��Ŀ����
�ȣ�Sr��������������Ԫ�أ��䵥�ʺͻ�����Ļ�ѧ������ơ��������ƣ�ʵ�����ú�̼���ȵķ�������SrCO3 38.40%��SrO12.62%��CaCO3 38.27%��BaCO3 2.54%���������������������8.17%���Ʊ������ȴ�Ʒ�IJ���ʵ��������£�
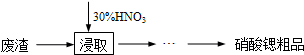
��1������Ũ�������������Ϊ65%���ܶ�Ϊ1.4g/cm3��Ҫ����30%ϡ����500mL������Ҫ���ĵ������� �������ƹ����в�ʹ����ƽ�������Ҫ����������� ������Ҫʹ�õ������� ��
��֪�����ε��ܽ�ȣ�g/100gˮ�������ʾ��
��2���ɽ�ȡ��õ��Ļ�����Ʊ������ȴ�Ʒ��ʵ�鲽������Ϊ�����ˡ� �� ��ϴ�ӣ����
��3����֪��������������л��ܼ�A�У�ʽ����Sr��NO3��2-212��Ba��NO3��2-261��Ca��NO3��2-164
�Ƶõ������ȴ�Ʒ�к�����Ca��NO3��2��Ba��NO3��2�����ʣ��ⶨ�����ȴ��ȵ�ʵ�����£�ȡ5.39g��������Ʒ�������������л��ܼ�A�������ˡ�ϴ�ӡ������ʣ�����5.26g�����˹������250mL����Һ��ȡ��25.00mL������pHΪ7������ָʾ������Ũ��Ϊ0.107mol/L��̼������Һ�ζ����յ㣬����̼������Һ22.98mL���ζ����̵ķ�Ӧ��Sr2++CO32-��SrCO3����Ba2++CO32-��BaCO3����
�ٵζ�ѡ�õ�ָʾ��Ϊ ���ζ��յ�۲쵽������Ϊ ��
�ڸ������ȴ�Ʒ�У������ȵ���������Ϊ ��С���������λ�������ζ�ǰ��Ʒ��Ca��NO3��2û�г��������ⶨ�������ȴ��Ƚ��� ���ƫ�ߡ�����ƫ�͡����䡱����
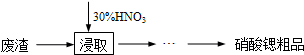
��1������Ũ�������������Ϊ65%���ܶ�Ϊ1.4g/cm3��Ҫ����30%ϡ����500mL������Ҫ���ĵ�������
��֪�����ε��ܽ�ȣ�g/100gˮ�������ʾ��
| �¶�/������ | 0 | 20 | 30 | 45 | 60 | 80 | 100 |
| Sr��NO3��2 | 28.2 | 40.7 | 47 | 47.2 | 48.3 | 49.2 | 50.7 |
| Ca��NO3��2?4H2O | 102 | 129 | 152 | 230 | 300 | 358 | 408 |
��3����֪��������������л��ܼ�A�У�ʽ����Sr��NO3��2-212��Ba��NO3��2-261��Ca��NO3��2-164
�Ƶõ������ȴ�Ʒ�к�����Ca��NO3��2��Ba��NO3��2�����ʣ��ⶨ�����ȴ��ȵ�ʵ�����£�ȡ5.39g��������Ʒ�������������л��ܼ�A�������ˡ�ϴ�ӡ������ʣ�����5.26g�����˹������250mL����Һ��ȡ��25.00mL������pHΪ7������ָʾ������Ũ��Ϊ0.107mol/L��̼������Һ�ζ����յ㣬����̼������Һ22.98mL���ζ����̵ķ�Ӧ��Sr2++CO32-��SrCO3����Ba2++CO32-��BaCO3����
�ٵζ�ѡ�õ�ָʾ��Ϊ
�ڸ������ȴ�Ʒ�У������ȵ���������Ϊ
���㣺���ʷ�����ᴿ�ķ����ͻ��������ۺ�Ӧ��,�Ʊ�ʵ�鷽�������
ר�⣺ʵ�������
