��Ŀ����
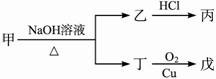
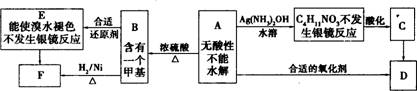
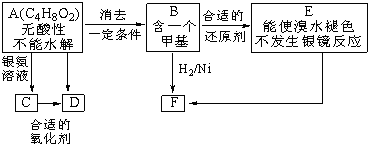
��֪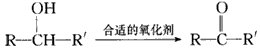 ��ע��R��R'Ϊ��������AΪ�л��ϳ��м��壬��һ�������·�����ȥ��Ӧ�����ܵõ����ֻ�Ϊͬ���칹��IJ�����е�һ��B��������ȡ�ϳ���֬��Ⱦ�ϵȶ��ֻ�����Ʒ��A�ܷ�������ͼ��ʾ�ı仯���Իش�
��ע��R��R'Ϊ��������AΪ�л��ϳ��м��壬��һ�������·�����ȥ��Ӧ�����ܵõ����ֻ�Ϊͬ���칹��IJ�����е�һ��B��������ȡ�ϳ���֬��Ⱦ�ϵȶ��ֻ�����Ʒ��A�ܷ�������ͼ��ʾ�ı仯���Իش�
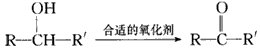 ��ע��R��R'Ϊ��������AΪ�л��ϳ��м��壬��һ�������·�����ȥ��Ӧ�����ܵõ����ֻ�Ϊͬ���칹��IJ�����е�һ��B��������ȡ�ϳ���֬��Ⱦ�ϵȶ��ֻ�����Ʒ��A�ܷ�������ͼ��ʾ�ı仯���Իش�
��ע��R��R'Ϊ��������AΪ�л��ϳ��м��壬��һ�������·�����ȥ��Ӧ�����ܵõ����ֻ�Ϊͬ���칹��IJ�����е�һ��B��������ȡ�ϳ���֬��Ⱦ�ϵȶ��ֻ�����Ʒ��A�ܷ�������ͼ��ʾ�ı仯���Իش� 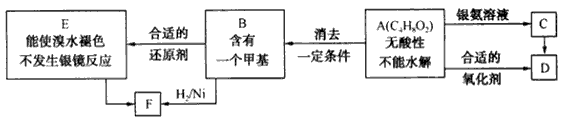
(1)д��������������A��ͬ���칹��Ľṹ��ʽ������дһ�֣��� a����������________��b���ܷ���ˮ�ⷴӦ_________��
(2)A�����еĹ�������____��D�Ľṹ��ʽ��____��
(3)C��D�ķ�Ӧ������____��E��F�ķ�Ӧ������____����д��Ӧ��ĸ����
a��������Ӧ
b����ԭ��Ӧ
c���ӳɷ�Ӧ
d��ȡ����Ӧ
(4)д����ѧ����ʽ��A��B___________ ��
(5)д��E���ɸ߾���Ļ�ѧ����ʽ_________ ��
(6)C��ͬ���칹��C1��C����ͬ�����ţ�������C1��ȥ������ˮ�γɺ�����Ԫ����C2��д��C2�Ľṹ��ʽ��________��
(2)A�����еĹ�������____��D�Ľṹ��ʽ��____��
(3)C��D�ķ�Ӧ������____��E��F�ķ�Ӧ������____����д��Ӧ��ĸ����
a��������Ӧ
b����ԭ��Ӧ
c���ӳɷ�Ӧ
d��ȡ����Ӧ
(4)д����ѧ����ʽ��A��B___________ ��
(5)д��E���ɸ߾���Ļ�ѧ����ʽ_________ ��
(6)C��ͬ���칹��C1��C����ͬ�����ţ�������C1��ȥ������ˮ�γɺ�����Ԫ����C2��д��C2�Ľṹ��ʽ��________��
(1)CH3CH2CH2COOH ��CH3CH2COOCH3
(2)�ǻ�,ȩ�� �� CH3COCH2COOH
(3)a��bc
(4)CH3CH(OH)CH2CHO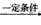 CH3CH=CHCHO+H2O
CH3CH=CHCHO+H2O
(5)nCH3CH=CHCH2OH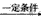
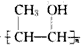
(6) ��
��
(2)�ǻ�,ȩ�� �� CH3COCH2COOH
(3)a��bc
(4)CH3CH(OH)CH2CHO
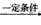 CH3CH=CHCHO+H2O
CH3CH=CHCHO+H2O(5)nCH3CH=CHCH2OH
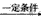
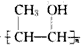
(6)
 ��
��

��ϰ��ϵ�д�
�����Ŀ
 ��ע��R��R?Ϊ������
��ע��R��R?Ϊ������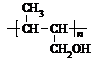

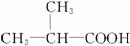 (ע��R��R��Ϊ����)
(ע��R��R��Ϊ����) 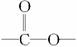
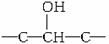 (ע��R��R���ʾ����)��
(ע��R��R���ʾ����)��