��Ŀ����
2�� ������Ƥ�����н�ǿ����ʴ�ԣ��������г��õĽ�������֮һ����п��������Ƥ�ı����㣬���Ĥ�ĺ�ȼ����ȶ�Ҳ�����ж϶Ʋ���������Ҫָ�꣮ij�о���ѧϰС��Ϊ�˲ⶨ��п��Ƥ�ĺ�ȣ�����������ʵ�鷽����
������Ƥ�����н�ǿ����ʴ�ԣ��������г��õĽ�������֮һ����п��������Ƥ�ı����㣬���Ĥ�ĺ�ȼ����ȶ�Ҳ�����ж϶Ʋ���������Ҫָ�꣮ij�о���ѧϰС��Ϊ�˲ⶨ��п��Ƥ�ĺ�ȣ�����������ʵ�鷽��������һ��ȡ�����п��Ƥ��A��B��C������ͬһ���п��Ƥ��������п��ƤA�����ձ��У�����40mL 6mol•L-1���ᣬ��Ӧʱ�ò�����С�ķ�����п��Ƥ����п�Ʋ㷴Ӧ��ȫʱ��������δ��Ӧ����Ƥȡ����ϴ�ӡ�С�ĺ�ɡ���������B��C�ظ������ʵ�飮
��1��п�Ʋ���ȫ��Ӧ�ı�־����������������ͻȻ������
��2����μ�����Ƭ�Ѿ�ϴ��ȡ���һ��ϴ��Һ��������������Һ���ް�ɫ��������������Ƭ��ϴ��
����������֪п��ǿ����Һ��Ӧ�����ӷ���ʽΪ��Zn+2OH-+2H2O=[Zn��OH��4]2-+H2����ijͬѧ����ͼװ������˲ⶨ��п��Ƥ�Ʋ��ȵ�ʵ�鷽�������������ΪS cm2������Ϊmg�Ķ�п��Ƥ����6mol•L-1 NaOH��Һ�У��ش��������⣺
��3��װ���е���a��������ʹ��Һ©������ƿ����ѹ��ͬ������������Һ��˳�����£�
��4����������ԣ���ҩƷ��ˮװ��������У����Ӻ�װ�ú�����еIJ������У�
�ټ�¼C��Һ��λ�ã��ڴ�B�в���������������ָ������£�����A��B�еμ�����NaOH��Һ���������ƶ�C��ʹ����ܺ�C��Һ����ƽ��
����������˳���Ǣܢ٢ۢڢܢ٣�����ţ���ſ��ظ�ʹ�ã���
��5��ʵ��ǰ���ʽ�ζ�����Һ������ֱ�ΪV1 mL��V2 mL����֪Zn���ܶ�Ϊ7.14g/cm3��ʵ������������Ħ�����ΪVm L/mol�������п��Ƥ�ĶƲ���Ϊ$\frac{\frac{V{\;}_{1}-V{\;}_{2}}{1000V{\;}_{m}}��65}{2S��7.14}$cm����д����ѧ����ʽ��
���� ��1����п�Ʋ���ȫ��Ӧ������������ᷴӦ������п�����Ļ����Բ�ͬ����Ӧ���ʾͲ�ͬ���ݴ��жϣ�
��2������ϴ��Һ�������ӵĴ�������������Ƿ�ϴ����
��3��װ���е���a�������DZ���ѹǿ��ͬ����������˳���ӷ�Һ©�����£�
��4����ʵ��Ĺؼ��Dz������������������ǿ�ʼ�Ȱ�����������Һ����ƽ������¼����������������Һ������������ų����ָ������ٰ�����������Һ����ƽ�ٶ�����
��5������п�Ʋ���=$\frac{V}{S}$��п�����=$\frac{m}{��}$��п������=n��Zn����M��Zn������Ӧ��п�����ɵ��������ʵ�����ͬ��
��� �⣺��1����п�Ʋ���ȫ��Ӧ������������ᷴӦ������п�����Ļ����Բ�ͬ����Ӧ���ʾͲ�ͬ���ⶨ������6mol/L������Һ��Ӧǰ�����������仯����Ӧ��ȫ�Dz�������������
�ʴ�Ϊ����������������ͻȻ������
��2������Һ��ȡ������Ƭ��Ȼ�������ἰ���Σ���ϴ��ʱҲ���Dz��ٺ������ἰ���Σ����Լ�����Ƭ�Ƿ�ϴ�����Ǽ��������ӣ�Ҳ�ɼ��������ԣ�������Ƭ��ϴ���ķ����Ǽ���ϴ��Һ���Ƿ��������ӣ�ȡ���һ��ϴ��Һ��������������Һ���ް�ɫ��������������Ƭ��ϴ����
�ʴ�Ϊ��ȡ���һ��ϴ��Һ��������������Һ���ް�ɫ��������������Ƭ��ϴ����
��3��װ���е���a��������ʹ��Һ©������ƿ����ѹ��ͬ������������Һ��˳�����£�
�ʴ�Ϊ��ʹ��Һ©������ƿ����ѹ��ͬ������������Һ��˳�����£�
��4�����ݴ�װ�õ�ԭ����Ӧ���μ�¼C�еĶ�����ȡ���ֵ���������������������Ե�һ����Ӧ���Ǣܣ�Ȼ���Ǣټ�¼C��Һ��λ�ã��پ��Ǣ���A��B�еμ�����NaOH��Һ���ڴ�B�в���������������ָ������£��������ܢ������ƶ�C��ʹ����ܺ�C��Һ����ƽ������Ǣټ�¼C�Ķ��������㣻
�ʴ�Ϊ���ܢ٢ۢڢܢ٣�
��5��V��H2��=��V2-V1�� mL��n��Zn��=n��H2��=$\frac{V{\;}_{2}-V{\;}_{1}}{1000•V{\;}_{m}}$��m��Zn��=65 g•mol-1 n��Zn��=$\frac{V{\;}_{2}-V{\;}_{1}}{1000•V{\;}_{m}}$��65g��п���ܶ���7.14 g•cm-3������V��Zn��=$\frac{m}{��}$=$\frac{m��Zn��}{7.14}$��������Ƭ�������ϡ������㣬����ÿһ��ĺ��h=$\frac{V��Zn��}{2S}$=$\frac{\frac{V{\;}_{1}-V{\;}_{2}}{1000V{\;}_{m}}��65}{2S��7.14}$��
�ʴ�Ϊ��$\frac{\frac{V{\;}_{1}-V{\;}_{2}}{1000V{\;}_{m}}��65}{2S��7.14}$��
���� ���⿼�����������ʵ�ʵ����ƺͲ�������жϣ�װ�÷����ǽ���ؼ���ע�������ȵļ��㷽��Ӧ�ã���Ŀ�Ѷ��еȣ�

| A�� | ����Ħ���������ֵ�ǹ̶�����ģ���ԼΪ22.4L | |
| B�� | ͨ�����������Ħ�����ԼΪ22.4L | |
| C�� | ��״���µ������Ħ�����ԼΪ22.4L | |
| D�� | ��״���£������Ħ�����ԼΪ22.4L/mol |
| A�� | ��ɫʳƷ����ָ��ɫΪ��ɫ��ʳƷ | B�� | �л�ʳƷ��ָ���л����ʳƷ | ||
| C�� | OTC��ʾ����ҩ | D�� | �ӵ�������ʳ���м���KIO3 |
| ���� | Mg��OH��2 | CaCO3 | MgCO3 | Ca��OH��2 | CaSO4 | MgSO4 |
| $\frac{�ܽ��}{g/100g{H}_{2}O}$ | 9.0��10-4 | 1.5��10-3 | 1.0��10-2 | 1.7��10-1 | 2.1��10-1 | 26.7 |
��2����¯�е�ˮ�������е�CaSO4����ֱ�������ȥ����Ҫ�����Լ�X��ʹ��ת��Ϊ���������������Y����ȥ���Լ�X��C������ţ���
A��ʳ�ס�������B���Ȼ�������C��̼���ơ���D̼������
CaSO4��ת��Ϊ����Y ��ԭ���ǣ�������������̼��Ƶ��ܽ��С������Ƶ��ܽ�ȣ�
��3������CaSO4��Mg��OH��2�⣬��¯ˮ���л����ܺ��е������ǣ�д��ѧʽ��MgCO3��CaCO3��
��4��ˮ�е�Ca2+��Mg2+������һ��Ũ�ȵ�M��Һ���вⶨ��M��Ca2+��Mg2+���Ե����ʵ������з�Ӧ����ȡ��Ca2+Ũ��Ϊ0.40g/L������ˮ��10mL������ƿ�н��еζ����ﵽ�յ�ʱ����ȥ��Ũ��Ϊ0.01mol/L��M��Һ20.00mL���������ˮ����Mg2+�����ʵ���Ũ����0.01mol/L��
 ��1����֪���ڳ����£�Ũ�Ⱦ�Ϊ0.1mol•L-1������������Һ��pH��
��1����֪���ڳ����£�Ũ�Ⱦ�Ϊ0.1mol•L-1������������Һ��pH��| ���� | CH3CO ONa | NaHCO3 | Na2CO3 | NaClO | NaCN | C6H5ONa |
| pH | 8.8 | 9.7 | 11.6 | 10.3 | 11.1 | 11.3 |
A��CH3COOH+Na2CO3=NaHCO3+CH3COONa B��CH3COOH+NaCN=CH3COONa+HCN
C��CO2+H2O+2NaClO=Na2CO3+2HClO D��CO2+H2O+2C6H5ONa=Na2CO3+2C6H5OH
��2������ǰ����Ϣ�жϣ�Ũ�Ⱦ�Ϊ0.05mol•L-1�������������ʵ���Һ�У�pH��С���Ǣ� �����ţ���������Һ�ֱ�ϡ��100����pH�仯��С���Ǣ� �����ţ���
��C6H5OH ��CH3COOH ��HCN ��HClO ��H2SO4 ��HClO4
��3������ǰ����Ϣ��д��������������ͨ�뵽������̼������Һ�з�Ӧ�����ӷ���ʽ��Cl2+H2O+2CO32-=2HCO3-+Cl-+ClO-��
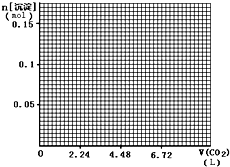
��4����״���£���1L��NaOH��Ca��OH��2��0.1mol����Һ�в���ͨ��CO2���������뻭�������ij��������ʵ�����mol����CO2ͨ�������L���ı仯����ͼ��
| A�� | 3s | B�� | 3p | C�� | 3d | D�� | 2d |
 ��ѧ����Դ����������������ʮ����Ҫ�����ã�
��ѧ����Դ����������������ʮ����Ҫ�����ã�
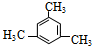
 2-��-2��4-����ϩ
2-��-2��4-����ϩ
 ��
��