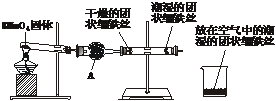
��Ŀ����
�������DZȽϳ���������ijʵ��С��Ϊ�о����������������������¿��١����еķ�����
���ȼ������װ�õ������ԣ�Ȼ��ͼ���Ӻ�װ�ã���ȼ�ƾ��Ƹ�ҩƷ���ȣ�����3�������ң��۲쵽��ʵ������Ϊ����ֱ�ι���������ˮ�����Ĺ�����˿������ɫ��ûҰ���������ʴ����ֱ�ι��и������˿������Ȼ������û�з�����ʴ�����ձ��г�ʪ����˿������Ȼ������
�Իش��������⣺
(1)����������Ӵ��Ľ��ʲ�ͬ��������ʴ�ֳɲ�ͬ���ͣ���ʵ��������������__________���ܱ�ʾ��ԭ���ĵ缫��ӦʽΪ_____________________________________________________��
(2)����A������Ϊ������װ��ҩƷ������__________________________________����������_____________________________________________________________________��
(3)��ʵ���֪�����������������Ϊ__________�����������������һ����Ҫ������________________________________________��
(1)�绯ѧ��ʴ
������Fe-2e-![]() Fe2+
Fe2+
������2H2O+O2+4e-![]() 4OH-
4OH-
(2)����� ��ʯ�һ�P2O5��CaCl2
(3)����O2�Ӵ� ����ˮ�Ӵ� ����Ũ��
�����������ڳ�ʪ�������γ�ԭ��أ������绯ѧ��ʴ����ʵ����ֱ�ι��е�����˵������ʪ��������ı���ǰ�ᣬ��û���漰��ʪ�̶ȶ����������Ӱ�죬ֱ�ι�ʵ�����ձ�ʵ��ĶԱȣ���˵��O2Ũ����Ӱ�����������һ����Ҫ���ء�

 ���ſ����ϵ�д�
���ſ����ϵ�д� ���Ŀ����ϵ�д�
���Ŀ����ϵ�д� ������ӱ������ͯ������ϵ�д�
������ӱ������ͯ������ϵ�д