��������̼���ȵķ�������SrCO3 38.40%��SrO12.62%��CaCO3 38.27%��BaCO3 2.54%���������������������8.17%����ϡ���ᷴӦ�����˺���Һ��Ӧ�ú���Sr��NO3��2��Ca��NO3��2��Ba��NO3��2���ɱ��е����ݿ��Կ�����Sr��NO3��2���ܽ�������¶ȵ����߱仯��������Ƶ��ܽ�����¶ȱ仯�ϴ���˿�ͨ�������ᾧ�����ȹ��ˣ�ϴ�ӣ�����������ȴֲ�Ʒ��
��1��Ũ�������������Ϊ65%���ܶ�Ϊ1.4g/cm3��Ҫ����30%ϡ����500mL��v��Ũ���ᣩ���ѣ�Ũ���ᣩ��Ũ�������������=m��ϡ���ᣩ��ϡ���������������m��ϡ���ᣩ=v��ϡ���ᣩ���ѣ�ϡ���ᣩ���ʻ���Ҫ����ϡ������ܶȣ������ƹ����в�ʹ����ƽ�������Ҫ���������Ϊv��Ũ���ᣩ��v��ϡ���ᣩ����ʵ����ȡ��Ӧ�ǣ�Ũ���������ˮ�����������ʹ�õ���������Ͳ���ձ�����������
��2���ɱ��е����ݿ��Կ�����Sr��NO3��2���ܽ�������¶ȵ����߱仯��������Ƶ��ܽ�����¶ȱ仯�ϴ���˿�ͨ���ɽ�ȡ��õ��Ļ�����Ʊ������ȴ�Ʒ��ʵ�鲽������Ϊ�������ᾧ�����ȹ��ˣ�ϴ�ӣ�����õ���
��3���ٷ����ζ����̿�֪����Ʒ��Һ��ɫ������̼���Ƴ�����ȫ���Ե����̪��Һָʾ�յ㣬�������һ����Һ�ʺ�ɫ��������ڲ���ɫ�������ζ�ǰ��Ʒ��Ca��NO3��2û�г����������ı���Һ̼���ƣ����ݵζ���������c������Һ��=
�����ı�Һ�࣬���ⶨ�������ȴ��Ȼ�ƫ�ߣ�
����֪��������������л��ܼ�A�У�ȡ5.39g��������Ʒ�������������л��ܼ�A�������ˡ�ϴ�ӡ������ʣ�����5.26g��ʣ�����Ϊ���ᱵ�������ȣ��������Ⱥ����ᱵ�����ʵ���Ϊx��y������������Ϊ5.26g������212x+261y=5.26�٣���̼�����Ӧ��ֻ�б����Ӻ������ӣ�Sr2++CO32-��SrCO3����Ba2++CO32-��BaCO3��������Ũ��Ϊ0.107mol/L��̼������Һ22.98mL�����ʵ���Ϊ0.00246mol������x+y=0.0246�ڣ��ɢ٢�ʽ��m�������ȣ�=212x=5.02g�������ȵ���������=
��100%=0.93�����ζ�ǰ��Ʒ��Ca��NO3��2û�г������ζ���Һʱ����̼������Һ�࣬�������������ʵ���ƫ�����ⶨ�������ȴ��Ƚ���ƫ�ߣ�
��1��Ũ�������������Ϊ65%���ܶ�Ϊ1.4g/cm3��Ҫ����30%ϡ����500mL��v��Ũ���ᣩ���ѣ�Ũ���ᣩ��Ũ�������������=m��ϡ���ᣩ��ϡ���������������m��ϡ���ᣩ=v��ϡ���ᣩ���ѣ�ϡ���ᣩ���ʻ���Ҫ����ϡ������ܶȣ������ƹ����в�ʹ����ƽ�������Ҫ���������Ϊv��Ũ���ᣩ��v��ϡ���ᣩ����ʵ����ȡ��Ӧ�ǣ�Ũ���������ˮ�����������ʹ�õ���������Ͳ���ձ�����������
��2���ɱ��е����ݿ��Կ�����Sr��NO3��2���ܽ�������¶ȵ����߱仯��������Ƶ��ܽ�����¶ȱ仯�ϴ���˿�ͨ���ɽ�ȡ��õ��Ļ�����Ʊ������ȴ�Ʒ��ʵ�鲽������Ϊ�������ᾧ�����ȹ��ˣ�ϴ�ӣ�����õ���
��3���ٷ����ζ����̿�֪����Ʒ��Һ��ɫ������̼���Ƴ�����ȫ���Ե����̪��Һָʾ�յ㣬�������һ����Һ�ʺ�ɫ��������ڲ���ɫ�������ζ�ǰ��Ʒ��Ca��NO3��2û�г����������ı���Һ̼���ƣ����ݵζ���������c������Һ��=
