��Ŀ����
��1��ʵ�����ﳣ��Ũ������������̷�Ӧ����ȡ��������������Ӧ�Ļ�ѧ����ʽΪ��MnO2+4HCl��Ũ��
MnCl2+Cl2��+2H2O��ȡһ������Ũ����ʹ����������̷�����Ӧ�������������ڱ�״���µ����Ϊ22.4L����ش��������⣺
��д���÷�Ӧ�����ӷ�Ӧ����ʽ ��
�ڷ�Ӧ�б�������HCl�����ʵ���Ϊ mol��
��ʵ���ұ��õ�Ũ������������Ϊ36.5%���ܶ�Ϊ1.19g?cm-3��Ϊʹ�÷��㣬��������Ũ��������ʵ���Ũ�� mol��L-1��
��2����һС��þ���Ͻ�����100mL�����У�Ȼ�������е���1mol��L-1NaOH��Һ�����ɳ����������ͼ���NaOH��Һ���������ͼ��ʾ��
�ٺϽ���þ������ g��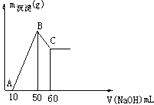
����������ʵ���Ũ�� mol��L-1��
| ||
��д���÷�Ӧ�����ӷ�Ӧ����ʽ
�ڷ�Ӧ�б�������HCl�����ʵ���Ϊ
��ʵ���ұ��õ�Ũ������������Ϊ36.5%���ܶ�Ϊ1.19g?cm-3��Ϊʹ�÷��㣬��������Ũ��������ʵ���Ũ��
��2����һС��þ���Ͻ�����100mL�����У�Ȼ�������е���1mol��L-1NaOH��Һ�����ɳ����������ͼ���NaOH��Һ���������ͼ��ʾ��
�ٺϽ���þ������

����������ʵ���Ũ��
���㣺��ѧ����ʽ���йؼ���,þ��������Ҫ������
ר�⣺
��������1���ٶ������̡����塢ˮ�����ӷ���ʽ��Ӧ������ѧʽ��
�ڸ���ClԪ�صĻ��ϼ۱仯��ԭ���غ��֪����������HCl�����ʵ����������������ʵ�����2����
�۸���c=
������Ũ��������ʵ���Ũ�ȣ�
��2������ͼ��֪���Ӽ���10m����������Һ��ʼ������������������������ҺΪ50mLʱ�����������ʱΪMg��OH��2��Al��OH��3���ý�������������40mL�����������غ��֪3n[Al��OH��3]+2n[Mg��OH��2]=n��NaOH��=��0.05L-0.01L����1mol/L=0.04mol���Ӽ�������������Һ50mL��60mL�ܽ������������ýη�����ӦAl��OH��3+NaOH=NaAlO2+2H2O������n[Al��OH��3]=��0.06L-0.05L����1mol/L=0.01mol��������ʽ����n[Mg��OH��2]����Ԫ���غ��֪n��Mg��=n[Mg��OH��2]��������m=nM����Mg��Al��������
�ڼ�������������ҺΪ60mLʱ�����������ʱΪMg��OH��2��Al��OH��3����ҺΪ�Ȼ�����Һ��������Ԫ���غ��ʱ��Һ��n��NaCl��=n��NaOH�����ݴ˼����n��HCl����������c=
������������ʵ���Ũ�ȣ�
�ڸ���ClԪ�صĻ��ϼ۱仯��ԭ���غ��֪����������HCl�����ʵ����������������ʵ�����2����
�۸���c=
| 1000��w |
| M |
��2������ͼ��֪���Ӽ���10m����������Һ��ʼ������������������������ҺΪ50mLʱ�����������ʱΪMg��OH��2��Al��OH��3���ý�������������40mL�����������غ��֪3n[Al��OH��3]+2n[Mg��OH��2]=n��NaOH��=��0.05L-0.01L����1mol/L=0.04mol���Ӽ�������������Һ50mL��60mL�ܽ������������ýη�����ӦAl��OH��3+NaOH=NaAlO2+2H2O������n[Al��OH��3]=��0.06L-0.05L����1mol/L=0.01mol��������ʽ����n[Mg��OH��2]����Ԫ���غ��֪n��Mg��=n[Mg��OH��2]��������m=nM����Mg��Al��������
�ڼ�������������ҺΪ60mLʱ�����������ʱΪMg��OH��2��Al��OH��3����ҺΪ�Ȼ�����Һ��������Ԫ���غ��ʱ��Һ��n��NaCl��=n��NaOH�����ݴ˼����n��HCl����������c=
| n |
| V |
���
�⣺��1���ٷ�Ӧ�Ļ�ѧ����ʽΪMnO2+4HCl��Ũ��
MnCl2+C12��+2H2O����дΪ���ӷ�Ӧʱ�������̡�������ˮ�����ӷ���ʽ��Ӧ������ѧʽ��
�����ӷ���ʽΪMnO2+4H++2Cl-
Cl2��+Mn2++2H2O���ʴ�Ϊ��MnO2+4H++2Cl-
Cl2��+Mn2++2H2O��
�������ڱ�״���µ����Ϊ22.4L��n��Cl2��=
=1mol����MnO2+4H++2Cl-
Cl2��+Mn2++2H2O��֪��4molHCl��Ӧʱֻ��2mol����ԭ����������
��������HCl�����ʵ���Ϊ1mol��2=2mol���ʴ�Ϊ��2mol��
����c=
��֪��c=
=11.9 mol?L-1���ʴ�Ϊ��11.9��
��2������ͼ��֪���Ӽ���10m����������Һ��ʼ������������������������ҺΪ50mLʱ�����������ʱΪMg��OH��2��Al��OH��3���ý�������������40mL�����������غ��֪3n[Al��OH��3]+2n[Mg��OH��2]=n��NaOH��=��0.05L-0.01L����1mol/L=0.04mol���Ӽ�������������Һ50mL��60mL�ܽ������������ýη�����ӦAl��OH��3+NaOH=NaAlO2+2H2O������n[Al��OH��3]=��0.06L-0.05L����1mol/L=0.01mol����3��0.01mol+2n[Mg��OH��2]=0.04mol�����n[Mg��OH��2]=0.005mol����Ԫ���غ��֪n��Mg��=n[Mg��OH��2]=0.005mol����m��Mg��=0.005mol��24g?mol-1=0.12g��m��Al��=0.01mol��27g?mol-1=0.27g��
�ʴ�Ϊ��0.12��
�ڼ�������������ҺΪ50mLʱ�����������ʱΪMg��OH��2��Al��OH��3����ҺΪ�Ȼ�����Һ��������Ԫ���غ��ʱ��Һ��n��NaCl��=n��NaOH��=0.05L��1mol/L=0.05mol��������Ԫ���غ�n��HCl��=0.05mol������������ʵ���Ũ��Ϊ
=0.5mol/L���ʴ�Ϊ��0.5��
| ||
�����ӷ���ʽΪMnO2+4H++2Cl-
| ||
| ||
�������ڱ�״���µ����Ϊ22.4L��n��Cl2��=
| 22.4L |
| 22.4L/mol |
| ||
��������HCl�����ʵ���Ϊ1mol��2=2mol���ʴ�Ϊ��2mol��
����c=
| 1000��w |
| M |
| 1000��1.19��36.5% |
| 36.5 |
��2������ͼ��֪���Ӽ���10m����������Һ��ʼ������������������������ҺΪ50mLʱ�����������ʱΪMg��OH��2��Al��OH��3���ý�������������40mL�����������غ��֪3n[Al��OH��3]+2n[Mg��OH��2]=n��NaOH��=��0.05L-0.01L����1mol/L=0.04mol���Ӽ�������������Һ50mL��60mL�ܽ������������ýη�����ӦAl��OH��3+NaOH=NaAlO2+2H2O������n[Al��OH��3]=��0.06L-0.05L����1mol/L=0.01mol����3��0.01mol+2n[Mg��OH��2]=0.04mol�����n[Mg��OH��2]=0.005mol����Ԫ���غ��֪n��Mg��=n[Mg��OH��2]=0.005mol����m��Mg��=0.005mol��24g?mol-1=0.12g��m��Al��=0.01mol��27g?mol-1=0.27g��
