��Ŀ����
4�� ��֪A��B��C��D��E�������ڱ���ǰ�����ڵ�Ԫ�أ����ǵ�ԭ������������������AԪ��ԭ�ӵĺ���p��������s��������1��C�ǵ縺������Ԫ�أ�Dԭ�Ӵ���������������������2����E�ǵڢ�����ԭ��������С��Ԫ�أ�
��֪A��B��C��D��E�������ڱ���ǰ�����ڵ�Ԫ�أ����ǵ�ԭ������������������AԪ��ԭ�ӵĺ���p��������s��������1��C�ǵ縺������Ԫ�أ�Dԭ�Ӵ���������������������2����E�ǵڢ�����ԭ��������С��Ԫ�أ���1��д����̬Cԭ�ӵĵ����Ų�ʽ1s22s22p5��
��2��A��B����Ԫ�صĵ�һ�������ɴ�С��˳��ΪN��O����Ԫ�ط��ű�ʾ����ԭ����Nԭ�ӵ�2p�ܼ����ڽ��ȶ��İ���״̬��
��3����֪DC4������Ϊ���壬������ʵľ��������Ƿ��Ӿ��壬�����������ԭ�ӵĹ���ӻ�����Ϊsp3���ռ乹�����������壮
��4��Cu2+������AH3�γ�������[Cu��AH3��4]2+����AC3������Cu2+�γ������ӣ���ԭ���ǵ縺��F��N��H����NF3�����У����õ��Ӷ�ƫ��Fԭ�ӣ�ʹ��Nԭ���Ϲµ��Ӷ�����ͭ�����γ���λ����
��5��A��B��Ԫ�طֱ���D�γɵĹ��ۼ��У����Խ�ǿ����Si-O��A��B��Ԫ�ؼ����γɶ��ֶ�Ԫ�����������A3-��Ϊ�ȵ���������ʵĻ�ѧʽΪN2O��
��6����֪E���ʵľ�����ͼ��ʾ��������Eԭ�ӵ���λ��Ϊ8��һ��E�ľ�������Ϊ$\frac{112}{{N}_{A}}$g��
���� AԪ��ԭ�ӵĺ���p��������s��������1������A�ĺ�������Ų�Ϊ��1s22s22p3�������ǵ�Ԫ�أ�C�ǵ縺������Ԫ���Ƿ�Ԫ�أ�Dԭ�Ӵ���������������������2���������ǹ�Ԫ�أ�E�ǵڢ�����ԭ��������С��Ԫ������Ԫ�أ���A��B��C��D��E�������ڱ���ǰ�����ڵ�Ԫ�أ����ǵ�ԭ������������������Ԫ��B����Ԫ�أ��ɴ˷������
��� �⣺��1����ԭ�ӵĵ����Ų�ʽ1s22s22p5���ʴ�Ϊ��1s22s22p5��
��2��N��O����Ԫ�صĵ�һ�����ܣ���Ԫ�ص�2p������ڰ���״̬�����Ե��ĵ�һ�����ܴ������ĵ�һ�����ܣ����Դ�С��˳��ΪN��O���ʴ�Ϊ��N��O��Nԭ�ӵ�2p�ܼ����ڽ��ȶ��İ���״̬��
��3����֪�ķ����賣����Ϊ���壬�����ķ������Ƿ��Ӿ��壬����ԭ�ӹ���sp3�ӻ����ռ乹������������ṹ���ʴ�Ϊ�����Ӿ��壻sp3���������壻
��4��NF3�����У����õ��Ӷ�ƫ��Fԭ�ӣ�ʹ��Nԭ���Ϲµ��Ӷ�����ͭ�����γ���λ��������NF3������Cu2+�γ������ӣ��ʴ�Ϊ���縺��F��N��H����NF3�����У����õ��Ӷ�ƫ��Fԭ�ӣ�ʹ��Nԭ���Ϲµ��Ӷ�����ͭ�����γ���λ����
��5����ΪOԪ�صķǽ����Դ���N������O-Si����N-Si�����ȵ�������ԭ�Ӹ�����ȼ��۵�������ȣ�N3-�к���3��ԭ�ӡ��۵�������16��������ӻ�Ϊ�ȵ�����ĵ�������ΪN2O���ʴ�Ϊ��O-Si����N2O��
