��Ŀ����
6���Ȼ���ͭ��CuCl���㷺Ӧ���ڻ�����ӡȾ����Ƶ���ҵ��CuCl�����ڴ���ˮ��������������Ũ�Ƚϴ����ϵ���ڳ�ʪ��������ˮ���������Ժ���ͭ����Ҫ�ɷ���Cu������CuO��Ϊԭ�ϣ���������������ֽ⼼������CuCl�Ĺ��չ�����ͼ��
�ش��������⣺
��1��д�����ܽ⡱�����з�����������ԭ��Ӧ�����ӷ���ʽ4Cu+NO3-+10H+=4Cu2++NH4++3H2O��NԪ�ر���ԭ����ͼۣ����ޣ��ܽ��¶�Ӧ������60-70�棬ԭ�����¶ȵ��ܽ��ٶ������¶ȹ�����ηֽ⣮
��2��д��������У��������뻹ԭ�������ʵ���֮��Ϊ2��1��
��3������ݰ�����pH=2����ϴ��ˮϴ������������ϴ���õ��������� ��д���ƣ������յ���Ҫ�����ǣ�NH4��2SO4 ��д��ѧʽ��
��4�����������У������ϴ�������Ǵ�ϴ�����ڼӿ��ȥCuCl����ˮ�ַ�ֹ��ˮ��������
��5����ȡ2.000g CuCl��Ʒ�����ʲ����뷴Ӧ��������һ������0.5000mol/L FeCl3��Һ�У���ˮϡ����200mL�õ����ڣ�ȡ20.00mL��0.1000mol/L��Ce��SO4��2��Һ�ζ����յ㣬����20.00mL Ce��SO4��2�йط�Ӧʽ��Fe3++CuCl�TFe2++Cu2++Cl- Ce4++Fe2+�TFe3++Ce3+������Ʒ��CuCl����������99.50%��
���� ������������������Ӿ��������ԣ�����������ͭ����Ҫ�ɷ���Cu������CuO����������ͭ�����˺�����Һ�м���������立���������ԭ��Ӧ����CuCl������2Cu2++SO32-+2Cl-+H2O=2CuCl+SO42-+2H+���õ���CuCl��������ϴ��ˮϴ�������Ҵ�ϴ�ӣ��ɿ��ٳ�ȥ��������ˮ�֣���ֹˮ�⡢����������ڡ��ܡ��ݡ��Ҫ���й�Һ���룬���ݻ�ϵ�ˮ���Խ��з��룬û�з�����ѧ�仯���������Ļ��Լ���ʽѹ�˻����Եõ��˱�����ϴ�ɿ��ٳ�ȥ��������ˮ�֡���ֹˮ�⣬��ɵõ�CuCl���Դ��������⣮
��� �⣺��1�������������������������������Ӿ��������ԣ�������Cu����CuSO4�����ӷ�ӦΪ4Cu+NO3-+10H+=4Cu2++NH4++3H2O���ܽ��¶�Ӧ������60-70�棬ԭ�����¶ȵ��ܽ��ٶ������¶ȹ�����ηֽ⣬
�ʴ�Ϊ��4Cu+NO3-+10H+=4Cu2++NH4++3H2O���¶ȵ��ܽ��ٶ������¶ȹ�����ηֽ⣻
��2��������з�ӦΪͭ������������立���������ԭ��Ӧ����CuCl������2Cu2++SO32-+2Cl-+H2O=2CuCl+SO42-+2H+������������Ϊͭ���ӣ���ԭ��Ϊ������泥��������뻹ԭ�������ʵ���֮��Ϊ2��1��
�ʴ�Ϊ��2��1��
��3�������̿�֪������ϴ��ˮϴ��õ�����泥���Ӧ�������ᣬΪ��ֹCuCl�ܽ⣬���ܼ���������������ᣬҲ���ܼ������ᣬ��ֹ���������ʣ�������ͼ��֪������炙��տ�ѭ��ʹ�ã�
�ʴ�Ϊ�������NH4��2SO4��
��4�������Ϊ��ϴ�����Ҵ��е�ͣ��ӷ��������Ҵ�ϴ�ӣ��ɿ��ٳ�ȥ��������ˮ�֣���ֹˮ�⡢������
�ʴ�Ϊ����ϴ�����ڼӿ��ȥCuCl����ˮ�ַ�ֹ��ˮ��������
��5��Fe3++CuCl=Fe2++Cu2++Cl-��Ce4++Fe2+=Fe3++Ce3+�ɵõ�Ce4+��Fe2+��CuCl��n��Ce4+��=0.1000mol•L-1��0.02L=0.002mol����2.000g��Ʒ�к���n��CuCl��=10��0.002mol=0.02mol������m��CuCl��=0.02mol��99.5g/mol=1.99g�������Ʒ��CuCl����������=$\frac{1.99g}{2.000g}$��100%=99.50%��
�ʴ�Ϊ��99.50%��
���� ����ѧ���Ķ���ȡ��Ϣ�����������û�ѧ���������ԭ�ζ���Ӧ�õȣ�������ѧ���ķ���������ʵ�������ͼ��������Ŀ��飬ע��������ʵ������Լ���Ӧ���̣��Ѷ��еȣ�ע�⣨6�������ù�ϵʽ�ļ��㣮

 �ϴ�̸�������������νӽ̳��Ͼ���ѧ������ϵ�д�
�ϴ�̸�������������νӽ̳��Ͼ���ѧ������ϵ�д�| A�� | ������Һ��Cu2+��Fe3+��NO3-��I- | |
