��Ŀ����
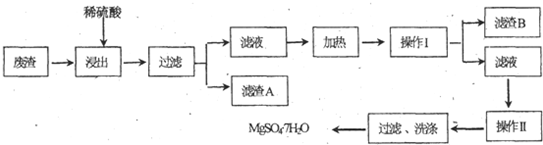
MgS04?7H20��������ֽ����֯���մɡ����Ṥҵ��Ҳ����ҽ��������к�Σ�ij��������Ҫ�ɷ���MgC03�������CaC03��Si02�����ʣ��Ӵ˷����л���MgS04?7H20�Ĺ����£�
��֪CaS04��MgS04���ܽ�ȣ�S�����¶ȣ�T���仯������ͼ��ʾ���Իش��������⣺

��1���������������У�Ϊ���þ�Ľ����ʣ��ɲ�ȡ�Ĵ�ʩ�� ��Ҫ��д������һ������
��2������B����Ҫ�ɷֵĻ�ѧʽ�� ��
��3��������������� ��������������� ��
��4��������Ͳ�������Ҫ�õ����������������в���������Ҫ������ ���������в���������Ҫ������ ��
��5��������ΪMg2+��ˮ�⣬����������ٹ��ˡ�ϴ�ӵò���MgS04?7H20���壬����Ϊ����˵���Ƿ���ȷ ����ǡ�����ԭ���� ��

��֪CaS04��MgS04���ܽ�ȣ�S�����¶ȣ�T���仯������ͼ��ʾ���Իش��������⣺

��1���������������У�Ϊ���þ�Ľ����ʣ��ɲ�ȡ�Ĵ�ʩ��
��2������B����Ҫ�ɷֵĻ�ѧʽ��
��3���������������
��4��������Ͳ�������Ҫ�õ����������������в���������Ҫ������
��5��������ΪMg2+��ˮ�⣬����������ٹ��ˡ�ϴ�ӵò���MgS04?7H20���壬����Ϊ����˵���Ƿ���ȷ
���㣺�Ʊ�ʵ�鷽�������,þ��������Ҫ������
ר�⣺������Ҫ�Ľ������仯����
��������1����ϡ�����ڷ����У�MgCO3��CaCO3�ܽ⣬�����ܽ��-�¶�ͼ��֪��MgSO4���¶����߶��ܽ������
��2���ɹ������̿�֪����������������ˣ���Һ�к���CaSO4��MgSO4�������ܽ��-�¶�ͼ��֪��CaSO4���¶����߶��ܽ���½���MgSO4���¶����߶��ܽ��������Һ��������CaSO4��
��3���ɹ������̿�֪�����������ڼ��ȵ�����½���������Һ���룬������������Һ���������壮
��4���������ǹ��ˣ�������Ϊ�������ã��������ǽᾧ��������Ϊ�������ã�
��5��MgS04ˮ������������þ�����ᣬ�����Ƿǻӷ����ᣮ
��2���ɹ������̿�֪����������������ˣ���Һ�к���CaSO4��MgSO4�������ܽ��-�¶�ͼ��֪��CaSO4���¶����߶��ܽ���½���MgSO4���¶����߶��ܽ��������Һ��������CaSO4��
��3���ɹ������̿�֪�����������ڼ��ȵ�����½���������Һ���룬������������Һ���������壮
��4���������ǹ��ˣ�������Ϊ�������ã��������ǽᾧ��������Ϊ�������ã�
��5��MgS04ˮ������������þ�����ᣬ�����Ƿǻӷ����ᣮ
���
�⣺��1����ϡ�����ڷ����У�MgCO3��CaCO3�ܽ⣬�����ܽ��-�¶�ͼ��֪��MgSO4���¶����߶��ܽ�����������������У�Ϊ���þ�Ľ����ʣ��ɲ�ȡ�Ĵ�ʩ�ʵ���߷�Ӧ�¶ȣ�ͬʱ���Ͻ��裬���ӽ���ʱ�䣮
�ʴ�Ϊ���ʵ���߷�Ӧ�¶ȣ�ͬʱ���Ͻ��裬���ӽ���ʱ�䣮
��2���ɹ������̿�֪����������������ˣ���Һ�к���CaSO4��MgSO4�������ܽ��-�¶�ͼ��֪��CaSO4���¶����߶��ܽ���½���MgSO4���¶����߶��ܽ��������Һ��������CaSO4���ʲ������ڼ��ȵ�����¹��ˣ�����B��CaSO4��
�ʴ�Ϊ��CaSO4��
��3��CaSO4���¶����߶��ܽ���½������ȵ�����£����Խ���CaSO4�ܽ�ȣ��������ڼ��ȵ�����½���������Һ���룬Ӧ�dz��ȹ��ˣ�������������Һ���������壬ӦΪ����Ũ������ȴ�ᾧ��
�ʴ�Ϊ�����ȹ��ˣ�����Ũ������ȴ�ᾧ��
��4���������ǹ��ˣ�������Ϊ�������ã��������ǽᾧ��������Ϊ�������ã�
�ʴ�Ϊ�����������裮
��5��MgS04ˮ������������þ�����ᣬ�������ѻӷ����ᣬ������ɵ�������������þ����������þ�����������ˡ�ϴ�ӿ��Եõ�MgS04?7H20���壮
�ʴ�Ϊ�����������ѻӷ����ᣮ
�ʴ�Ϊ���ʵ���߷�Ӧ�¶ȣ�ͬʱ���Ͻ��裬���ӽ���ʱ�䣮
��2���ɹ������̿�֪����������������ˣ���Һ�к���CaSO4��MgSO4�������ܽ��-�¶�ͼ��֪��CaSO4���¶����߶��ܽ���½���MgSO4���¶����߶��ܽ��������Һ��������CaSO4���ʲ������ڼ��ȵ�����¹��ˣ�����B��CaSO4��
�ʴ�Ϊ��CaSO4��
��3��CaSO4���¶����߶��ܽ���½������ȵ�����£����Խ���CaSO4�ܽ�ȣ��������ڼ��ȵ�����½���������Һ���룬Ӧ�dz��ȹ��ˣ�������������Һ���������壬ӦΪ����Ũ������ȴ�ᾧ��
�ʴ�Ϊ�����ȹ��ˣ�����Ũ������ȴ�ᾧ��
��4���������ǹ��ˣ�������Ϊ�������ã��������ǽᾧ��������Ϊ�������ã�
�ʴ�Ϊ�����������裮
��5��MgS04ˮ������������þ�����ᣬ�������ѻӷ����ᣬ������ɵ�������������þ����������þ�����������ˡ�ϴ�ӿ��Եõ�MgS04?7H20���壮
�ʴ�Ϊ�����������ѻӷ����ᣮ
���������⿼��ѧ���Ķ���Ŀ��ȡ��Ϣ�������Թ������̵����⡢�����ƶϡ����ʷ����ᴿ���������Ƶȣ��Ѷ��еȣ���Ҫѧ���߱���ʵ�Ļ���֪ʶ���ۺ�����֪ʶ����Ϣ���н�������������

��ϰ��ϵ�д�
 һ����������ϵ�д�
һ����������ϵ�д�
�����Ŀ
��6.5gп����������ϡ�����У���п��Ӧ��ȫ����H2������ڱ�״����Ϊ��������
| A��1.12L |
| B��2.24L |
| C��11.2L |
| D��22.4L |
��NAΪ�����ӵ��������Ƶ�Ħ������ΪM g/mol���Ƶ��ܶ�Ϊ��g/cm3��������˵���У���ȷ���ǣ�������
��1g Na����ԭ����Ϊ��NA�� ��1cm3Na����ԭ����Ϊ��NA/M�� ��1����ԭ�ӵ�����ΪM/NAg
��1����ԭ��ռ�����ΪM/��NAcm3 ��1g��������O2��ȼ��ʧȥ�ĵ�����Ϊ2NA/M����
��1g Na����ԭ����Ϊ��NA�� ��1cm3Na����ԭ����Ϊ��NA/M�� ��1����ԭ�ӵ�����ΪM/NAg
��1����ԭ��ռ�����ΪM/��NAcm3 ��1g��������O2��ȼ��ʧȥ�ĵ�����Ϊ2NA/M����
| A���٢ڢ� | B���ڢۢ� |
| C���٢ڢ� | D���ۢܢ� |
�����ࡱ��һ��˼�뷽�����ڻ�ѧ��չ��������Ҫ���ã����з�����������ǣ�������
| A��������ԭ��Ӧ�У�һ��Ԫ�ػ��ϼ�������һ������һ��Ԫ�ػ��ϼ��½� |
| B�����ݷ�ɢϵ�Ƿ���ж��������ɢϵ��Ϊ��Һ�������Һ |
| C������ˮ��Һ�ܷ罫�������Ϊ����ʺͷǵ���� |
| D������������һ���ǽ�����������������ﲻһ���Ǽ��������� |
�����£����и���������ָ����Һ���ܴ���������ǣ�������
| A��c��H+��=1.0��10-13mol/L��Һ�У�C6H5O-��K+��SO42-��Br-- |
| B��������ˮ�У�Cl-��NO3-��Na+��SO32- |
| C��Na2S��Һ�У�SO42-��K+��Cl-��Cu2+ |
| D��pH=12����Һ�У�NO3-��I-��Na+��Al3+ |


 ��1�������£���20.0g 14%��NaCl��Һ��30.0g 24%��NaCl��Һ��ϣ��õ��ܶ�Ϊ1.17g/cm3�Ļ����Һ���û����Һ��NaCl�����ʵ���Ũ��Ϊ
��1�������£���20.0g 14%��NaCl��Һ��30.0g 24%��NaCl��Һ��ϣ��õ��ܶ�Ϊ1.17g/cm3�Ļ����Һ���û����Һ��NaCl�����ʵ���Ũ��Ϊ