��Ŀ����
ij���Ͻ�Ӳ�����к�������þ��ͭ���裬Ϊ�˲ⶨ�úϽ������ĺ������������������ʵ�飺����֪�費�������ᣩ
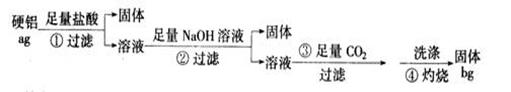
��1����ȫ�����٢ڢܸۢ�����Ӧ�����ӷ���ʽ
��Mg+2H+===Mg2++H2�� ��___________________________
��_______________________�� _______________________
Mg2++2OH��===Mg(OH)2��
�� ___________________________________
CO2+H2O+ A1O2��===Al(OH)3��+3HCO3��
|
��2A1(OH)3===Al2O3+H2O
��2������Ʒ����������������
��3���ڢڲ��м���NaOH��Һ����ʱ����ʹ�ⶨ��� ���ڢܲ��еij���û��������ˮϴ��ʱ����ʹ�ⶨ��� ���ڢܲ��Գ������ղ����ʱ����ʹ�ⶨ��� �����A������B����C����
A��ƫ�� B��ƫ�� C����Ӱ��
��1��2Al+6H+ ===2 A13++3 H2�� , H++OH-=== H20[
A13++ 4OH��===A102-+2H20�� CO2+ OH-=== HCO3��
��2�� ![]()
��3�� B��A��A

��ϰ��ϵ�д�
�����Ŀ