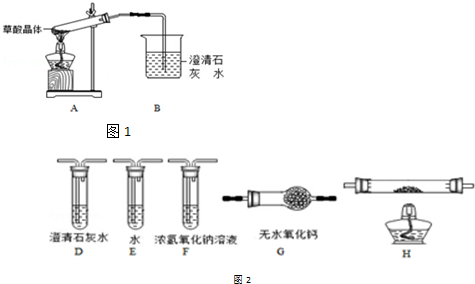
��Ŀ����
10��ij�еش��غ�����ˮ��Դ�ḻ����ˮ��ȡʳ�κ�Br2�Ժ����±���������Ʊ�������MgCl2��MgO����±�к���Mg2+��Cl-������������Na+��Fe2+��Fe3+��SO42-��CO��NH2��2�ȣ��Ʊ�������ͼ1����1�������ijɷ���Fe��OH��3��CaSO4��д��ѧʽ������Һ����������Ҫ������������Na+��д���ӷ��ţ���
��2����NaClO��ȥ����CO��NH2��2ʱ������������⣬�����ܲ������ѭ�������ʣ���÷�Ӧ�Ļ�ѧ����ʽΪ3NaClO+CO��NH2��2=3NaCl+CO2��+N2��+2H2O������NaClO����һ�������Ǽ���NaClO������Fe2+ʹ�����Fe��OH��3��������ȥ��
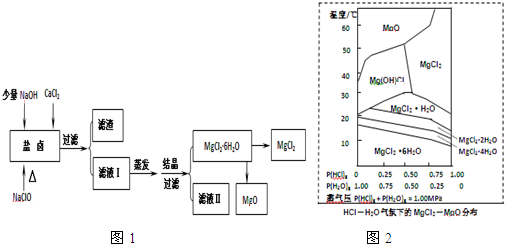
��3���¶Ⱥ�ѹǿP��HCl��g��MgCl2•6H2O�����ȷֽ�����Ӱ����ͼ2��ʾ����ش��������⣺
��д��P��HCl��g=0.25MPa���¶ȴ�300�����ߵ�550��ʱ��Ӧ�Ļ�ѧ����ʽMg��OH��Cl=MgO+HCl����
��ʵ�������У���MgCl2•6H2O������ȵ�600��Ĺ����м����ò�����ˮMgCl2����ԭ����P��HCl��g��С��þ����ˮ������������þ���ò����Ȼ�þ����Ҫ�õ���ˮMgCl2���ȡ�Ĵ�ʩ��ͨ��HCl��������P��HCl��g������þ���ӵ�ˮ�⣮
���� ��1��������±�ijɷ�����������NaClO������Fe2+ʹ�����Fe��OH��3�����������Ȼ��Ƶ��������γ�����Ƴ�������ȥ���е���������ӣ���Һ��ijɷ�Ϊ�Ȼ�þ���Ȼ��ƣ���ȴ�ᾧ���õ��Ȼ�þ���壬��Һ�ijɷ�Ϊ�Ȼ��ƺ��Ȼ�þ��
��2������NaClO������CO��NH2��2��Ӧʱ������Ԫ���غ��Լ����������ѭ�������ʣ���֪��������NaCl������CO2��N2��H2O������NaClO������Fe2+ʹ�����Fe��OH��3��������ȥ��
��3������ͼ��֪P��HCl��g=0.25MPa���¶ȴ�300�����ߵ�550��ʱMgCl2•6H2O������Mg��OH��Clת���MgO��
��������MgCl2•6H2O���壬P��HCl��g��С������þ����ˮ������������þ���ò����Ȼ�þ����Ҫ�õ���ˮ�Ȼ�þ����Ӧͨ��HCl��������P��HCl��g������þ���ӵ�ˮ�⣮
��� �⣺��1��������±�ijɷ�����������NaClO������Fe2+ʹ�����Fe��OH��3�����������Ȼ��Ƶ��������γ�����Ƴ�������ȥ���е���������ӣ����������ijɷ���Fe��OH��3��CaSO4��
��Һ��ijɷ�Ϊ�Ȼ�þ���Ȼ��ƣ���ȴ�ᾧ���õ��Ȼ�þ���壬��Һ�ijɷ�Ϊ�Ȼ��ƺ��Ȼ�þ����Һ������������Ҫ����������Na+��
����Fe��OH��3��CaSO4��Na+��
��2��NaClO������CO��NH2��2��Ӧʱ����������NaCl������CO2��N2��H2O����÷�Ӧ�Ļ�ѧ����ʽΪ3NaClO+CO��NH2��2=3NaCl+CO2��+N2��+2H2O������NaClO����������������Fe2+ʹ�����Fe��OH��3��������ȥ��
