��Ŀ����
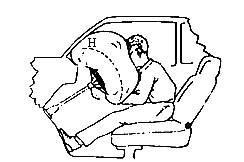
��һ��������ȫ�������ڲ����л�ѧ���ʣ�����������ǰ���ܵ�ײ��ʱ����ѧ���ʻ���˲�䱬��������������������������˾���ͳ�ǰ���粣����������֮�䣬��ֹ˾�����ˡ���
��1������������������泥�NH4NO3�����������ҳ������Ѹ�ٷֽ⣬����������һ�����������壬��д���仯ѧ����ʽ��
��2����Ҫ����30 dm3��һ������������Ҫ��������Ħ��������泥�
��3����ij���¹��У������ٶ���35 m��s-1��˾������������0.2 sֹͣ�˶���˾����������������Ϊ40 kg���������������ϵ������700 cm2����ʱ���ܵ�������ѹǿ�ж��
��1��2NH4NO3![]() 2N2O��+4H2O
2N2O��+4H2O
��2��1.34 mol
(3)1.0��105 Pa
����:
��1��2NH4NO3![]() 2N2O��+4H2O
2N2O��+4H2O
��2��n(N2O)=![]() =1.34 mol
=1.34 mol
�ɣ�1��֪n(NH4NO3)=n(N2O)=1.34 mol
(3)ƽ��ѹǿ��p=![]() F=m
F=m![]()
����p=![]() =
=![]() Pa=1.0��105 Pa
Pa=1.0��105 Pa
ֻ�൱��һ������ѹ���ܰ�ȫ��

��ϰ��ϵ�д�
�����Ŀ