��Ŀ����
���б��������ڱ���һ���֣����е���Ŷ�Ӧ����Ԫ�أ���ش��������⣺
��1����ԭ�ӹ�����ص���ʽ����Ԫ�آٺ͢��γɵ���Է���������С�ķ����У��� ����Ԫ�آٺ͢��γɻ�������Ư�����õĵ���ʽ ��
��2��ijԪ�صļ۵����Ų�ʽΪnsnnpn+1����Ԫ����Ԫ�آ��γɵ���̬��������ӵĿռ乹��Ϊ�� ��
��3���Ȣ��Ԫ��ԭ��������2��Ԫ��ͨ����������γ��������ӣ������ӷ��ŷֱ��� �� �����н��ȶ����� ����ԭ���� ��
��4���ܢݢޢ�����Ԫ�صĵ�һ�������ɴ�С��˳���ǣ���Ԫ�ط��ţ� ��
��5��Ԫ�آ������ڱ��е�λ��Ϊ ��

��1����ԭ�ӹ�����ص���ʽ����Ԫ�آٺ͢��γɵ���Է���������С�ķ����У���
��2��ijԪ�صļ۵����Ų�ʽΪnsnnpn+1����Ԫ����Ԫ�آ��γɵ���̬��������ӵĿռ乹��Ϊ��
��3���Ȣ��Ԫ��ԭ��������2��Ԫ��ͨ����������γ��������ӣ������ӷ��ŷֱ���
��4���ܢݢޢ�����Ԫ�صĵ�һ�������ɴ�С��˳���ǣ���Ԫ�ط��ţ�
��5��Ԫ�آ������ڱ��е�λ��Ϊ
���㣺Ԫ�������ɺ�Ԫ�����ڱ����ۺ�Ӧ��
ר�⣺Ԫ����������Ԫ�����ڱ�ר��
��������Ԫ�������ڱ���λ�ã���֪��ΪH����ΪC����ΪO����ΪMg����ΪAl����ΪS����ΪCl����ΪCa����ΪNi����ΪCu��
��1��Ԫ�آٺ͢��γɵ���Է���������С�ķ���ΪCH4����ԭ�ӹ�����ص���ʽ���ۼ���Ϊ�Ҽ����м�������Ϊ�Ҽ���
Ԫ�آٺ͢��γɻ�������Ư�����ã��û�����ΪH2O2��������Oԭ��֮���γ�1�Թ��õ��Ӷԣ���ԭ������ԭ��֮���γ�1�Թ��õ��Ӷԣ�
��2��Ԫ�صļ۵����Ų�ʽΪnsnnpn+1��s�ܼ�����2�����ӣ��ʸ�Ԫ�صļ۵����Ų�ʽΪ2s22p3��Ϊ��Ԫ�أ�
��3���Ȣ��Ԫ��ԭ��������2��Ԫ��ΪFe��ͨ����������γ�Fe2+ Fe3+�������ӣ�������Χ�����Ų��Ƿ�Ϊ�ȶ�״̬���з������
��4��ͬ����������ң���һ�����ܳ��������ƣ��ܼ�Ϊȫ����������ȫ���ȶ�״̬�������ϵͣ���һ�����ܸ���ͬ��������Ԫ�أ�
��5���ɢ�Ԫ�������ڱ��е�λ�ÿ�֪���䴦�ڵ������ڢ�B�壮
��1��Ԫ�آٺ͢��γɵ���Է���������С�ķ���ΪCH4����ԭ�ӹ�����ص���ʽ���ۼ���Ϊ�Ҽ����м�������Ϊ�Ҽ���
Ԫ�آٺ͢��γɻ�������Ư�����ã��û�����ΪH2O2��������Oԭ��֮���γ�1�Թ��õ��Ӷԣ���ԭ������ԭ��֮���γ�1�Թ��õ��Ӷԣ�
��2��Ԫ�صļ۵����Ų�ʽΪnsnnpn+1��s�ܼ�����2�����ӣ��ʸ�Ԫ�صļ۵����Ų�ʽΪ2s22p3��Ϊ��Ԫ�أ�
��3���Ȣ��Ԫ��ԭ��������2��Ԫ��ΪFe��ͨ����������γ�Fe2+ Fe3+�������ӣ�������Χ�����Ų��Ƿ�Ϊ�ȶ�״̬���з������
��4��ͬ����������ң���һ�����ܳ��������ƣ��ܼ�Ϊȫ����������ȫ���ȶ�״̬�������ϵͣ���һ�����ܸ���ͬ��������Ԫ�أ�
��5���ɢ�Ԫ�������ڱ��е�λ�ÿ�֪���䴦�ڵ������ڢ�B�壮
���
�⣺��1��Ԫ�آ٢��γɵ���Է���������С�ķ���ΪCH4�����Ӵ���C-H����������Ϊ�Ҽ���
Ԫ�آٺ͢��γɻ�������Ư�����ã��û�����ΪH2O2��������Oԭ��֮���γ�1�Թ��õ��Ӷԣ���ԭ������ԭ��֮���γ�1�Թ��õ��Ӷԣ������ʽΪ��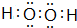 ��
��
�ʴ�Ϊ���ң�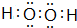 ��
��
��2��Ԫ�صļ۵����Ų�ʽΪnsnnpn+1��s�ܼ�����2�����ӣ��ʸ�Ԫ�صļ۵����Ų�ʽΪ2s22p3��Ϊ��Ԫ�أ���Ԫ������Ԫ���γɵ���̬���������ΪNH3���ռ乹��Ϊ�����ͣ�
�ʴ�Ϊ�������ͣ�
��3���Ȣ��Ԫ��ԭ��������2��Ԫ��ΪFe��ͨ����������γ�Fe2+ Fe3+�������ӣ�Fe3+��Χ�����Ų�Ϊ3d5 �����������ȶ�����Fe2+�����ȶ���
�ʴ�Ϊ��Fe2+��Fe3+��Fe3+��Fe3+��Χ�����Ų�Ϊ3d5�������������ȶ���
��4��ͬ����������ң���һ�����ܳ��������ƣ�Mg��2s�ܼ�����2�����ӣ�Ϊȫ���ȶ�״̬�������ϵͣ���һ�����ܸ���ͬ��������Ԫ�أ��ʵ�һ�����ܣ�Cl��S��Mg��Al���ʴ�Ϊ��Cl��S��Mg��Al��
��5���ɢ�Ԫ�������ڱ��е�λ�ÿ�֪���䴦�ڵ������ڢ�B�壬�ʴ�Ϊ���������ڢ�B�壮
Ԫ�آٺ͢��γɻ�������Ư�����ã��û�����ΪH2O2��������Oԭ��֮���γ�1�Թ��õ��Ӷԣ���ԭ������ԭ��֮���γ�1�Թ��õ��Ӷԣ������ʽΪ��
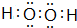 ��
���ʴ�Ϊ���ң�
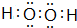 ��
����2��Ԫ�صļ۵����Ų�ʽΪnsnnpn+1��s�ܼ�����2�����ӣ��ʸ�Ԫ�صļ۵����Ų�ʽΪ2s22p3��Ϊ��Ԫ�أ���Ԫ������Ԫ���γɵ���̬���������ΪNH3���ռ乹��Ϊ�����ͣ�
�ʴ�Ϊ�������ͣ�
��3���Ȣ��Ԫ��ԭ��������2��Ԫ��ΪFe��ͨ����������γ�Fe2+ Fe3+�������ӣ�Fe3+��Χ�����Ų�Ϊ3d5 �����������ȶ�����Fe2+�����ȶ���
�ʴ�Ϊ��Fe2+��Fe3+��Fe3+��Fe3+��Χ�����Ų�Ϊ3d5�������������ȶ���
��4��ͬ����������ң���һ�����ܳ��������ƣ�Mg��2s�ܼ�����2�����ӣ�Ϊȫ���ȶ�״̬�������ϵͣ���һ�����ܸ���ͬ��������Ԫ�أ��ʵ�һ�����ܣ�Cl��S��Mg��Al���ʴ�Ϊ��Cl��S��Mg��Al��
��5���ɢ�Ԫ�������ڱ��е�λ�ÿ�֪���䴦�ڵ������ڢ�B�壬�ʴ�Ϊ���������ڢ�B�壮
�����������Ƕ����ʽṹ�Ŀ��飬�漰Ԫ�����ڱ�����ѧ�������ӽṹ�������ܵȣ��ѶȲ���ע��ͬ���ڵ�һ�����ܵ������ԣ�

��ϰ��ϵ�д�
 ������ÿ�ʱ�Ż���ҵϵ�д�
