��Ŀ����
5�� ������һ�ּ��߷�չDZ���������Դ��
������һ�ּ��߷�չDZ���������Դ����1�����ѭ���ֽ�ˮ�����DZ�������ⷽ�����䷴Ӧԭ�����£�
��SO2+2H2O+I2=H2SO4+2HI
��.2H2SO4$\frac{\underline{\;\;��\;\;}}{\;}$2SO2+O2+2H2O
��.2HI$\frac{\underline{\;\;��\;\;}}{\;}$H2+I2��g��
��ӦI�Ļ�ԭ����SO2���ѧʽ�������ѭ���ֽ�ˮ�ܷ�Ӧ�Ļ�ѧ����ʽΪ2H2O$\frac{\underline{����}}{��}$2H2��+O2����
��2������һ��������������ϣ����ڲ�ͬ�����·ֽⷽʽ��ͬ�� �ڸ����£�N2H4����ȫ�ֽ�ΪNH3��N2��H2��ʵ���÷ֽ������N2��H2���ʵ����仯��ͼ��ʾ���÷ֽⷴӦ����ʽΪ7N2H4$\frac{\underline{\;����\;}}{\;}$8NH3+3N2+2H2��
��3��̼���£���Է�������Ϊ90������DEC��N2H4����ȡ����Ӧ�Ƶõģ���֪DEC�Ľṹ��ʽΪ
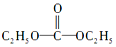 ����̼���µĽṹ��ʽΪ
����̼���µĽṹ��ʽΪ ��
��
���� ��1��SO2+2H2O+I2=H2SO4+2HI�ж�����������Ļ��ϼ������ǻ�ԭ������+��+��õ�ѭ���ֽ�ˮ�ܷ�Ӧ�Ļ�ѧ����ʽ��
��2���ڸ����£�N2H4����ȫ�ֽ�ΪNH3��N2��H2�����ԷֽⷴӦ����ʽΪ��7N2H4$\frac{\underline{\;����\;}}{\;}$8NH3+3N2+2H2��
��3��̼���£���Է�������Ϊ90������DEC��N2H4����ȡ����Ӧ�Ƶõģ�����̼���µĽṹ��ʽΪ ��
��
��� �⣺��1��SO2+2H2O+I2=H2SO4+2HI�ж�����������Ļ��ϼ������ǻ�ԭ������+��+��õ�ѭ���ֽ�ˮ�ܷ�Ӧ�Ļ�ѧ����ʽ�����Է���ʽΪ��2H2O$\frac{\underline{����}}{��}$2H2��+O2�����ʴ�Ϊ��SO2��2H2O$\frac{\underline{����}}{��}$2H2��+O2����
��2���ڸ����£�N2H4����ȫ�ֽ�ΪNH3��N2��H2�����ԷֽⷴӦ����ʽΪ��7N2H4$\frac{\underline{\;����\;}}{\;}$8NH3+3N2+2H2���ʴ�Ϊ��7N2H4$\frac{\underline{\;����\;}}{\;}$8NH3+3N2+2H2��
��3��̼���£���Է�������Ϊ90������DEC��N2H4����ȡ����Ӧ�Ƶõģ�����̼���µĽṹ��ʽΪ ���ʴ�Ϊ��
���ʴ�Ϊ�� ��
��
���� ���⿼��������ԭ��Ӧ����ѧ����ʽ����д���ṹʽ������ȣ��ۺ���ǿ������һ�����Ѷȣ�

| A�� | ��ͭ˿����ϡ�����У�Cu+4H++2NO3-�TCu2++2NO2��+2H2O | |
| B�� | ��Fe2��SO4��3��Һ�м���������ۣ�Fe3++Fe�T2Fe2+ | |
| C�� | ��Al2��SO4��3��Һ�м��������ˮ��Al3++3NH3•H2O�TAl��OH��3��+3NH4+ | |
| D�� | ϡH2SO4��Ba��OH��2��Һ��Ӧ��H++SO42-+Ba2++OH-�TBaSO4��+H2O |
| A�� | C+H2O$\frac{\underline{\;\;��\;\;}}{\;}$CO+H2 | B�� | H2+CuO$\frac{\underline{\;\;��\;\;}}{\;}$Cu+H2O | ||
| C�� | Cl2+H2O?HCl+HClO | D�� | SO3+H2O=H2SO4 |
| A�� | .���³�ѹ�£�18 g H2O�к��еĵ�������Ϊ10NA | |
| B�� | .1 mol•L-1 NaCl��Һ����NA��Na+ | |
| C�� | ���³�ѹ�£�92 g��NO2��N2O4������庬�е�ԭ����Ϊ3 NA | |
| D�� | ��״���£�2.24LCH4����������ΪNA |
| A�� | ��ͥ�о�����ʳ������ˮ����ˮ�� | |
| B�� | ���ͼ�ˮ�������� | |
| C�� | ��ͨ�����������ظ���ؼ��˾���Ƿ�ƺ�ݳ� | |
| D�� | ����ʱ�������������Ͼƺ�ʳ�ɼ�����ζ��������ζ |
| A�� | +2��+2 | B�� | +2��+3 | C�� | +2��+4 | D�� | +3��+3 |
 2HI��g�� ��H��0������ƽ�⡣HI���������w��HI����ʱ��仯��ͼ���ߣ�����ʾ��
2HI��g�� ��H��0������ƽ�⡣HI���������w��HI����ʱ��仯��ͼ���ߣ�����ʾ��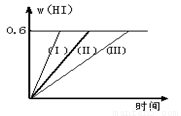
 An��g����ƽ�ⳣ��ΪK������1/2An ��g��
An��g����ƽ�ⳣ��ΪK������1/2An ��g��