��Ŀ����
6��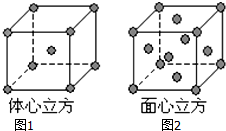 ��֪A��B��C��D��E����Ԫ�����ڱ���ǰ�����ڵ�Ԫ�أ����ǵĺ˵����A��B��C��D��E��Bԭ�ӵ�p�����������γɵ��⻯��ķе���ͬ����Ԫ�ص��⻯������͵ģ�Dԭ�ӵõ�һ�����Ӻ�3p���ȫ������A+��Dԭ���γɵ�������һ�����Ӳ㣮C��A�γ�A2C�����ӻ����E��ԭ������Ϊ26��Eԭ�ӻ�������Χ�н϶���������Ŀչ��������һЩ���ӻ������γ��������������������ش��������⣺������ʱ��A��B��C��D��E������Ӧ��Ԫ�ط��ű�ʾ��
��֪A��B��C��D��E����Ԫ�����ڱ���ǰ�����ڵ�Ԫ�أ����ǵĺ˵����A��B��C��D��E��Bԭ�ӵ�p�����������γɵ��⻯��ķе���ͬ����Ԫ�ص��⻯������͵ģ�Dԭ�ӵõ�һ�����Ӻ�3p���ȫ������A+��Dԭ���γɵ�������һ�����Ӳ㣮C��A�γ�A2C�����ӻ����E��ԭ������Ϊ26��Eԭ�ӻ�������Χ�н϶���������Ŀչ��������һЩ���ӻ������γ��������������������ش��������⣺������ʱ��A��B��C��D��E������Ӧ��Ԫ�ط��ű�ʾ����1��A��B��C��D�ĵ�һ��������С�����˳��ΪNa��S��P��Cl��
��2��C���⻯������Ǽ��ԣ�����ԡ��Ǽ��ԡ������ӣ�
��3��������BD3�ķ��ӿռ乹���������Σ�����ԭ�ӵ��ӻ���ʽΪSP3
��4������E���ʵľ����ڲ�ͬ�¶��������ֶѻ���ʽ��������ͼ��1��2����ʾ������������������������������ʵ�ʺ��е�Eԭ�Ӹ���֮��Ϊ1��2��
���� Bԭ�ӵ�p�����������γɵ��⻯��ķе���ͬ����Ԫ�ص��⻯������͵ģ������Ų�ʽӦΪ1S22S22P63S23P3��ΪPԪ�أ��������ǵ�Ԫ�أ�����е㲻����ͣ�Dԭ�ӵõ�һ�����Ӻ�3p���ȫ������ԭ�ӵĵ����Ų�ʽӦΪ1S22S22P63S23P5��ΪClԪ�أ�A+��Dԭ���γɵ�������һ�����Ӳ㣬��AӦΪNaԪ�أ�C��A�γ�A2C�����ӻ������CӦΪ�ڢ�A����Ԫ�أ����ݺ˵����A��B��C��D��E��֪ӦΪSԪ�أ�E��ԭ������Ϊ26��ΪFeԪ�أ��Դ˽����⣮
��� �⣺�����Ϸ�����֪AΪNa��BΪP��CΪS��DΪCl��EΪFe��
��1����Ԫ�����ڱ��У�ͬһ����Ԫ�صĵ�һ�����ܴ�����������ͬһ����Ԫ�صĵ�һ�����ܴ��ϵ�����С���ݴ˿��ж�����Ԫ�صĵ�һ�����ܵ�˳��Ϊ��Na��S��P��Cl���ʴ�Ϊ��Na��S��P��Cl��
��2��C���⻯��ΪH2S���������幹�ͺ�ˮ�������ƣ�ΪV�Σ�����������������IJ��ص���Ϊ���Է��ӣ��ʴ�Ϊ�����ԣ�
��3��PCl3�к���3���ļ����µ��Ӷ���Ϊ$\frac{5-1��3}{2}$=1�����Է��ӵĿռ乹���������Σ�Ϊ SP3�ӻ����ʴ�Ϊ�������Σ� SP3��
��4�����ݾ������������ķ��䷽�����㣬������������ʵ�ʺ��е�Eԭ�Ӹ���Ϊ$\frac{1}{8}$��8+1=2����������������ʵ�ʺ��е�Eԭ�Ӹ���Ϊ$\frac{1}{8}$��8+$\frac{1}{2}$��6=4���ʶ��߱�ֵΪ1��2���ʴ�Ϊ��1��2��
���� ���⿼��Ԫ���ƶ��⣬Ϊ��Ƶ����ͳ������ͣ�������ѧ���ķ��������������Ŀ��飬�ƶϳ�Ԫ�ص������ǽ����Ĺؼ����ƶ�ʱע���ԭ�ӵĺ�������Ų��ص��Լ�Ԫ�ص���������Ϊͻ�ƿڽ�𣬱������һ���Ѷȣ�

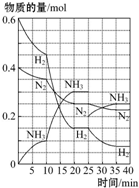 500�桢20MPaʱ����H2��N2����һ�ݻ�Ϊ2L���ܱ������з�����Ӧ��3H2+N2?2NH3��H��0����Ӧ������H2��N2��NH3���ʵ����仯��ͼ��ʾ����ͼ�ж�������������ȷ���ǣ�������
500�桢20MPaʱ����H2��N2����һ�ݻ�Ϊ2L���ܱ������з�����Ӧ��3H2+N2?2NH3��H��0����Ӧ������H2��N2��NH3���ʵ����仯��ͼ��ʾ����ͼ�ж�������������ȷ���ǣ�������| A�� | ��Ӧ��ʼ����һ��ƽ��ʱ��N2��ƽ����Ӧ����Ϊ0.062 5 mol•L-1•min-1��H2��ת����Ϊ75% | |
| B�� | ��Ӧ35 minʱ���ﵽ�ڶ���ƽ��ʱ����Ӧ��ƽ�ⳣ����K=$\frac{0.1{5}^{2}}{0.07{5}^{3}•0.125}$ | |
| C�� | �����߱仯���Կ�������Ӧ���е�10��20 minʱ���ܲ�ȡ�Ĵ�ʩ��ʹ���˴��� | |
| D�� | �����߱仯���Կ�������Ӧ������25 minʱ����ȡ�Ĵ�ʩ�Ƿ����0.1 mol�İ�����ƽ�����������ƶ� |
| A�� | ��ʱ�����ɵİ�Һ������ | B�� | ���������ڰ������� | ||
| C�� | ʹ�ô����ٽ�����ת�� | D�� | ����ѹǿ�������ڰ������� |
 ��ϩ��C2H4�ṹʽ��
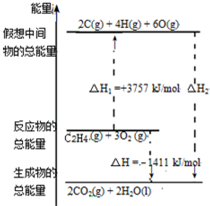
��ϩ��C2H4�ṹʽ��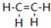 ���ȿ�������ʵ�Ĵ������Ҳ������ȼ�ϣ�ȼ��ʱ�йػ�ѧ�����仯��ͼ��ʾ������1mol��ѧ�������������kJ����C=CΪ615��O=OΪ500��C-HΪ414��C=OΪ750�������1mol O-H�����������ǣ�����������֪��C2H4+3O2=2CO2+2H2O��
���ȿ�������ʵ�Ĵ������Ҳ������ȼ�ϣ�ȼ��ʱ�йػ�ѧ�����仯��ͼ��ʾ������1mol��ѧ�������������kJ����C=CΪ615��O=OΪ500��C-HΪ414��C=OΪ750�������1mol O-H�����������ǣ�����������֪��C2H4+3O2=2CO2+2H2O��| A�� | 542 | B�� | 391 | C�� | 1834 | D�� | 917 |
| �� ���� | ��A | 0 | ||||||
| 1 | ��A | ��A | ��A | ��A | ��A | ��A | ||
| 2 | �� | |||||||
| 3 | �� | �� | �� | �� | ||||
��2���ܺ͢�����Ԫ�ص�����������Ӧˮ�������Խ�������H2SO4���ѧʽ�����ں͢�����Ԫ���У�ԭ�Ӱ뾶�ϴ����Al����Ԫ�ط��ţ���
��3��Ԫ�آٵ�����������Ԫ�آڵĵ��ʷ�Ӧ�����ӷ�Ӧ����ʽΪ��2Al+2OH-+2H2O=2AlO2-+3H2����
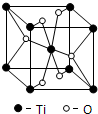
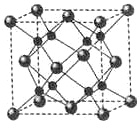 A��B��C��DΪԭ�������������������Ԫ�أ�A2-��B+������ͬ�ĵ��ӹ��ͣ�C��DΪͬ����Ԫ����C�������������������������3����DԪ���������һ��δ�ɶԵ��ӣ��ش��������⣺
A��B��C��DΪԭ�������������������Ԫ�أ�A2-��B+������ͬ�ĵ��ӹ��ͣ�C��DΪͬ����Ԫ����C�������������������������3����DԪ���������һ��δ�ɶԵ��ӣ��ش��������⣺