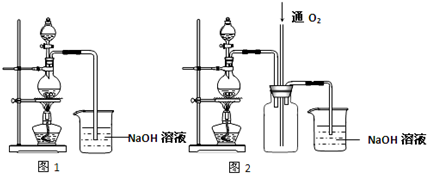
��Ŀ����
��1����һ���¶Ⱥ�ѹǿ�£�1���X2��g����3���Y2��g����������2���Z��g������Z����Ļ�ѧʽ�� ��
��2����ͬ�����£���1molCl2��1molO2�Ļ������22.4L����û�������ƽ����Է�������Ϊ ��
��3���������0.5mol/L��������Һ�����ơ�����þ���������У������ӣ�SO42-���ĸ������� ��
��4��1mol/L��H2SO4��Һ1000ml��0.5molNa2CO3��Ӧ�������ɱ���µ��������Ϊ ��
��2����ͬ�����£���1molCl2��1molO2�Ļ������22.4L����û�������ƽ����Է�������Ϊ
��3���������0.5mol/L��������Һ�����ơ�����þ���������У������ӣ�SO42-���ĸ�������
��4��1mol/L��H2SO4��Һ1000ml��0.5molNa2CO3��Ӧ�������ɱ���µ��������Ϊ
���㣺���ʵ�������ؼ���
ר�⣺������
��������1��һ���¶���ѹǿ�£���������֮�ȵ������ʵ���֮�ȣ����ڻ�ѧ������֮�ȣ�Ȼ�����������غ㶨����ȷ����ѧʽ��
��2���������ʵ������ԼӺͼ������������ʵ��������ݱ��������Ħ��������ݼ�������������������m=nM����1molCl2��1mol O2������������ƽ��Ħ�����������������������������ܵ����ʵ��������������ƽ��Ħ�������������������������������������������
��3���������Ũ�ȵIJ�ͬ���ʣ������ӣ�SO42-���ĸ����Ⱦ͵��ڻ�ѧʽ�е�SO42-�ĸ�����
��4������n=cV����n��H2SO4�������ݷ���ʽ�������ɶ�����̼���
��2���������ʵ������ԼӺͼ������������ʵ��������ݱ��������Ħ��������ݼ�������������������m=nM����1molCl2��1mol O2������������ƽ��Ħ�����������������������������ܵ����ʵ��������������ƽ��Ħ�������������������������������������������
��3���������Ũ�ȵIJ�ͬ���ʣ������ӣ�SO42-���ĸ����Ⱦ͵��ڻ�ѧʽ�е�SO42-�ĸ�����
��4������n=cV����n��H2SO4�������ݷ���ʽ�������ɶ�����̼���
���
�⣺��1��һ���¶Ⱥ�ѹǿ�£�1���X2������3���Y2���廯������2������廯�����û�����ΪZ��
����������֮�ȵ������ʵ���֮�ȣ����ڻ�ѧ������֮�ȣ�
��X2+3Y2�T2Z����ԭ���غ��֪��ZΪXY3��
�ʴ�Ϊ��XY3��
��2��1molCl2��1molO2��Ϻ����ʵ���Ϊ��1mol+1mol=2mol��1molCl2������Ϊ��1mol��71g/mol=71g��1mol����������Ϊ��1mol��32g/mol=32g���ʻ������ƽ��Ħ������Ϊ
=51.5g/mol�����û�������ƽ����Է�������Ϊ51.5��
�ʴ�Ϊ��51.5��
��3���������Ũ�ȵIJ�ͬ���ʣ������ӣ�SO42-���ĸ����Ⱦ͵��ڻ�ѧʽ�е�SO42-�ĸ����ȣ������ơ�����þ����������ѧʽ�ֱ�Ϊ��Na2SO4��MgSO4��Al2��SO4��3���������ӣ�SO42-���ĸ�������1��1��3��
�ʴ�Ϊ��1��1��3��
