��Ŀ����
4��ij�����A������Al2��SO4��3��Al2O3��Fe2O3����һ�������¿�ʵ����ͼ��ʾ�ı仯��
��ش��������⣮
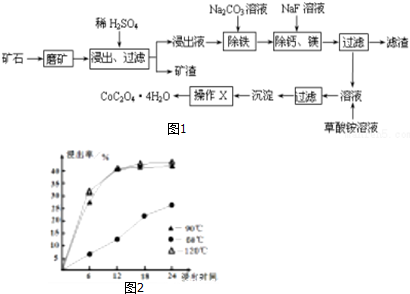
��1��ͼ���漰������Һ������ķ����ǹ��ˣ�
��2��B��C��D��E 4�����ʵĻ�ѧʽΪ��
BAl2O3��CFe2O3��DNaAlO2��EAl��OH��3��
��3������F��NaOH��Һ��Ӧ�����ӷ���ʽΪAl2O3+2OH-=2AlO2-+H2O��
����E��ϡ���ᷴӦ�����ӷ���ʽΪAl��OH��3+3H+=Al3++3H2O��
��ҺG�����ϡ��ˮ��Ӧ�Ļ�ѧ����ʽΪAl2��SO4��3+6NH3•H2O=2Al��OH��3��+3��NH4��2SO4��
���� A����Al2��SO4��3��Al2O3��Fe2O3��Al2��SO4��3����ˮ��Al2O3��Fe2O3��������ˮ�������A��ˮ�ܽ����Һ��G�к�Al2��SO4��3������F��Al2O3��Fe2O3�������F�м�NaOH��Һ��Fe2O3����Ӧ��Al2O3����NaOH��Һ��Ӧ����NaAlO2����CΪFe2O3����ҺDΪNaAlO2��Һ������ҺG�мӹ�����ˮ����Һ�������ˮ��Ӧ��Al3+���������õ�EΪAl��OH��3��Al��OH��3��������BΪAl2O3���Դ������
��� �⣺A����Al2��SO4��3��Al2O3��Fe2O3��Al2��SO4��3����ˮ��Al2O3��Fe2O3��������ˮ�������A��ˮ�ܽ����Һ��G�к�Al2��SO4��3������F��Al2O3��Fe2O3�������F�м�NaOH��Һ��Fe2O3����Ӧ��Al2O3����NaOH��Һ��Ӧ����NaAlO2����CΪFe2O3����ҺDΪNaAlO2��Һ������ҺG�мӹ�����ˮ����Һ�������ˮ��Ӧ��Al3+���������õ�EΪAl��OH��3��Al��OH��3��������BΪAl2O3��
��1��������Һ������ķ����ǹ��ˣ��ʴ�Ϊ�����ˣ�
��2��������������֪��BΪAl2O3��CΪFe2O3��DΪNaAlO2��EΪAl��OH��3��
�ʴ�Ϊ��Al2O3��Fe2O3��NaAlO2��Al��OH��3��
��3��F��Al2O3��Fe2O3������F��NaOH��Һ��Ӧ�����ӷ���ʽΪAl2O3+2OH-=2AlO2-+H2O��
EΪAl��OH��3������E��ϡ���ᷴӦ�����ӷ���ʽΪ Al��OH��3+3H+=Al3++3H2O��
G�к�Al2��SO4��3����ҺG�����ϡ��ˮ��Ӧ�Ļ�ѧ����ʽΪAl2��SO4��3+6NH3•H2O=2Al��OH��3��+3��NH4��2SO4��
�ʴ�Ϊ��Al2O3+2OH-=2AlO2-+H2O��Al��OH��3+3H+=Al3++3H2O��Al2��SO4��3+6NH3•H2O=2Al��OH��3��+3��NH4��2SO4��
���� ���⿼��������ƶϣ�Ϊ�߿��������ͣ������ڿ���ѧ���ķ����������ƶ��������������ʵ����ʼ������ķ�ӦΪ���Ĺؼ���ע���������������Լ�Ԫ�ػ�����֪ʶ���ۺ�Ӧ�ã���Ŀ�ѶȲ���

 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д�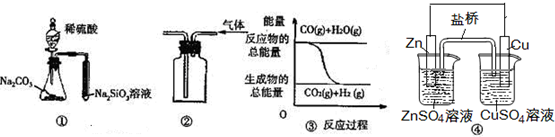
| A�� | ͼ ���е�װ�ÿ��γɳ����ȶ��ĵ�������пƬ����CuSO4��Һ�з����Ļ�ѧ��Ӧ��ͼ ���е�ԭ���װ�÷�Ӧ����ͬ | |
| B�� | ��ͼ ����ʾʵ��װ���ſ������ռ�H2���� | |
| C�� | ͼ �۱�ʾ���淴Ӧ��CO��g��+H2O��g��?CO2��g��+H2��g����Ϊ���ȷ�Ӧ | |
| D�� | ��ͼ ����ʾʵ��ɱȽ���̼��������Ԫ�صķǽ�����ǿ�� |
| A�� | Ϊ����±�����е�±ԭ�ӣ��ȼ���NaOH��Һ���ȣ��ټ���AgNO3��Һ�۲���ɫ�仯 | |
| B�� | Ϊ��ȡ������������ϡH2SO4���Ҵ��������ϼ��ȣ��ų�������ͨ��ʢ��NaOH��Һ���Թ��� | |
| C�� | Ϊ����ȩ���Ĵ��ڣ������Թ��м���2mL2%��NaOH��Һ������CuSO4��Һ���ټ���ȩ����� | |
| D�� | Ϊ��ȡ��ϩ����ϡH2SO4���Ҵ���ϼ��� |
| A�� | �������� | B�� | �� | C�� | �Ҵ� | D�� | ���� |
| A�� | BCl3 | B�� | NCl3 | C�� | H2S | D�� | BeCl2 |