��6���þ�������������������������ԭ�ӵ���λ��Ϊ8���þ�����Feԭ�Ӹ���=1+8��$\frac{1}{8}$=2�������ϵ�Feԭ�ӱ������ϵ�8��ԭ�Ӱ�Χ���������������8��ÿ��Feԭ�ӵ�����=$\frac{M}{{N}_{A}}$g����þ���������������Feԭ������=2��$\frac{M}{{N}_{A}}$g=2��$\frac{56}{{N}_{A}}$g=$\frac{112}{{N}_{A}}$g��
�ʴ�Ϊ��8��$\frac{112}{{N}_{A}}$g��
���� ���⿼�����ʽṹ�����ʣ�Ϊ��Ƶ���㣬�漰�������㡢�ȵ������жϡ�ԭ�ӽṹ�����ռ乹���жϵ�֪ʶ�㣬���ؿ���ѧ�������жϼ���������

| A�� | п�̸ɵ�ع���һ��ʱ���̼����ϸ | |
| B�� | �ں������������п��ɼ�������ĸ�ʴ���Dz������������������������� | |
| C�� | ͭпԭ��ع���ʱ�����������·��ͭ�缫����п�缫 | |
| D�� | ���MgCl2������Һ�����Ƶý���þ |
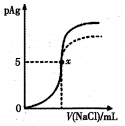 ��֪��pAg=-lg[c��Ag+��]��Ksp��AgCl��=1��l0-10��Ksp��AgI��=l��l0-17����ͼ����l0mLAgNO3��Һ�м���0��lmol/LNaCI��Һʱ����Һ��pAg���ż���NaCl��Һ������仯��ͼ��ʵ�ߣ�������ͼ���������н�����ȷ���ǣ�������
��֪��pAg=-lg[c��Ag+��]��Ksp��AgCl��=1��l0-10��Ksp��AgI��=l��l0-17����ͼ����l0mLAgNO3��Һ�м���0��lmol/LNaCI��Һʱ����Һ��pAg���ż���NaCl��Һ������仯��ͼ��ʵ�ߣ�������ͼ���������н�����ȷ���ǣ�������| A�� | ԭAgNO3��Һ�����ʵ���Ũ��Ϊ0��l mol/L | |
| B�� | ͼ��x���ʾ��Һ��c��Ag+��=c��Cl-�� | |
| C�� | ͼ��x�������Ϊ��10��5�� | |
| D�� | ��NaCl��Һ����0.1 mol/L��MaCl��Һ����ͼ�����յ���Ϊ���߲��� |
��2KClO3$\frac{\underline{\;MnO_{2}\;}}{��}$2KCl=3O2��
��2HgO$\frac{\underline{\;\;��\;\;}}{\;}$2Hg+O2��
��2KMnO4$\frac{\underline{\;\;��\;\;}}{\;}$K2MnO4+MnO2+O2��
��2H2O2$\frac{\underline{\;MnO_{2}\;}}{\;}$O2��+2H2O
��Ҫ�Ƶ���ͬ��������������Ӧ�е���ת����Ŀ֮��Ϊ��������
| A�� | 3��1��1��2 | B�� | 2��2��2��1 | C�� | 3��1��4��2 | D�� | 2��1��1��1 |
| ����� | �Լ� | ���뷽�� | |
| A | �������ӣ� | ��ˮ | ���� |
| B | ���飨��ϩ�� | ���Ը��������Һ | ϴ�� |
| C | �������������ᣩ | ����Na2CO3��Һ | ��Һ |
| D | �屽���嵥�ʣ� | �� | ��ȡ |
| A�� | A | B�� | B | C�� | C | D�� | D |
| A�� | ԭ�Ӱ뾶��A��B��C | |
| B�� | ����Ԫ�ص�����������Ӧˮ��������ɻ��Ϸ�Ӧ�õ� | |
| C�� | A���⻯���ȶ���С��C���⻯���ȶ��� | |
| D�� | B ��A�����������ɷ����û���Ӧ |