������ÿ�ʱ�Ż���ҵϵ�д�
�����Ŀ
 ��ѧ��������Ƴ�������Ϊ��Ч����ƽ�����N��NO2��3����ͼ��ʾ������֪�÷�����N-N-N���Ƕ���108.1�㣬�����й�N��NO2��3��˵����ȷ���ǣ�������
��ѧ��������Ƴ�������Ϊ��Ч����ƽ�����N��NO2��3����ͼ��ʾ������֪�÷�����N-N-N���Ƕ���108.1�㣬�����й�N��NO2��3��˵����ȷ���ǣ�������| A�����������й��ۼ���Ϊ�Ǽ��Լ� |
| B���������ĸ���ԭ�ӹ�ƽ�� |
| C��N��NO2��3�������ڼ��Է��� |
| D��N��NO2��3�����е�Nֻ��һ�ֻ��ϼ� |
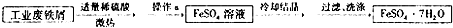
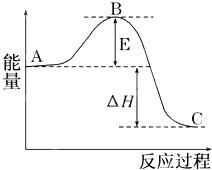 2SO2��g��+O2��g��?2SO3��g����Ӧ���̵������仯��ͼ��ʾ����֪1mol SO2��g������Ϊ1mol SO3��g���ġ�H=-99kJ/mol��
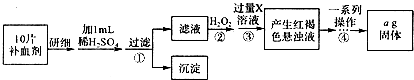
2SO2��g��+O2��g��?2SO3��g����Ӧ���̵������仯��ͼ��ʾ����֪1mol SO2��g������Ϊ1mol SO3��g���ġ�H=-99kJ/mol�� ÿ��COCl2�����ں���
ÿ��COCl2�����ں���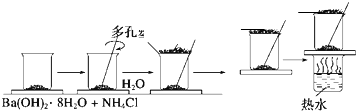 ���о���ѧ��Ӧ�е������仯ʱ������ͨ���������ʵ�飺��һ��С�ձ������20g����ĥ�ɷ�ĩ��Ba��OH��2?8H2O����С�ձ����������ѵ���3��4��ˮ�IJ���Ƭ�ϣ�Ȼ�����ձ��м���Լ10g NH4Cl���壬����ʵ�鲽�裬��д�±������ش����⣮
���о���ѧ��Ӧ�е������仯ʱ������ͨ���������ʵ�飺��һ��С�ձ������20g����ĥ�ɷ�ĩ��Ba��OH��2?8H2O����С�ձ����������ѵ���3��4��ˮ�IJ���Ƭ�ϣ�Ȼ�����ձ��м���Լ10g NH4Cl���壬����ʵ�鲽�裬��д�±������ش����⣮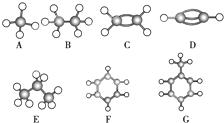 A��G�Ǽ������ķ������ģ�ͣ���ͼ�����ݴ˻ش��������⣺
A��G�Ǽ������ķ������ģ�ͣ���ͼ�����ݴ˻ش��������⣺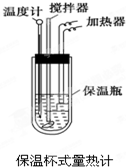 �����ȼ��У���ͼ����100mL 0.50mol?L-1��CH3COOH��Һ��100mL 0.55mol?L-1��NaOH��Һ��ϣ��¶ȴ�298.0K������300.7K����֪���ȼƵ����ݳ��������ȼƸ�����ÿ����1K����Ҫ��������150.5J?K-1����Һ�ܶȾ�Ϊ1g?mL-1��������Һ�ı�����c=4.184J?��g?K��-1��
�����ȼ��У���ͼ����100mL 0.50mol?L-1��CH3COOH��Һ��100mL 0.55mol?L-1��NaOH��Һ��ϣ��¶ȴ�298.0K������300.7K����֪���ȼƵ����ݳ��������ȼƸ�����ÿ����1K����Ҫ��������150.5J?K-1����Һ�ܶȾ�Ϊ1g?mL-1��������Һ�ı�����c=4.184J?��g?K��-1��