| c(��Һ)v(��Һ) |
| v(����Һ) |
����֪��������������л��ܼ�A�У�ȡ5.39g��������Ʒ�������������л��ܼ�A�������ˡ�ϴ�ӡ������ʣ�����5.26g��ʣ�����Ϊ���ᱵ�������ȣ��������Ⱥ����ᱵ�����ʵ���Ϊx��y������������Ϊ5.26g������212x+261y=5.26�٣���̼�����Ӧ��ֻ�б����Ӻ������ӣ�Sr2++CO32-��SrCO3����Ba2++CO32-��BaCO3��������Ũ��Ϊ0.107mol/L��̼������Һ22.98mL�����ʵ���Ϊ0.00246mol������x+y=0.0246�ڣ��ɢ٢�ʽ��m�������ȣ�=212x=5.02g�������ȵ���������=
| 5.02g |
| 5.39g |
���
�⣺��̼���ȵķ�������SrCO3 38.40%��SrO12.62%��CaCO3 38.27%��BaCO3 2.54%���������������������8.17%����ϡ���ᷴӦ�����˺���Һ��Ӧ�ú���Sr��NO3��2��Ca��NO3��2��Ba��NO3��2���ɱ��е����ݿ��Կ�����Sr��NO3��2���ܽ�������¶ȵ����߱仯��������Ƶ��ܽ�����¶ȱ仯�ϴ���˿�ͨ�������ᾧ�����ȹ��ˣ�ϴ�ӣ�����������ȴֲ�Ʒ��
��1��Ũ�������������Ϊ65%���ܶ�Ϊ1.4g/cm3��Ҫ����30%ϡ����500mL��v��Ũ���ᣩ���ѣ�Ũ���ᣩ��Ũ�������������=m��ϡ���ᣩ��ϡ���������������m��ϡ���ᣩ=v��ϡ���ᣩ���ѣ�ϡ���ᣩ���ʻ���Ҫ����ϡ������ܶȣ������ƹ����в�ʹ����ƽ�������Ҫ���������Ϊv��Ũ���ᣩ��v��ϡ���ᣩ����ʵ����ȡ��Ӧ�ǣ�Ũ��������ˮ�����������ʹ�õ���������Ͳ���ձ�����������
�ʴ�Ϊ��30%ϡ������ܶȣ�Ũ���������ˮ���������Ͳ���ձ�����������
��2���ɱ��е����ݿ��Կ�����Sr��NO3��2���ܽ�������¶ȵ����߱仯��������Ƶ��ܽ�����¶ȱ仯�ϴ���˿�ͨ���ɽ�ȡ��õ��Ļ�����Ʊ������ȴ�Ʒ��ʵ�鲽������Ϊ�������ᾧ�����ȹ��ˣ�ϴ�ӣ�����õ���
�ʴ�Ϊ�������ᾧ�����ȹ��ˣ�
��3���ٷ����ζ����̿�֪����Ʒ��Һ��ɫ������̼���Ƴ�����ȫ���Ե����̪��Һָʾ�յ㣬�������һ����Һ�ʺ�ɫ��������ڲ���ɫ�������ζ�ǰ��Ʒ��Ca��NO3��2û�г����������ı���Һ̼���ƣ����ݵζ���������c������Һ��=
�����ı�Һ�࣬���ⶨ�������ȴ��Ȼ�ƫ�ߣ�
�ʴ�Ϊ����̪����Һ��Ϊ��ɫ����30s�ڲ���ɫ��
����֪��������������л��ܼ�A�У�ȡ5.39g��������Ʒ�������������л��ܼ�A�������ˡ�ϴ�ӡ������ʣ�����5.26g��ʣ�����Ϊ���ᱵ�������ȣ��������Ⱥ����ᱵ�����ʵ���Ϊx��y������������Ϊ5.26g������212x+261y=5.26�٣���̼�����Ӧ��ֻ�б����Ӻ������ӣ�Sr2++CO32-��SrCO3����Ba2++CO32-��BaCO3��������Ũ��Ϊ0.107mol/L��̼������Һ22.98mL�����ʵ���Ϊ0.00246mol������x+y=0.0246�ڣ��ɢ٢�ʽ��m�������ȣ�=212x=5.02g�������ȵ���������=
��100%=0.93�����ζ�ǰ��Ʒ��Ca��NO3��2û�г������ζ���Һʱ����̼������Һ�࣬�������������ʵ���ƫ�����ⶨ�������ȴ��Ƚ���ƫ�ߣ�
