��Ŀ����
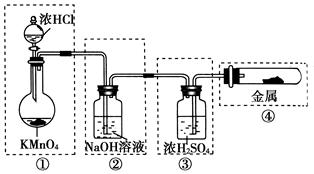
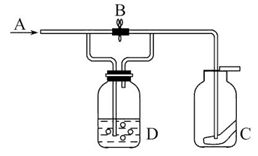
ij��ѧ��ȤС��Ϊ��̽��ijЩ���������,�������ͼ��ʾ��ʵ��װ�á�ʵ��ʱ��A��D�в���������ͬʱͨ��C�С�(KΪֹˮ�У����ּг���������ȥ)
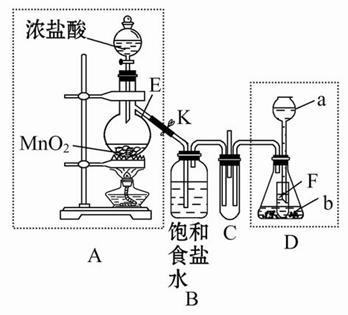
�ش��������⣺
(1)����ʵ��ǰ���A����װ�������Եķ����ߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߡ�
(2)����E�������ǣߣߣߣߣߣߣ���ƿ��С�Թ�F�������ǣߣߣߣߣߣߡ�
(3)��b����ʯ�ң�a�ǣߣߣߣߣߣ�ʱ����C���а��̲�����д�����ɰ��̵Ļ�ѧ����ʽ�ߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߡ�
(4)��a����������Ϊ75%�����ᣬb���������Ʒ�ĩ����C��ʢ������BaCl2��Һʱ��д��C�з�����Ӧ�����ӷ���ʽ���ߣߣߣߣߣߣߣߣߣߣߣߡ�
(5)�ӻ����ĽǶȳ���������ʵ��װ������Ҫ�Ľ����ǣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߡ�

�ش��������⣺
(1)����ʵ��ǰ���A����װ�������Եķ����ߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߡ�
(2)����E�������ǣߣߣߣߣߣߣ���ƿ��С�Թ�F�������ǣߣߣߣߣߣߡ�
(3)��b����ʯ�ң�a�ǣߣߣߣߣߣ�ʱ����C���а��̲�����д�����ɰ��̵Ļ�ѧ����ʽ�ߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߡ�
(4)��a����������Ϊ75%�����ᣬb���������Ʒ�ĩ����C��ʢ������BaCl2��Һʱ��д��C�з�����Ӧ�����ӷ���ʽ���ߣߣߣߣߣߣߣߣߣߣߣߡ�
(5)�ӻ����ĽǶȳ���������ʵ��װ������Ҫ�Ľ����ǣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߡ�
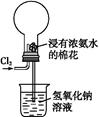
(1)�ر�ֹˮ��K����Һ©������������Һ©����������ƿ�м�ˮ,�Ժ���ˮ���ܼ��룬˵��A������������(����B�м�����ˮ����A�ĵ��ܳ��ڽ���ˮ�У��رշ�Һ©���������þƾ��ƻ��ָ�������ƿ�ȣ���B�е��ܳ����������ݳ���Ϩ��ƾ��ƻ��ɿ��ֺ������и߳�Һ���ˮ����һ��ʱ�䲻�½�,˵������������)
(2)������ƿ ����Һ���������γ�Һ��,��ֹ�����ݳ�
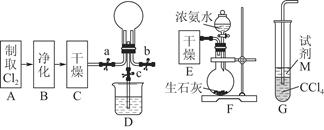
(3)Ũ��ˮ 3Cl2+8NH3=6NH4Cl+N2(�ֲ�д��ѧ����ʽҲ��)
(4)Cl2+SO2+Ba2++2H2O=2Cl-+BaSO4��+4H+ (�ֲ�д���ӷ���ʽҲ��)
(5)��C�ܿ����β������װ�ã���ֹ��Ⱦ����
(2)������ƿ ����Һ���������γ�Һ��,��ֹ�����ݳ�
(3)Ũ��ˮ 3Cl2+8NH3=6NH4Cl+N2(�ֲ�д��ѧ����ʽҲ��)
(4)Cl2+SO2+Ba2++2H2O=2Cl-+BaSO4��+4H+ (�ֲ�д���ӷ���ʽҲ��)
(5)��C�ܿ����β������װ�ã���ֹ��Ⱦ����
��ʵ���ǿ���������ȡ��̽���������ʵ�ʵ�顣A�����ɵ���Cl2��D����ƿ�м�һС�Թܿ��Լ���Һ���������ͬʱ�Գ���©���γ�Һ�⡣��D������ʯ�Һ�Ũ��ˮ���û�����NH3��NH3��Cl2��C�з�����Ӧ����NH4Cl���������̣���D����Ũ�����Na2SO3�����ĩ��Ӧ������SO2����C��Cl2��SO2��BaCl2��Һ������Ӧ����BaSO4���ɡ���װ��ȱ��β������װ�ã���Կ���������Ⱦ��

��ϰ��ϵ�д�
 ��������һ���þ�ϵ�д�
��������һ���þ�ϵ�д� Сѧ��10����Ӧ����ϵ�д�
Сѧ��10����Ӧ����ϵ�д�
�����Ŀ