| B�� | �������軯����Ѫ��ɫ����Һ��K+��NH4+��Cl-��NO3- | |
| C�� | ����Al�ܷų�����H2����Һ�У�NH4+��Fe2+��NO3-��SO42- | |
| D�� | �����£�c��H+��/c��OH-��=1��1012����Һ��K+��AlO2-��CO32-��Na+ |
| A�� | H2O2������ֻ���ڼ��Լ� | B�� | NH3�ĽṹʽΪ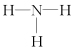 | ||
| C�� | HCl�ĵ���ʽΪ | D�� | ÿ��ˮ�����ں���2����� |
| A�� | Ԫ��Y����������ϼ�Ϊ+6 | |
| B�� | ���Ӱ뾶�Ĵ�С˳��ΪW��Q��Z��X��Y | |
| C�� | �����£�1molQ���ʺ�������NaOH��Һ��Ӧת��1mol���� | |
| D�� | Ԫ��W������������Ӧ��ˮ�������Ա�Q��ǿ |
| A�� | ����ͱ�ϩ�������2��1 | B�� | �������ϩ�������2��1 | ||
| C�� | C2H6��C2H2�������Ϊ1��1 | D�� | C2H6��C2H2�������2��1 |
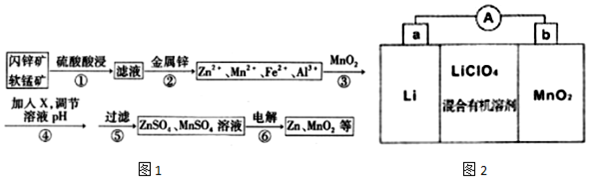
��֪��I����ʯ�����н���Ԫ������Һ�о���������ʽ���ڣ�
���ֽ���������ȫ������pH�����
| Zn2+ | Mn2+ | Fe2+ | Fe3+ | Al3+ | |
| pH | 8.0 | 10.1 | 9.0 | 3.2 | 4.7 |
��1��������з��������Ӧ������MnO2��FeS�����Ṳ��ʱ�е���ɫ������������Һ��Ϊ�ػ�ɫ��д��MnO2��FeS�����Ṳ�ȷ�����Ӧ�Ļ�ѧ����ʽ3MnO2+2FeS+6H2SO4$\frac{\underline{\;\;��\;\;}}{\;}$3MnSO4+Fe2��SO4��3+2S+6H2O��
��2������ڼ������п��Ϊ�˻��ս��������ս�������Ҫ�ɷ���Cu��Cd��
��3���������MnO2����������ԭ��Ӧ�����ӷ���ʽMnO2+2Fe2++4H+=2Fe3++Mn 2++2H2O������X������BD������ĸ��ʾ��
A��Cu2��OH��2CO3 B��MnCO3 C��MgO D��Zn��OH��2
��4�������п���ѭ�����õ�������H2SO4���ѧʽ����
��5��MnO2��Li����LiMnO2���õ�ط�Ӧԭ����ͼ2��ʾ�����е����LiClO4�����ڻ���л��ܼ��У�Li+ͨ�������Ǩ����MnO2�����У�����LiMnO2���ش��������⣺
�����·�еĵ����ƶ���������a������b��������ĸ����
��д��������ӵ�ص������缫��ӦʽMnMnO2+e-+Li+=LiMnO2��
��6����֪��25��ʱ��HCN�ĵ��볣��K=4.9��10-10��H2S�ĵ��볣��K1=1.3��10-7��K2=7.0��10-15����NaCN��Һ��ͨ��������H2S���壬�÷�Ӧ�Ļ�ѧ����ʽΪNaCN+H2S=HCN+NaHS��
����MΪ����Ͻ�MHΪ��������ԭ�ӵĴ���Ͻ�KOH��Һ�����Һ�����������أ�����˵������ȷ���ǣ�������
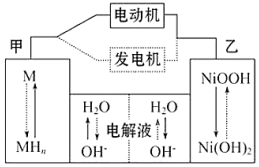
| A�� | ���ʱ����������pH���� | |
| B�� | �綯������ʱ��Һ��OH-����ƶ� | |
| C�� | �ŵ�ʱ������ӦʽΪ��NiOOH+H2O+e-�TNi��OH��2+OH- | |
| D�� | �缫�ܷ�ӦʽΪ��MH+NiOOH$?_{���}^{�ŵ�}$M+Ni��OH��2 |
| A�� | �ں�A13+����ΪNA��A1C13��Һ�У�C1-����Ϊ3NA | |
| B�� | ��״���£�5.6L CO2�к��еĹ��õ��Ӷ���Ϊ0.5NA | |
| C�� | Na2O2��������CO2��Ӧ����0.1mol O2��ת�Ƶĵ�����Ϊ0.2NA | |
| D�� | �����£�56g��ƬͶ������ŨH2SO4�У���ַ�Ӧ������NA��SO2���� |
 ��Ӧ���ͼӾ۷�Ӧ
��Ӧ���ͼӾ۷�Ӧ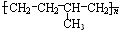 ����ṹ��ʽΪCH2=CH2��CH3-CH=CH2��
����ṹ��ʽΪCH2=CH2��CH3-CH=CH2��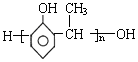 ����ṹ��ʽΪ
����ṹ��ʽΪ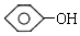 ��CH3CHO��
��CH3CHO��