��4��n��H2SO4��=1L��1mol/L=1mol��
�ݷ���ʽ��Na2CO3+H2SO4=Na2SO4+CO2��+H2O
1 1 1
Na2CO3���㣬H2SO4��������Na2CO3���㣬������CO2���ʵ���Ϊ0.5mol������µ��������Ϊ0.5mol��22.4mol/L=11.2L��
�ʴ�Ϊ��11.2L
����������֮�ȵ������ʵ���֮�ȣ����ڻ�ѧ������֮�ȣ�
��X2+3Y2�T2Z����ԭ���غ��֪��ZΪXY3��
�ʴ�Ϊ��XY3��
��2��1molCl2��1molO2��Ϻ����ʵ���Ϊ��1mol+1mol=2mol��1molCl2������Ϊ��1mol��71g/mol=71g��1mol����������Ϊ��1mol��32g/mol=32g���ʻ������ƽ��Ħ������Ϊ
| 71g+32g |
| 2mol |
�ʴ�Ϊ��51.5��
��3���������Ũ�ȵIJ�ͬ���ʣ������ӣ�SO42-���ĸ����Ⱦ͵��ڻ�ѧʽ�е�SO42-�ĸ����ȣ������ơ�����þ����������ѧʽ�ֱ�Ϊ��Na2SO4��MgSO4��Al2��SO4��3���������ӣ�SO42-���ĸ�������1��1��3��
�ʴ�Ϊ��1��1��3��
��4��n��H2SO4��=1L��1mol/L=1mol��
�ݷ���ʽ��Na2CO3+H2SO4=Na2SO4+CO2��+H2O
1 1 1
Na2CO3���㣬H2SO4��������Na2CO3���㣬������CO2���ʵ���Ϊ0.5mol������µ��������Ϊ0.5mol��22.4mol/L=11.2L��
�ʴ�Ϊ��11.2L
���������⿼�����ʵ������йؼ��㣬����٤�����ɼ������ۣ�����ʽ���еļ��㣬�漰�������㣬��ȷһ���¶���ѹǿ����������뻯ѧ�������Ĺ�ϵ�ǽ����Ĺؼ�����Ŀ�ѶȲ���

��ϰ��ϵ�д�
�����Ŀ
��������У��γɹ��ۼ���ԭ�ӹ���ǣ�������
| A����ԭ�ӵ�2p�������ԭ�ӵ�1s��� |
| B����ԭ�ӵ�2p�������ԭ�ӵ�2p��� |
| C����ԭ�ӵ�3p�������ԭ�ӵ�1s��� |
| D����ԭ�ӵ�2p�������ԭ�ӵ�3p��� |
��NAΪ�����ӵ�����������������ȷ���ǣ�������
| A��24 gþ��ԭ������������ΪNA |
| B��1 L 0.1 mol?L-1������Һ��H+��Ϊ0.1NA |
| C��1 mol�����������������Ϊ10NA |
| D����״���£�22.4 L�Ҵ��ķ�����ΪNA |
��nAΪ�����ӵ���������ֵ������˵����ȷ���ǣ�������
| A��1mol ������6nA��C-H�� |
| B����״���£�22.4L��ˮ����nA��NH3���� |
| C��18gH2O����10nA������ |
| D��56g��ƬͶ������ŨH2SO4������nA��SO2���� |
Ϊ��������ƿ��ɫ��Һ��HCl��AlCl3��Ba��NO3��2��NaCl��Na2CO3����λͬѧ��û�þƾ��ƣ���λͬѧ�������Լ���һλͬѧû�������κ��Լ������в�������һ�����ٵ��ǣ�������
| A���������˷�̪��Һ |
| B����������NaOH��Һ |
| C����������ʯ����Һ |
| D�����������κ��Լ� |
���б��﷽ʽ��ȷ���ǣ�������
A����̬̼ԭ�ӵļ۵����Ų�ͼ��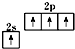 |
| B��HClO�Ľṹʽ��H-Cl-O |
| C����̬26Fe�ļ۵����Ų�ʽ��3d64s2 |
D����̬ͭԭ�ӵļ۵����Ų�ͼ��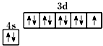 |