��Ŀ����
18������ͭ�ڸ����¿ɷֽ������ͭ��δ֪���ʣ�ij����С��ͬѧ����ϸͼ1��ʾ��װ�ã�ͨ��ʵ���������ͭ�ķֽ�������֪SO3���۵���16.8�棬�е���44.8�棩��1����ͬѧ��쵽װ��B������ɫ������֣������ɫ����ΪSO3��д��ѧʽ��������Bװ�û�ΪDװ�ã���ͼ2������ʵ������пɹ۲쵽�������Dz���������
��2������ͬѧ��쵽װ��B�������Թ���װ��C�г���ʯ��ˮ����ǣ�������ͭ�ֽ�Ļ�ѧ����ʽΪ2CuSO4$\frac{\underline{\;\;��\;\;}}{\;}$ 2CuO+2SO2��+O2����
��3����ͬѧ��쵽B�г��־��壬C�г��ֻ��ǣ�������C�г��ֻ��ǵ�ԭ�������װ���е�CO2�йأ�Ϊ����֤�Լ����뷨�����������ͼ3װ�����Cװ�ã�
�ɹ�ѡ����Լ��У�����KMnO4��Һ��Ʒ����Һ�����ͳ���ʯ��ˮ��
��a�е��Լ�������KMnO4��Һ��
��b��Ʒ����Һ�������Ǽ�����������Ƿ����������ֵ�b��Ʒ�첻��ɫ��װ��c�г���ʯ��ˮ����ǵ�������˵��ȷ��CO2������װ���и���ʵ�飮
��4�����ʵ��֤��������Ϊ��Ԫ��õ�Ũ�ȵ�����������Һ�ζ���������Һ����������������Һ��������������2������֤�����������Ƕ�Ԫ�ᣮ

���� ��1������SO3���۵���16.8�棬�е���44.8���֪��ˮʹ�������������ɹ��壻����������ˮ���ղ���������
��2��B�г��־��壬C�г��ֻ��ǣ�˵�����������������ʹʯ��ˮ����ǵ������Ƕ�����������ͭ����Ԫ�ػ��ϼ۽��ͣ�ֻ������Ԫ�ػ��ϼ����ߣ�����������
��3��Ϊ��֤��ʵ���������̼�йأ���Ҫ�ڳ������������ǰ���¹۲�ʯ��ˮ�Ƿ����ǣ�
��4���õ�Ũ�ȵ�����������Һ�ζ���������Һ����������������Һ��������������2����
��� �⣺��1������SO3���۵���16.8�棬�е���44.8���֪��ˮʹ�������������ɹ��壻����������ˮ���ղ���������
�ʴ�Ϊ��SO3������������
��2��B�г��־��壬C�г��ֻ��ǣ�˵�����������������ʹʯ��ˮ����ǵ������Ƕ�����������ͭ����Ԫ�ػ��ϼ۽��ͣ�ֻ������Ԫ�ػ��ϼ����ߣ�������������ѧ����ʽΪ��2CuSO4$\frac{\underline{\;\;��\;\;}}{\;}$ 2CuO+2SO2��+O2����
�ʴ�Ϊ��2CuSO4$\frac{\underline{\;\;��\;\;}}{\;}$ 2CuO+2SO2��+O2����
��3����Ϊ��֤��ʵ���������̼�йأ���Ҫ�ڳ������������ǰ���¹۲�ʯ��ˮ�Ƿ����ǣ����������������Ը��������Һ��a���Լ�Ϊ���Ը��������Һ��
�ʴ�Ϊ������KMnO4��Һ��
��a���Լ�Ϊ���Ը��������Һ��������������Ϊȷ��������Ʒ�������飬��b��Ʒ�첻��ɫ��װ��c�г���ʯ��ˮ����ǣ���֤���˶�����̼�ĸ�£��
�ʴ�Ϊ��������������Ƿ�������b��Ʒ�첻��ɫ��װ��c�г���ʯ��ˮ����ǣ�
��4���õ�Ũ�ȵ�����������Һ�ζ���������Һ����������������Һ��������������2������֤�����������Ƕ�Ԫ�ᣬ
�ʴ�Ϊ���õ�Ũ�ȵ�����������Һ�ζ���������Һ����������������Һ��������������2������֤�����������Ƕ�Ԫ�ᣮ
���� ����̽������ͭ�ֽܷ�õ�������ʲô��ͨ������ʵ�鲢���������ԭ��Ӧ�Ĺ����ƶϳ��������ʵ����ƣ���������ѧ������֪ʶ��������

 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д�| A�� | ��Ȼ�������н���C��H��0��N����Ԫ�� | |
| B�� | ���루NH4��2S04������Һ��ʹ�����ʱ��� | |
| C�� | �ؽ�������ʹ���������ᣬ������ʳ�ؽ����λ��ж� | |
| D�� | ���������ʳ�Σ���ʹ�����ʱ��� |
 ijѧ��������֪���ʵ���Ũ�ȵ�����ȥ�ⶨ�ڿ�����¶��һ��ʱ����NaOH����Ĵ��ȣ�������·�����
ijѧ��������֪���ʵ���Ũ�ȵ�����ȥ�ⶨ�ڿ�����¶��һ��ʱ����NaOH����Ĵ��ȣ�������·������ٳ�ȡW g��Ʒ��ȷ���500mL��Һ��
��ȷ����0.1000mol/L�ı����ᣮ
���õζ���ȡ�����Ƽ�Һ25.00mL����ƿ������ƿ�еμ��μ�����Һ��
������ƿ����ε�������ᣬֱ���յ㣮
�ݼ�¼���ݣ����㣮��ش�
��1���ڢٲ�����Ҫʹ�õĶ����IJ���������500ml����ƿ��
��2��������װ������ĵζ�������ʽ�ζ��ܣ�
��3���ζ����������ֿ�����ʽ�ζ��ܵĻ�����������һ������ҡ����ƿ���۾�Ӧʼ��ע����ƿ����Һ����ɫ�仯���жϵζ������յ�������ǵμ����һ�����ᣬ��Һ����ɫ�ɻ�ɫ��Ϊ��ɫ�����ڰ�����ڲ���ɫ��
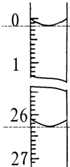
��4�����ζ���ʼ�ͽ���ʱ����ʽ�ζ����е�Һ����ͼ��ʾ��������������Һ�����Ϊ26.10ml��
��5��ijѧ������3��ʵ��ֱ��¼�й����������
| �ζ� ���� | ����NaOH��Һ�����/mL | 0.1000mol/L��������/mL | ||
| �ζ�ǰ�̶� | �ζ���̶� | ƽ���������� ���/mL | ||
| ��һ�� | 25.00 | 0.00 | 26.15 | ��V=26.20 |
| �ڶ��� | 25.00 | 0.56 | 30.30 | |
| ������ | 25.00 | 0.20 | 26.45 | |
�۸�������������NaOH�Ĵ���$\frac{2.096}{W}$��100%��
��6�����в����У�����ʹ�ⶨ���ƫ�͵���ACF��
A����ȡ��Ʒʱ����Ʒ������λ�÷ŷ���
B�����Ʊ���������У���ȡ��Ͳ����ʱ�����ӿ̶���
C����ʽ�ζ�����ϴ��δ�ô���Һ��ϴ
D����ʽ�ζ�����ϴ��δ�ô���Һ��ϴ
E����ƿװҺǰδ�����ˮ�鸽����ƿ��
F����ȡ��ʽ�ζ��ܶ���ʱ����һ�ζ������ӣ��ڶ��ζ������ӣ�
| A�� | ��ij�����еμ����������ʹ����ʯ��ˮ����ǵ���ɫ��ζ���������֤����CO32- | |
| B�� | ��ij�����������ɫʵ�飬����ɫ�ܲ����۲죬��ɫΪ��ɫ��֤������ | |
| C�� | ����Һ��μ���NaOH��Һ���а�ɫ��״����������������������ʧ����ԭ��Һ�п��ܺ���Al3+ | |
| D�� | ����Һ���ȼ���KSCN��Һ�������������ټ�����ˮ����Һ��Ѫ��ɫ�������Һ��һ������Fe2+ |
| A�� | һ�������£���1 mol N2��3 mol H2��Ϸ�����Ӧ��ת�Ƶĵ�������Ϊ6 NA | |
| B�� | 1 L 0.1 mol•L-1��Na2CO3��Һ�������ӵ���������0.1 NA | |
| C�� | ��״���£�44.8LHF�к��з��ӵ���ĿΪ2NA | |
| D�� | 1 mol-CH3�������ĵ�������Ϊ10 NA |

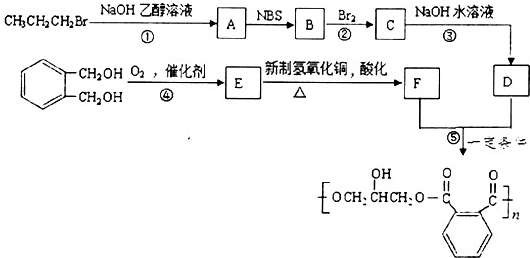
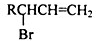
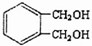 ��ͬ���칹����ͬʱ�������������ķ����廯�����д������һ�ֵĽṹ��ʽ
��ͬ���칹����ͬʱ�������������ķ����廯�����д������һ�ֵĽṹ��ʽ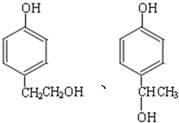 ������֮һ����
������֮һ����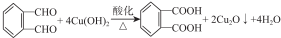 ��
��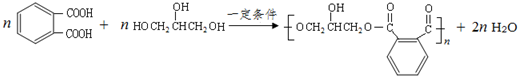 ��
��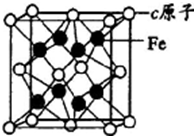 a��b��c��d������ԭ��������������Ķ�����Ԫ�أ�aԭ�ӵĵ��Ӳ���Ϊn��������������2n2-1������������Ϊ2n+l��b��dͬ���壬���γ�������ѧ�����Ļ����c��b��ɵĻ�������һ�������������ҵ��ͨ�����û������ұ��c���ʣ�eԭ�����ĸ��ܲ㣬��δ�ɶԵ�������ͬ���������ģ��ش��������⣺
a��b��c��d������ԭ��������������Ķ�����Ԫ�أ�aԭ�ӵĵ��Ӳ���Ϊn��������������2n2-1������������Ϊ2n+l��b��dͬ���壬���γ�������ѧ�����Ļ����c��b��ɵĻ�������һ�������������ҵ��ͨ�����û������ұ��c���ʣ�eԭ�����ĸ��ܲ㣬��δ�ɶԵ�������ͬ���������ģ��ش��������⣺