| A�� | ��ɱ������� | B�� | �����������ʯ�ҵ��ۻ� | ||
| C�� | �Ȼ��ƺ������ۻ� | D�� | ���ʯ���ۻ��ͱ����ڻ� |
��ҵ������úת��ΪCO��������CO��ˮ������Ӧ��H2ʱ����������ƽ�⣺CO��g��+H2O��g��?CO2��g��+H2��g��
��1����1L�����ܱ������г���CO��H2O��g����800��ʱ��ò����������±���
| t/min | 0 | 1 | 2 | 3 | 4 |
| n��H2O��/mol | 0.600 | 0.520 | 0.450 | 0.350 | 0.350 |
| n��CO��/mol | 0.400 | 0.320 | 0.250 | 0.150 | 0.150 |
��2����ͬ�����£���2L�����ܱ������г���1molCO��1mol H2O��g����2molCO2��2mo1H2��
��ʱ���������� �����棩���������=����������
��3��һ���¶��£��ں����ܱ�������NO2��O2��Ӧ���£�4NO2��g��+O2��g��?2N2O5��g��������֪K350�棼K300�棬��÷�Ӧ�Ƿ��ȷ�Ӧ������ȡ����ȡ�����
����֪CO��g����H2��g����CH3OH��l����ȼ���ȷֱ�Ϊ283kJ•mol-1��286kJ•mol-1��726kJ•mol-1
��4������CO��H2�ϳ�Һ̬�״����Ȼ�ѧ����ʽΪCO��g��+2H2��g��=CH3OH��l����H=-129kJ•mol-1��
��5�����ݻ�ѧ��Ӧԭ������������ѹǿ���Ʊ��״���Ӧ��Ӱ������ѹǿʹ��Ӧ���ʼӿ죬ͬʱƽ�����ƣ�CH3OH��������
��Ϊ���Ѷ�ʯ�͵Ĺ���������������Ա��úҺ���Ʊ����ͣ������������ȼ�ϵ�أ���ع���ԭ����ͼ��ʾ��

һ���缫ͨ����������һ�缫ͨ������������������Dz�����Y2O3��ZrO2���壬���ڸ������ܴ���O${\;}_{2}^{-}$
��6�������飨C8H18���������ͣ�д���õ�ع���ʱ�ĸ�����Ӧ����ʽC8H18-50e-+25O2-=8CO2+9H2O��
��7����֪һ�����ӵĵ�����1.602��10-19C���øõ�ص�ⱥ��ʳ��ˮ������·��ͨ��1.929��105C�ĵ���ʱ������NaOH80g��
����úȼ�ղ�����CO2���������ЧӦ����Ҫ����֮һ��
��8����CO2ת�����л������Ч��ʵ��̼ѭ�����磺
a��6CO2+6H2O$\stackrel{����/Ҷ����}{��}$C6H12O6+6O2
b��2CO2+6H2$��_{��}^{����}$C2H5OH+3H2O
c��CO2+CH4$��_{��}^{����}$ CH3COOH
d��2CO2+6H2$��_{��}^{����}$CH2=CH2+4H2O
���Ϸ�Ӧ�У�����ܵ��ǣ���Ӧc��������ԭ��������Ϊ100%��
 ijС�����о�ǰ18��Ԫ��ʱ���֣����ݲ�ͬ�ı����ɣ�Ԫ�����ڱ��в�ͬ��������ʽ����������ǰ�ԭ������������˳�����У����γ�ͼ��ʾ�ġ���ţ����״��ͼ��ÿ������������һ��Ԫ�أ�����P�������Ԫ�أ�
ijС�����о�ǰ18��Ԫ��ʱ���֣����ݲ�ͬ�ı����ɣ�Ԫ�����ڱ��в�ͬ��������ʽ����������ǰ�ԭ������������˳�����У����γ�ͼ��ʾ�ġ���ţ����״��ͼ��ÿ������������һ��Ԫ�أ�����P�������Ԫ�أ