�ʴ�Ϊ��0.12��
�ڼ�������������ҺΪ50mLʱ�����������ʱΪMg��OH��2��Al��OH��3����ҺΪ�Ȼ�����Һ��������Ԫ���غ��ʱ��Һ��n��NaCl��=n��NaOH��=0.05L��1mol/L=0.05mol��������Ԫ���غ�n��HCl��=0.05mol������������ʵ���Ũ��Ϊ
| 0.05mol |
| 0.1L |
���������⿼�黯ѧ��Ӧ���йؼ��㣬Ϊ��Ƶ���㣬���շ����ķ�Ӧ����Ӧ�����ʵ�����ϵ��ͼ��Ӧ�Ķ�Ӧ��ϵ���ǽ��Ĺؼ������ط�������������Ŀ��飬��Ŀ�Ѷ��еȣ�

��ϰ��ϵ�д�
�����Ŀ
��ˮ��һ����Ļ�ѧ��Դ�⣬�����йغ�ˮ�ۺ����õ�˵��������ǣ�������
| A����ˮ�к�����Ԫ�أ��辭����ѧ�仯�ſ��Եõ��嵥�� | ||||||||
| B�������Ǻ�ˮ���˷��绹�dz�ϫ���磬���ǽ���ѧ��ת��Ϊ���� | ||||||||
| C���Ӻ�ˮ�п��Եõ�NaCl���������NaCl���Ʊ������� | ||||||||
D���Ӻ�ˮ����ȡþ���Բ������з�������ˮ��ˮ
|
���¡���ѹ�£�1mol A��n mol B��һ���ݻ��ɱ�������з������·�Ӧ��A��g��+2B��g��?2C��g����һ��ʱ���ﵽƽ�⣬����a mol C��������˵����ȷ���ǣ�������
| A������A��ת����Ϊa | ||
B����ʼʱ�̺ʹﵽƽ��ʱ�����е�ѹǿ��Ϊ��1+n������1+n-
| ||
| C����Ӧ��ƽ������������г���amolHe������B��ת���ʼ�С | ||
| D����v����A��=2v�棨C��ʱ�����жϷ�Ӧ�ﵽƽ�� |
����pH=3��CH3COOH��Һ��pH=11��NaOH��Һ��V mL�������к͵ζ������ζ���pH=7ʱ����ƿ����Һ�����Ϊ���ζ�ǰ����Һδϡ�͡���������䣩��������
| A������2V mL |
| B����2V mL |
| C������2V mL |
| D����ȷ�� |
��һԪ���һԪ��������Һ���� pH�ֱ�Ϊ2��12������Һ�������Ϻ��pHΪ��������
| A������7 |
| B����7 |
| C������7 |
| D�����ڡ�С�ڻ����7���п��� |
�����й�ʵ��ԭ����ʵ�������ȷ���ǣ�������
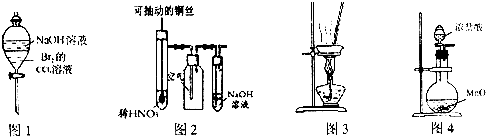

| A��ͼ1װ�ÿ������ڳ�ȥ�ܽ���CCl4�е�Br2 |
| B��ͼ2��ʾװ����ϡHNO3���ڹ��ƿ�п��ռ�NO���� |
| C����ͼ3װ����������������CuSO4?5H2O�����Գ�ȥ�ᾧˮ |
| D��ʵ���ҳ���ͼ4װ����ȡ�������� |
�ͳ���ʯ��ˮ�м�������CaC2����ַ�Ӧ��ָ���ԭ���¶ȣ�������Һ�У�������
| A��c��Ca2+����c��OH-�������� |
| B��c��Ca2+����c��OH-�������ֲ��� |
| C��n��Ca2+����n��OH-�������� |
| D��c��OH-������c��H+����С |
���л�����ĺ˴Ź��������г�����������ǣ�������
| A��2��5�������� |
| B��3��4�������� |
| C��2��3��4�������� |
| D��2��2��3��3�ļ����� |