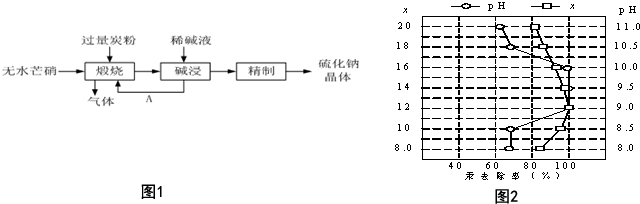
��Ŀ����
9����������Ҫ�ɷ�ΪFeS2�����ҹ���������᳧��ȡ�������Ҫԭ�ϣ�ij��ѧѧϰС���ij������ʯ��������ʵ��̽����[ʵ��һ]�ⶨ��Ԫ�صĺ���
��m1 g�û�������Ʒ������ͼ��ʾװ�ã��гֺͼ���װ��ʡ�ԣ���ʯӢ���У���a�����ϵػ���ͨ���������������ʯӢ���еĻ�������Ʒ����Ӧ��ȫ��ʯӢ���з�����Ӧ�Ļ�ѧ����ʽΪ��4FeS2+11O2$\frac{\underline{\;����\;}}{\;}$2Fe2O3+8SO2
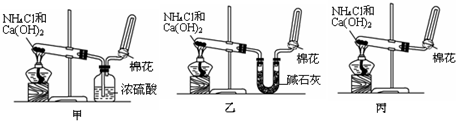
��Ӧ��������ƿ�е���Һ�������´�����
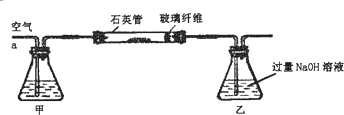
[ʵ���]�ⶨ��Ԫ�صĺ�����
�ⶨ��Ԫ�غ�����ʵ�鲽����ͼ��ʾ��
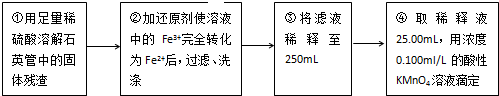
��������ϡ�����ܽ�ʯӢ���еĹ�������ڼӻ�ԭ��ʹ��Һ�е�Fe3+��ȫת��ΪFe2+���ˡ�ϴ�Ӣ۽���Һϡ����250mL��ȡϡ��Һ25.00mL����Ũ�� 0.100ml/L������KMnO4��Һ�ζ�
�������ۣ�
��1�����У���ƿ����ʢ�Լ��� ��Һ����ƿ�ڷ�����Ӧ�����ӷ���ʽΪSO2+2OH-=SO32-+H2O��2SO32-+O2=2SO42-��
��2�����У���ƿ����H2O2��Һʱ��Ӧ�����ӷ���ʽΪSO32-+H2O2�TSO42-+H2O��
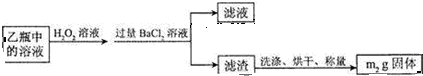
��3���û���������Ԫ�ص���������Ϊ$\frac{32m{\;}_{2}}{233m{\;}_{1}}$��100%��
��4����IJ�����У���Ҫ�õ����������ձ�������������ͷ�ι��⣬����250mL����ƿ��
��5����IJ�����У���ʾ�ζ��Ѵ��յ�����������һ�θ��������Һ����ʱ����Һ��ɫͻ��Ϊ��ɫ������30s�ڲ���ɫ
��6����IJ���ܽ���������ƽ��ʵ�飬�������KMnO4��Һ����ֱ�Ϊ24.98mL��24.80mL��25.02mL��KMnO4����ԭΪMn2+���������������ݣ��ɼ�����û�������Ʒ��Ԫ�ص���������Ϊ$\frac{7}{m{\;}_{1}}$��
���� ��1��Ϊ��ֹ�������Һ�л���BaCO3������Ӱ��ʵ������Ӧ�������еĶ�����̼��ȥ�����Լ�װ���п���NaOH��Һ������NaOH��Һ��SO2��Ӧ����SO32-��H2O��������������ܱ���������������������ӣ�
��2�������������ǿ�����ԣ������������������Ϊ�������ͬʱ����ˮ��
��3�����յõ��ij���Ϊ���ᱵ��������ԭ���غ����������������������Ӷ�����������������
��4������250mlһ�����ʵ���Ũ�ȵ���Һ��Ҫ�ձ�������������ͷ�ιܡ���Ͳ��250ml����ƿ��������
��5�����һ�θ��������Һ����ʱ����Һ��ɫͻ��Ϊ��ɫ������30s�ڲ���ɫ��˵���ζ������յ㣻
��6��������������Ԫ�ص���������Ϊa�����ݹ�ϵʽMnO4-��5Fe2+��5Fe��������������Ԫ�ص�����������
��� �⣺��1��Ϊ��ֹ�������Һ�л���BaCO3������Ӱ��ʵ������Ӧ�������еĶ�����̼��ȥ�����Լ�װ���п���NaOH��Һ������NaOH��Һ��SO2��Ӧ����SO32-��H2O��������������ܱ���������������������ӣ����з�Ӧ���ӷ���ʽΪSO2+2OH-=SO32-+H2O\2SO32-+O2=2SO42-��
�ʴ�Ϊ��NaOH��SO2+2OH-=SO32-+H2O��2SO32-+O2=2SO42-��
��2�������������ǿ�����ԣ������������������Ϊ�������ͬʱ����ˮ����Ӧ���ӷ���ʽΪ��SO32-+H2O2�TSO42-+H2O��
�ʴ�Ϊ��SO32-+H2O2�TSO42-+H2O��
��3��������Ԫ���غ㣬m1gFeS2�е���Ԫ������ȫ������BaSO4������n��BaSO4��=$\frac{m{\;}_{2}}{233}$mol������n��S��=$\frac{m{\;}_{2}}{233}$mol�����Ի���������Ԫ�ص���������Ϊ��$\frac{\frac{{m}_{2}}{233}mol��32g/mol}{m{\;}_{1}}$��100%=$\frac{32m{\;}_{2}}{233m{\;}_{1}}$��100%��
�ʴ�Ϊ��$\frac{32m{\;}_{2}}{233m{\;}_{1}}$��100%��
��4����IJ�����У�һ�����ʵ���Ũ����Һ�������У������ձ�������������ͷ�ι��⣬����Ҫ250mL����ƿ��
�ʴ�Ϊ��250mL����ƿ��
��5�����һ�θ��������Һ����ʱ����Һ��ɫͻ��Ϊ��ɫ������30s�ڲ���ɫ��˵���ζ������յ㣬
�ʴ�Ϊ�����һ�θ��������Һ����ʱ����Һ��ɫͻ��Ϊ��ɫ������30s�ڲ���ɫ��
��6��������������Ԫ�ص���������Ϊa����
MnO4-��5Fe2+��5Fe
1mol 5��56g
0.1mol/L��25��10-3L��10 m1a g
����1mol��0.1mol/L��25��10-3L��10=5��56g��m1a g
���a=$\frac{7}{m{\;}_{1}}$��
�ʴ�Ϊ��$\frac{7}{m{\;}_{1}}$��
���� ���⿼�����ʳɷֺͺ����IJⶨ���漰���û�ѧ�����Һ�����ơ�������ԭ�ζ�����ѧ����ȣ����н�ǿ���ۺ��ԣ�����ʱע��������غ����ϵʽ�ĽǶȷ�����

| A�� | $\frac{m}{a}L$ | B�� | $\frac{2m}{3a}L$ | C�� | $\frac{m+n}{a}L$ | D�� | $\frac{2��m+n��}{3a}L$ |
| A�� | CuSO4•3H2O | B�� | CuSO4•2H2O | C�� | CuSO4•H2O | D�� | CuSO4 |
| A�� | CH3COOH+CH3CH2OH $?_{��}^{Ũ����}$CH3COOCH2CH33+H2O | |
| B�� | CH2�TCH2+HBr��CH3CH2Br | |
| C�� | CH4+Cl2$\stackrel{��}{��}$CH3Cl+HCl | |
| D�� | 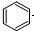 +Br2$\stackrel{FeBr_{3}}{��}$ +Br2$\stackrel{FeBr_{3}}{��}$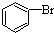 +HBr +HBr |
| A�� | H2SO4��NaHSO4�������� | B�� | ���ʹ�����ڼ� | ||
| C�� | KOH��NH3•H2O�����ڵ���� | D�� | Na2O��Na2O2�����ڼ��������� |