�ʴ�Ϊ��3NaClO+CO��NH2��2=3NaCl+CO2��+N2��+2H2O������NaClO������Fe2+ʹ�����Fe��OH��3��������ȥ��
��3������ͼ��֪P��HCl��g=0.25MPa���¶ȴ�300�����ߵ�550��ʱMgCl2•6H2O������Mg��OH��Clת���MgO������ʽΪ��Mg��OH��Cl=MgO+HCl����
�ʴ�Ϊ��Mg��OH��Cl=MgO+HCl����
�ۼ���MgCl2•6H2O���壬P��HCl��g��С��þ����ˮ������������þ���ò����Ȼ�þ����Ҫ�õ���ˮ�Ȼ�þ����Ӧͨ��HCl��������P��HCl��g������þ���ӵ�ˮ�⣬
�ʴ�Ϊ��P��HCl��g��С��þ����ˮ������������þ���ò����Ȼ�þ��ͨ��HCl��������P��HCl��g������þ���ӵ�ˮ�⣮
���� ������Ҫ��������±�����ӵij���������Լ��¶Ⱥ�ѹǿP��HCl��g��MgCl2•6H2O�����ȷֽ�����Ӱ�죬������ѧ���������⣬������Ϣ����������Ŀ�Ѷ��еȣ�

| A�� | �ú������۵���С����ʳƷһ���ܷ��װ�������dz��õ������� | |
| B�� | Ũ�������ʢ������Ͱ�У�˵����������Ũ���ᷴӦ | |
| C�� | �����õ��IJ�������������̨���ձ���©��������������ֽ | |
| D�� | ��������̲�����˵���������ǿ�� |
| A�� | 1mol/LBaCl2��Һ�к��е�Ba2+��ΪNA | |
| B�� | 1molþ�����к��еĵ�����Ϊ2NA | |
| C�� | ���³�ѹ�£�11.2LH2�к���H2��Ϊ0.5NA | |
| D�� | 16gO3�к�����ԭ����ΪNA |
| A�� | 1molNa����ȫ��������Na2O2ʱʧȥ2mole- | |
| B�� | Na2O2������������ߵĹ�������ʹ�ù������漰������ԭ��Ӧ | |
| C�� | ���ȶ��ԣ�NaHCO3��Na2CO3 | |
| D�� | ������������ͷۺͼ���ϴ�Ӽ�����Ҫ�ɷ�֮һ |
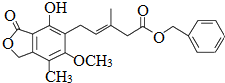 �����й��ڸû������˵������ȷ���ǣ�������
�����й��ڸû������˵������ȷ���ǣ�������| A�� | ���л���ķ���ʽΪC23H24O6 | |
| B�� | 1mol�û�������������9molH2�����ӳɷ�Ӧ | |
| C�� | �ȿ���ʹ������Ȼ�̼��Һ��ɫ���ֿ���ʹ����KMnO4��Һ��ɫ | |
| D�� | �ȿ�����FeCl3��Һ������ɫ��Ӧ���ֿ�����Na2CO3��Һ��Ӧ |
| ���ᴿ���������� | ��ѡ�Լ� |
| Fe ��Al�� | |
| Al2O3��Al�� | |
| FeCl2 ��FeCl3�� | |
| FeCl3 ��FeCl2�� | |
| SiO2 ��CaCO3�� |
��3����400mL2mol•L-1 Ba��OH��2��Һ�У����ʵ����ʵ�����0.8mol������Һ��OH-�����ʵ���Ũ��Ϊ4mol•L-1��Ba2+�����ʵ���Ũ��Ϊ2mol•L-1��