�ʴ�Ϊ��0.93��ƫ��
��1��Ũ�������������Ϊ65%���ܶ�Ϊ1.4g/cm3��Ҫ����30%ϡ����500mL��v��Ũ���ᣩ���ѣ�Ũ���ᣩ��Ũ�������������=m��ϡ���ᣩ��ϡ���������������m��ϡ���ᣩ=v��ϡ���ᣩ���ѣ�ϡ���ᣩ���ʻ���Ҫ����ϡ������ܶȣ������ƹ����в�ʹ����ƽ�������Ҫ���������Ϊv��Ũ���ᣩ��v��ϡ���ᣩ����ʵ����ȡ��Ӧ�ǣ�Ũ��������ˮ�����������ʹ�õ���������Ͳ���ձ�����������
�ʴ�Ϊ��30%ϡ������ܶȣ�Ũ���������ˮ���������Ͳ���ձ�����������
��2���ɱ��е����ݿ��Կ�����Sr��NO3��2���ܽ�������¶ȵ����߱仯��������Ƶ��ܽ�����¶ȱ仯�ϴ���˿�ͨ���ɽ�ȡ��õ��Ļ�����Ʊ������ȴ�Ʒ��ʵ�鲽������Ϊ�������ᾧ�����ȹ��ˣ�ϴ�ӣ�����õ���
�ʴ�Ϊ�������ᾧ�����ȹ��ˣ�
��3���ٷ����ζ����̿�֪����Ʒ��Һ��ɫ������̼���Ƴ�����ȫ���Ե����̪��Һָʾ�յ㣬�������һ����Һ�ʺ�ɫ��������ڲ���ɫ�������ζ�ǰ��Ʒ��Ca��NO3��2û�г����������ı���Һ̼���ƣ����ݵζ���������c������Һ��=
| c(��Һ)v(��Һ) |
| v(����Һ) |
�ʴ�Ϊ����̪����Һ��Ϊ��ɫ����30s�ڲ���ɫ��
����֪��������������л��ܼ�A�У�ȡ5.39g��������Ʒ�������������л��ܼ�A�������ˡ�ϴ�ӡ������ʣ�����5.26g��ʣ�����Ϊ���ᱵ�������ȣ��������Ⱥ����ᱵ�����ʵ���Ϊx��y������������Ϊ5.26g������212x+261y=5.26�٣���̼�����Ӧ��ֻ�б����Ӻ������ӣ�Sr2++CO32-��SrCO3����Ba2++CO32-��BaCO3��������Ũ��Ϊ0.107mol/L��̼������Һ22.98mL�����ʵ���Ϊ0.00246mol������x+y=0.0246�ڣ��ɢ٢�ʽ��m�������ȣ�=212x=5.02g�������ȵ���������=
| 5.02g |
| 5.39g |
�ʴ�Ϊ��0.93��ƫ��
���������⿼���������Ʊ�ʵ�鷽���ķ����жϣ�������������̣���ѧ��Ӧ���ʵ�Ӱ�����ط������ζ�ʵ��IJ����ָʾ��ѡ��������Ӧ�ã�

��ϰ��ϵ�д�
 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д�
�����Ŀ
������Ϊ�Ƚ�Ԫ��ԭ�ӵ�ʧ�����������ǿ���������ǣ�������
| A��Ԫ����̬�⻯����ȶ��� |
| B��Ԫ�ص����ۡ��е�ߵ� |
| C�������䷢�����û� |
| D���ǽ��������ӵĻ�ԭ��ǿ�� |
 ��������Ȼ�̼��Һ��Ӧ���ɲ�������� �������������칹����������
��������Ȼ�̼��Һ��Ӧ���ɲ�������� �������������칹����������| A��1�� | B��2�� | C��3�� | D��4�� |
����ʵ���������ȷ���ǣ�������
| A��NaCl��Һ�����ᾧʱ�����������о���������ʣ������Һ��ʱ��ֹͣ���� |
| B����ȥFe��OH��3�����л��е�Cl-���ӣ����������ķ��� |
| C���������ʱ��Ӧʹ�¶ȼ�ˮ����������ƿ��֧�ܿڴ� |
| D�������Ȼ�̼��ȡ��ʱ��������Ȼ�̼��Һ�ӷ�Һ©���Ͽڵ��� |