��Ŀ����
N2O4
 2NO2��ƽ�⡣��ͼ�Ǹ�һ��ʱ��ⶨ����N2O4��Ũ�ȣ�������ΪN2O4��Ũ�ȣ�������Ϊʱ�䣩
2NO2��ƽ�⡣��ͼ�Ǹ�һ��ʱ��ⶨ����N2O4��Ũ�ȣ�������ΪN2O4��Ũ�ȣ�������Ϊʱ�䣩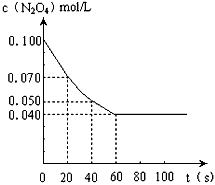
��1��������20��40��ʱ���ڣ�NO2��ƽ����������Ϊ____________mol��L-1��s-1��
��2���÷�Ӧ�Ļ�ѧƽ�ⳣ������ʽΪK=____________��
��3���±��Dz�ͬ�¶��²ⶨ�õ��ĸ÷�Ӧ�Ļ�ѧƽ�ⳣ����

��2��K= C2(NO2)/C(N2O4)
��3������

 ��������ϵ�д�
��������ϵ�д�(14��) ���¶�Ϊ373Kʱ����0.400 mol��ɫ��N2O4����ͨ��4 L���
���ܱ������У����̳��ֺ���ɫ��ֱ������N2O4 2NO2 ��ƽ
�⡣��ͼ�Ǹ�һ��ʱ��ⶨ����N2O4��Ũ�ȣ�������ΪN2O4��Ũ�ȣ�
������Ϊʱ�䣩��1���÷�Ӧ�Ļ�ѧƽ�ⳣ������ʽΪK = �����¶���,Kֵ= ��
��2��������0��20��ʱ���ڣ�NO2��ƽ����������Ϊ mol•L��1•s��1��
��3���±��Dz�ͬ�¶��²ⶨ�õ��ĸ÷�Ӧ�Ļ�ѧƽ�ⳣ����
| T/ K | 323 | 373 |
| Kֵ | 0.022 | �� |
�ݴ˿��Ʋ�÷�Ӧ������NO2���� ��Ӧ��ѡ����ȡ����ȡ�����
��4���������������䣬��Ӧ��373Kʱ�ﵽƽ���N2O4��NO2��Ũ�ȶ�������0.4mol/L��ƽ�⽫�� ����������桱����Ӧ�����ƶ���
(5) ��Ӧ��373Kʱ�ﵽƽ��ʱ��N2O4��ת����=_________________,������Ӧ�������ݻ���СΪԭ����һ�룬��N2O4��ת���ʽ�_________(ѡ���������С�����䡱)
 ���¶�Ϊ373Kʱ����0.100mol��ɫ��N2O4�������1L��յ��ܱ������У����̳��ֺ���ɫ��ֱ������N2O4?2NO2��ƽ�⣮��ͼ�Ǹ�һ��ʱ��ⶨ����N2O4��Ũ�ȣ�������ΪN2O4��Ũ�ȣ�������Ϊʱ�䣩
���¶�Ϊ373Kʱ����0.100mol��ɫ��N2O4�������1L��յ��ܱ������У����̳��ֺ���ɫ��ֱ������N2O4?2NO2��ƽ�⣮��ͼ�Ǹ�һ��ʱ��ⶨ����N2O4��Ũ�ȣ�������ΪN2O4��Ũ�ȣ�������Ϊʱ�䣩 ���¶�Ϊ373Kʱ����0.100mol��ɫ��N2O4�������1L��յ��ܱ������У����̳��ֺ���ɫ��ֱ������N2O4
���¶�Ϊ373Kʱ����0.100mol��ɫ��N2O4�������1L��յ��ܱ������У����̳��ֺ���ɫ��ֱ������N2O4  2NO2��ƽ�⣮��ͼ�Ǹ�һ��ʱ��ⶨ����N2O4��Ũ�ȣ�������ΪN2O4��Ũ�ȣ�������Ϊʱ�䣩
2NO2��ƽ�⣮��ͼ�Ǹ�һ��ʱ��ⶨ����N2O4��Ũ�ȣ�������ΪN2O4��Ũ�ȣ�������Ϊʱ�䣩 N2O4(g)
.��Ӧ���е�2sʱ��N2O4����Ϊ0.05mol�����е�60sʱ���ﵽ�������·�Ӧ������ȣ���ʱ�����л�������ƽ����Է�������Ϊ47.5����
N2O4(g)
.��Ӧ���е�2sʱ��N2O4����Ϊ0.05mol�����е�60sʱ���ﵽ�������·�Ӧ������ȣ���ʱ�����л�������ƽ����Է�������Ϊ47.5����