͣ�ζ���
��2���ζ�ʱ�ߵα�ҡ����ƿ���۾�Ӧ�۲�B
A���ζ�����Һ��ı仯 ��B����ƿ����Һ��ɫ�ı仯
��3����С���ڲ�����еĴ����Ǵ�����Һ��ϴ��ƿ�ɴ���ɵIJⶨ���ƫ�ߣ�ƫ�ߡ�ƫ�ͻ���Ӱ�죩
��4�������ȱ�ٵIJ����Ǿ���ʽ�ζ���������ˮϴ����Ӧ�ñ�Һ��ϴ
��5����ͼ����ij�εζ�ʱ�ĵζ����е�Һ�棬�����Ϊ22.60mL
��6��
���������ռ���Һ��Ũ��0.0800mol/L��
 ij������ȤС�����ⶨijNaOH��Һ��Ũ�ȣ�������������£�
ij������ȤС�����ⶨijNaOH��Һ��Ũ�ȣ�������������£��ٽ���ʽ�ζ���������ˮϴ�����ô�����Һ��ϴ����ע�������Һ�����ڵζ��ܵļ��첿�ֳ�����Һ����ʹҺ�洦�ڡ�0���̶����µ�λ�ã����¶���������ƿ������ˮϴ�����ô�����Һ��ϴ��ƿ2��3�Σ��Ӽ�ʽ�ζ����з�
��25.00mL������Һ����ƿ�У�
�ڽ���ʽ�ζ���������ˮϴ��������������ע��0.1000mol/L�����ᣬ���ڵζ��ܵļ��첿�ֳ�����Һ����ʹҺ�洦�ڡ�0���̶����µ�λ�ã����¶�����
������ƿ�е����̪��ָʾ�������еζ����ζ���ָʾ���պñ�ɫ���Ҳ������ϱ�ɫΪֹ�����������������ΪV1mL��
���ظ����Ϲ��̣����ڵζ�����������ƿ����5mL������ˮ�����������������ΪV2mL���Իش��������⣺
��1����ƿ�е���Һ�Ӻ�ɫ��Ϊ�� ɫʱ��ֹͣ�ζ���
��2���ζ�ʱ�ߵα�ҡ����ƿ���۾�Ӧ�۲�B
A���ζ�����Һ��ı仯 ��B����ƿ����Һ��ɫ�ı仯
��3����С���ڲ�����еĴ����Ǵ�����Һ��ϴ��ƿ�ɴ���ɵIJⶨ���ƫ�ߣ�ƫ�ߡ�ƫ�ͻ���Ӱ�죩
��4�������ȱ�ٵIJ����Ǿ���ʽ�ζ���������ˮϴ����Ӧ�ñ�Һ��ϴ
��5����ͼ����ij�εζ�ʱ�ĵζ����е�Һ�棬�����Ϊ22.60mL
��6��
| �ζ����� | ����Һ�����mL�� | �����������mL�� | |
| �ζ�ǰ������mL�� | �ζ��������mL�� | ||
| ��һ�� | 25.00 | 0.50 | 20.40 |
| �ڶ��� | 25.00 | 4.00 | 24.10 |
11�����������غ�˫�������أ��ṹ��ͼ��������˵���У�������ǣ�������
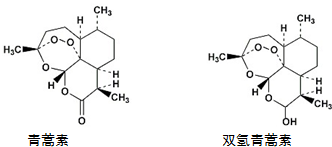

| A�� | �������ܷ�����ԭ��Ӧ | |
| B�� | ˫�������ط���������Ӧ��������Ӧ | |
| C�� | �����ط����к��й��������������Ѽ� | |
| D�� | ˫�������ط�������2����Ԫ����2����Ԫ�� |
15����NAΪ�����ӵ�������6ֵ������˵����ȷ���ǣ�������
| A�� | ���³�ѹ�£�16 g O2��32g O3�Ļ�����У�����Oԭ����ĿΪ3NA | |
| B�� | ��״���£�1 mol Na2O��1 mol Na2O2�Ļ�����У�������������Ϊ7NA | |
| C�� | 1 mol NaBH4�����Ӽ�����ĿΪ2NA | |
| D�� | ��K35ClO3+6H37Cl�TKCl+3Cl2��+3H2O�У�������71 g Cl2��ת�Ƶĵ�����ĿΪ$\frac{5}{3}$NA |
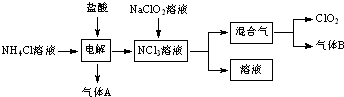
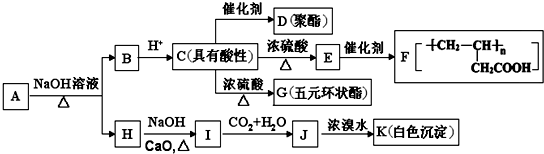
 +NaOH$��_{��}^{CaO}$R-H+Na2CO3
+NaOH$��_{��}^{CaO}$R-H+Na2CO3 ��K�Ľṹ��ʽΪ
��K�Ľṹ��ʽΪ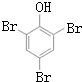 ��
��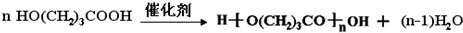 ��
��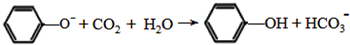 ��
�� ��д�ṹ��ʽ����
��д�ṹ��ʽ����