��Ŀ����
9�������˵�վй©�ķ����������к���$\stackrel{131}{53}$I�������й�$\stackrel{131}{53}$I��˵����ȷ���ǣ�������| A�� | �ɴ˿�ȷ����Ԫ�ص����ԭ������Ϊ131 | |
| B�� | $\stackrel{131}{53}$I�ǵ�Ԫ�ص�һ��ͬλ�� | |
| C�� | $\stackrel{131}{53}$I�����к�������Ϊ53 | |
| D�� | $\stackrel{131}{53}$I2��Ħ������Ϊ262 g |
���� 53131I��������λ53��������Ϊ131��������+������=��������������ͬ����������������ͬ��ͬ��Ԫ�ص�ԭ�ӻ�Ϊͬλ�أ���ע��Ħ�������ĵ�λΪg/mol��
��� �⣺A��ԭ�ӵ����ԭ������Ϊ131����Ԫ�ص����ԭ���������ͬλ��ԭ�ӵĺ����йأ���A����
B��������ָ����һ����Ŀ���Ӻ�һ����Ŀ���ӵ�һ��ԭ�ӣ�53131I�ǵ�Ԫ�ص�һ�ֺ��أ���B��ȷ��
C��53131IԪ���к�������Ϊ131-53=78����C����
D��53131I2��Ħ������Ϊ262g/mol����D����
��ѡB��
���� ���⿼��ԭ�ӵĹ��ɼ�ԭ���е�������ϵ��ѡ��A��ѧ������е��ѵ���״��㣬�ѶȲ���

��ϰ��ϵ�д�
 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д�
�����Ŀ
7��ijͬѧ����ͼ��ʾ��װ�ý��е��ʵ�飮����˵����ȷ���ǣ�������
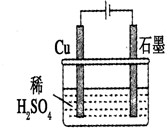

| A�� | ʯī�ǵ��صĸ�����Cu�ǵ��ص����� | |
| B�� | �������У�ͭ�缫������ûʲô�仯 | |
| C�� | ���һ��ʱ���ʯī�缫�ϻ���ͭ���� | |
| D�� | �����������У���Һ��pH����仯 |
17����ҵ����������ˮ��̼���ȣ�SrCO3����ĩΪԭ�ϣ��������������Ļ�����Ʊ��ߴ���ˮ�Ȼ��Ⱦ��壨SrCl2•6H2O���������Ϊ��
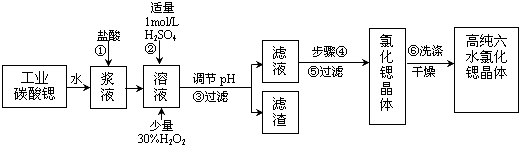
��֪�����й��������������pH��
��SrCl2•6H2O ������61��ʱ��ʼʧȥ�ᾧˮ��100��ʱʧȥȫ���ᾧˮ��
��1����������̼���������ᷴӦ�����ӷ���ʽSrCO3+2H+=Sr2++CO2��+H2O��
��2���ڲ����-�۵Ĺ����У�����Һ��pHֵ��1������B�����õ��Լ�ΪE��
A��1.5 B��3.7 C��9.7
D��ˮ E�������ȷ�ĩ F̼���ƾ���
��3���������м���H2O2������Ӧ�����ӷ���ʽ��2Fe2++H2O2+2H+=2Fe3++2H2O��
��4����������������������Ҫ�ɷ���Fe��OH��3��BaSO4���ѧʽ����
��5����ҵ�����ȷ紵����ˮ�Ȼ��ȣ����˵��¶���A��
A��50��60��B��80��100��C��100������
��6���������ѡ�õ���ϴ�Ӽ��DZ����Ȼ�����Һ��

