��Ŀ����
11��A��B��D��E��FΪԭ������������������ֶ���������Ԫ�أ��ǽ���Ԫ��A����������������������ͬ��B����������������������������2����B������D�����г��ȼ������������ۻ�����BD2��E+��D2-������ͬ�ĵ�������A������F������ȼ�գ���������ˮ�õ�һ��ǿ�ᣮ����Ӧ�Ļ�ѧ����ش��������⣺��1��A�����ڱ��е�λ���ǵ�һ����IA�壮AFD�ĵ���ʽΪ
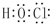 ��
����2��BD2�ĽṹʽΪO=C=O�� BD2�γɾ��������Ϊ���Ӿ��壮
��3��Ԫ��D��E��F�γɵļ����ӵİ뾶�ɴ�СΪCl-��O2-��Na+�������ӷ��ű�ʾ����
��4��д��ʵ�����Ʊ�����F�����ӷ���ʽMnO2+4H++2Cl-$\frac{\underline{\;\;��\;\;}}{\;}$Mn2++Cl2��+2H2O��
��5��д��E��D�γɵļȺ����Ӽ��ֺ����ۼ��Ļ�������ˮ��Ӧ�Ļ�ѧ����ʽ2Na2O2+2H2O=4NaOH+O2����
���� A��B��C��D��E��FΪԭ������������������ֶ���������Ԫ�أ��ǽ���Ԫ��A����������������������ͬ����AΪHԪ�أ�E+��D2-������ͬ�ĵ���������E����IA�壬D���ڢ�A�壬��EΪNa��DΪOԪ�أ�B��D�г��ȼ����������ۻ�����BD2��B����������ϼ�Ϊ+4��B����������������������������2������B���ڵڶ����ڣ���BΪ̼Ԫ�أ�C��ԭ����������̼����֮�䣬��CΪNԪ�أ�A��F��ȼ�գ���������ˮ�õ���ǿ�ᣬ��FΪCl���ݴ˽��
��� �⣺A��B��C��D��E��FΪԭ������������������ֶ���������Ԫ�أ��ǽ���Ԫ��A����������������������ͬ����AΪHԪ�أ�E+��D2-������ͬ�ĵ���������E����IA�壬D���ڢ�A�壬��EΪNa��DΪOԪ�أ�B��D�г��ȼ����������ۻ�����BD2��B����������ϼ�Ϊ+4��B����������������������������2������B���ڵڶ����ڣ���BΪ̼Ԫ�أ�C��ԭ����������̼����֮�䣬��CΪNԪ�أ�A��F��ȼ�գ���������ˮ�õ���ǿ�ᣬ��FΪCl��
��1��AΪHԪ�أ������ڱ��е�λ���ǣ���һ����IA�壮AFD�Ļ�ѧʽΪHClO������ʽΪ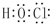 ��
��
�ʴ�Ϊ����һ����IA�壻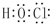 ��
��
��2��BD2�Ļ�ѧʽΪCO2���ṹʽΪO=C=O�����ڷ��Ӿ��壬
�ʴ�Ϊ��O=C=O�����Ӿ��壻
��3��Ԫ��D��E��F�γɵļ����ӷֱ�ΪO2-��Na+��Cl-�����Ӳ�ṹ��ͬ�����ӣ��˵����Խ�����Ӱ뾶ԽС�����ӵ��Ӳ�Խ�����Ӱ뾶Խ�����Ӱ뾶��Cl-��O2-��Na+��
�ʴ�Ϊ��Cl-��O2-��Na+��
��4��ʵ�����Ʊ����������ӷ���ʽ��MnO2+4H++2Cl-$\frac{\underline{\;\;��\;\;}}{\;}$Mn2++Cl2��+2H2O��
�ʴ�Ϊ��MnO2+4H++2Cl-$\frac{\underline{\;\;��\;\;}}{\;}$Mn2++Cl2��+2H2O��
��5��Na��O�γɵļȺ����Ӽ��ֺ����ۼ��Ļ�����ΪNa2O2����ˮ��Ӧ�Ļ�ѧ����ʽ��2Na2O2+2H2O=4NaOH+O2����
�ʴ�Ϊ��2Na2O2+2H2O=4NaOH+O2����
���� ���⿼��ṹ����λ�ù�ϵ�ۺ�Ӧ�ã��ƶ�Ԫ���ǽ���ؼ����ǶԻ���֪ʶ���ۺϿ��飬�ѶȲ����������ʽ��дΪ�״��㣬�����ܷ���ʽ��Ӱ�죮

 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д�| A�� | X��Y����Ԫ�ؿ������γɶ��ֻ����� | |
| B�� | X��Y��X���⻯����ȶ���ǿ | |
| C�� | �������ܺ��������Ʒ�Ӧ��Ԫ�ؿ�����4�� | |
| D�� | Ԫ��W��ijЩ���������Ϊˮ������ |
| A�� | ��ϩ | B�� | ���� | C�� | ��Ȳ | D�� | ��ȩ |
| A�� | NaCl | B�� | H2O | C�� | KOH | D�� | CH3Cl |
| A�� | Y�ֱ���Z��W�γɵĻ������л�ѧ��������ͬ | |
| B�� | ԭ�Ӱ뾶�Ĵ�С˳��r��W����r��Z����r��Y����r��X�� | |
| C�� | Y����̬���⻯������ȶ��Ա�W��ǿ | |
| D�� | X������������Ӧ��ˮ��������Ա�W���� |
| A�� | ˮ���ɱ����ۻ� | B�� | ʳ�κͱ������ۻ� | ||
| C�� | �Ȼ�狀�ˮ�ķֽ� | D�� | ������ռ���ۻ� |
| A�� | ����̼�������Һ�������ռ���Һ��ϼ��ȣ�NH4++OH-$\frac{\underline{\;\;��\;\;}}{\;}$NH3��+H2O | |
| B�� | ͭƬ��ϡ���ᷴӦ��3Cu+8H++2NO3-�T3Cu2++2NO��+4H2O | |
| C�� | ��ˮ��ϡ���ᷴӦ��OH-+H+�TH2O | |
| D�� | ��Ƭ��ϡ���ᷴӦ��2Fe+6H+�T2Fe3++3H2�� |
| A�� | �����Ҫ�ɷֱ�ʾΪ��C6H12O6��n | |
| B�� | ˿����Ҫ�ɷ��ǵ����� | |
| C�� | ���˿��Ҫ�ɷ־����ڸ߷��ӻ����� | |
| D�� | ���˿����ͨ�����ս������� |