��Ŀ����
��16�֣�Ϊ�˺������û�ѧ�ܣ�ȷ����ȫ���������������Ҫ��ֿ��ǻ�ѧ��Ӧ���ʱ䣬����ȡ��Ӧ��ʩ����ѧ��Ӧ���ʱ�ͨ����ʵ����вⶨ��Ҳ�ɽ����������㡣
��ʵ���ã�5g�״��������г��ȼ�����ɶ�����̼�����Һ̬ˮʱ�ͷų�113.5kJ����������д���״�ȼ�յ��Ȼ�ѧ����ʽ��____________________________________��
����ͼ��ij�ʼDZ������ü״�ȼ�ϵ�صĽṹʾ��ͼ��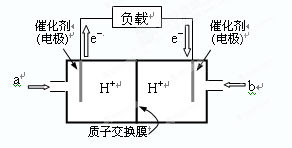
�ŵ�ʱ�״�Ӧ��______��ͨ�루�a����b����������ڲ�H����_____������ҡ����ƶ���д����ظ����ĵ缫��Ӧʽ��_______________________________��
������̬��̬ԭ���γ�1mol��ѧ���ͷŵ���������м��ܡ��ӻ�ѧ���ĽǶȷ�������ѧ��Ӧ�Ĺ��̾��Ƿ�Ӧ��Ļ�ѧ�����ƻ���������Ļ�ѧ�����γɹ��̡��ڻ�ѧ��Ӧ�����У���ѧ����Ҫ�����������γɻ�ѧ���ֻ��ͷ�������
| ��ѧ�� | H��H | N��H | N��N |
| ����/kJ��mol��1 | 436 | a | 945 |
 2NH3(g) ��H����93 kJ��mol��1���Ը��ݱ������м������ݼ���a����ֵ��_______________��
2NH3(g) ��H����93 kJ��mol��1���Ը��ݱ������м������ݼ���a����ֵ��_______________�������ݸ�˹���ɿ��Զ�ijЩ����ͨ��ʵ��ֱ�Ӳⶨ�Ļ�ѧ��Ӧ���ʱ�������㡣
��֪��C(s��ʯī)��O2(g)��CO2(g) ��H1����393.5kJ��mol��1
2H2(g)��O2(g)��2H2O(l) ��H2����571.6kJ��mol��1
2C2H2(g)��5O2(g)��4CO2(g)��2H2O(l) ��H3����2599kJ��mol��1
���ݸ�˹���ɣ�����2C(s��ʯī)��H2(g)��C2H2(g)��Ӧ���ʱ��H��________��
��1��2CH3OH(l)+3O2(g)=2CO2(g)+2H2O(l) ��H=����1452.8kJ��mol-1
��2��a �� CH3OH+H2O-6e-=CO2+6H+ ��3��391 ��4����H="+226.7" kJ��mol-1
����

��ϰ��ϵ�д�
�����Ŀ