��֪�����й��������������pH��
| �������� | Fe��OH��3 | Fe��OH��2 |
| ��ʼ������pH | 1.5 | 6.5 |
| ������ȫ��pH | 3.7 | 9.7 |
��1����������̼���������ᷴӦ�����ӷ���ʽSrCO3+2H+=Sr2++CO2��+H2O��
��2���ڲ����-�۵Ĺ����У�����Һ��pHֵ��1������B�����õ��Լ�ΪE��
A��1.5 B��3.7 C��9.7
D��ˮ E�������ȷ�ĩ F̼���ƾ���
��3���������м���H2O2������Ӧ�����ӷ���ʽ��2Fe2++H2O2+2H+=2Fe3++2H2O��
��4����������������������Ҫ�ɷ���Fe��OH��3��BaSO4���ѧʽ����
��5����ҵ�����ȷ紵����ˮ�Ȼ��ȣ����˵��¶���A��
A��50��60��B��80��100��C��100������
��6���������ѡ�õ���ϴ�Ӽ��DZ����Ȼ�����Һ��
14����������ɸ�������ɺ����ʽ��з��࣬���磺

��1����ͼ��ʾ�����ʷ������������״���෨��
��2����K��Na��H��O��S��N�������ֻ�����Ԫ����ɺ��ʵ����ʣ��ֱ������±��Тڡ����森
��3���ڢ٢ۢߢ���������ʵ��Ǣ٢ۣ�����ţ���
��4��д������۷����кͷ�Ӧ���ӷ���ʽH++OH-=H2O��
��5��д��ʵ�����Ʊ���Ļ�ѧ����ʽ2NH4Cl+Ca��OH��2$\frac{\underline{\;\;��\;\;}}{\;}$2NH3��+2H2O+CaCl2��
��6�������Ħ��������63g/mol��3.4g������0.2mol����������0.2NA��������ԭ��0.6mol���ڱ�״���£�4.48L CO2����6.4g O2�����ʵ�����ȣ�

��1����ͼ��ʾ�����ʷ������������״���෨��
��2����K��Na��H��O��S��N�������ֻ�����Ԫ����ɺ��ʵ����ʣ��ֱ������±��Тڡ����森
| ������� | �� | �� | �� | ������ | �⻯�� |
| ��ѧʽ | ��H2SO4 ��HNO3 | ��NaOH ��KOH | ��Na2SO4 ��K2SO4��KNO3 | ��SO2 ��SO3 | ��NH3 |
��4��д������۷����кͷ�Ӧ���ӷ���ʽH++OH-=H2O��
��5��д��ʵ�����Ʊ���Ļ�ѧ����ʽ2NH4Cl+Ca��OH��2$\frac{\underline{\;\;��\;\;}}{\;}$2NH3��+2H2O+CaCl2��
��6�������Ħ��������63g/mol��3.4g������0.2mol����������0.2NA��������ԭ��0.6mol���ڱ�״���£�4.48L CO2����6.4g O2�����ʵ�����ȣ�
19������������������Ȼ�߷��ӻ�������ǣ�������
| A�� | ��ά�� | B�� | ���� | C�� | ��֬ | D�� | ����ϩ |
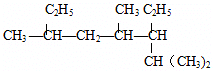 ���ƣ�2��4��6-����-3-�һ�����
���ƣ�2��4��6-����-3-�һ����� 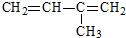 ���ƣ�2-��-1��3-����ϩ
���ƣ�2-��-1��3-����ϩ
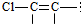 �ṹ����4��
�ṹ����4�� ��
�� ��KMnO4��H+����Һ��
��KMnO4��H+����Һ�� ��
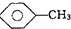
��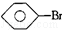 ��C6H12����ϩ������ˮ��
��C6H12����ϩ������ˮ��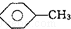 ��CCl4���Ҵ���ˮ��
��CCl4���Ҵ���ˮ��
 ���ڹ�ҵ��չ��ȼ���豸�������࣬�豸��ģ����������Щ�����ŷŵ������ж����д�����SO2��������ͳ�ƣ��ҹ�1995�깤ҵSO2���ŷ���Ϊ1396��֣�2006�깤ҵSO2���ŷ����ﵽ��3800��֣�����SO2����Ⱦ���ҹ�ÿ����ʧ�ߴ�1100��Ԫ��
���ڹ�ҵ��չ��ȼ���豸�������࣬�豸��ģ����������Щ�����ŷŵ������ж����д�����SO2��������ͳ�ƣ��ҹ�1995�깤ҵSO2���ŷ���Ϊ1396��֣�2006�깤ҵSO2���ŷ����ﵽ��3800��֣�����SO2����Ⱦ���ҹ�ÿ����ʧ�ߴ�1100��Ԫ��