��Ŀ����
16��������������ȷ���ǣ�������| A�� | ��̼���low-carbon life����һ��ʱ�е�������������������������ŷţ�CO2��CH4���dz������������� | |
| B�� | �������ǽ���������;�㷺�����������������¯�ܡ��������մɿ�����������ͻ����������� | |
| C�� | �ƹ������̫���ܡ����ܵij�������ϵͳ���������С���������ӿ��з����ö�����̼�ϳɵľ�̼������ɽ������ϣ����ڷ�չ��̼���á�ѭ������ | |
| D�� | ���������������̷��¼�����Ⱦ��Ϊ�����谷���ṹ��ͼ������Ħ������Ϊ126g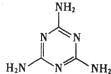 |
���� A�����ݵ�̼��������������������жϣ�C02��CH4���dz������������壻
B�������������ijɷ��������͵������մɾ������µ����ʷ�����
C����չ��̼���á�ѭ�����á��������С���������ӿ��з����ö�����̼�ϳɵľ�̼������ɽ������ϣ��ɼ��ٰ�ɫ��Ⱦ��
D��Ħ��������λΪg/mol��
��� �⣺A�����ݳ�����̼�����չ��̼�����ѳ�Ϊһ�ֻ���ʱ�У���̼������һ���Ե��ܺĺ�Ч�ܵ�Ϊ��Ҫ�������Խ��ٵ����������ŷŻ�ýϴ�������¾��÷�չģʽ����A��ȷ��
B���������ijɷ������������������иߵ��۷е㣬�����²��ϣ��������մɾ��нϸߵ�ǿ�ȺͿ�����ԣ���ѹ�����裬�����������Ӳ������֮һ�����Ⱥ��۳����壬һֱ��1900��Ż�ֽ⣬���о��˵��ͻ�ѧ��ʴ���ܣ�ͬʱ����һ�ָ����ܵ��Ե���ϣ�������������ͻ�����B��ȷ��
C����չ��̼���á�ѭ�����á��������С���������ӿ��з����ö�����̼�ϳɵľ�̼������ɽ������ϣ��ɼ��ٰ�ɫ��Ⱦ������������Ȼ���������ã���C��ȷ��
D�������谷���仯ѧʽΪC3H6N6��Ħ��������λΪ126g/mol����D����
��ѡD��
���� ���⿼�黯ѧ�뻷�������ϡ���Ϣ����Դ��ϵ������ص�֪ʶ��Ϊ��Ƶ���㣬�����ڻ���֪ʶ���ۺ���������õĿ��飬�ѶȲ���

 �����������һ��һ��ϵ�д�
�����������һ��һ��ϵ�д�| A�� | 1 mol �κ����������ķ�������ΪNA | |
| B�� | 14 g���������ĵ�ԭ����ΪNA | |
| C�� | ��״���£�22.4 Lˮ������ˮ������ΪNA | |
| D�� | �����������ķ�Ӧ�У�1 mol��ʧȥ���ӵ���ĿΪNA |
| A�� | 46gC2H6O�к��е�C-H����һ��Ϊ5NA | |
| B�� | ���³�ѹ�£�46g��NO2��N2O4��ɵĻ�������к���ԭ�ӵ�����Ϊ3NA | |
| C�� | ��15.6gNa2O2Ͷ�뵽����ˮ�У���Ӧ����Һ�е���ԭ����Ϊ0.4NA | |
| D�� | 2molSO2��1molO2��ϣ���������ķ�����Ϊ2NA |

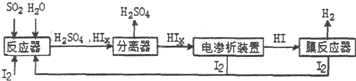
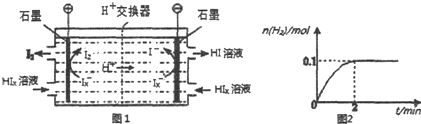
��1��Aװ������������Ӧ�����ӷ���ʽΪ��CaCO3+2H+=Ca2++CO2��+H2O��Cװ����ϡ���������Ϊ�����մ�Bװ���е��Թ����ݳ���
��2���������г�������������ڲ�ͬ�¶��µ��ܽ�����ݣ�g/100gˮ��
| 0�� | 10�� | 20�� | 30�� | 40�� | 50�� | |
| NaCl | 35.7 | 35.8 | 36.0 | 36.3 | 36.6 | 37.0 |
| NaHCO3 | 6.9 | 8.1 | 9.6 | 11.1 | 12.7 | 14.5 |
| NH4Cl | 29.4 | 33.3 | 37.2 | 41.4 | 45.8 | 50.4 |
��3����Уѧ���ڼ�������װ�������Ժ����ʵ�飬���û�еõ�̼�����ƾ��壬ָ����ʦָ��Ӧ��A��Bװ��֮�䣨��д��ĸ������һ��ʢ�б���NaHCO3��Һ��ϴ��װ�ã��������dz�ȥCO2�л�ϵ�HCl���壮
��4������Уѧ������ʵ��ʱ�����ñ���ʳ��ˮ�к�NaCl������Ϊ5.85g��ʵ���õ������NaHCO3���������Ϊ5.04g����NaHCO3�IJ���Ϊ60%��
| A�� | OH- | B�� | Ba2+ | C�� | NO3- | D�� | CO32- |
| A�� | 5�� | B�� | 6�� | C�� | 7�� | D�� | 8�� |
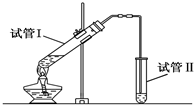 ʵ��������ͼ��ʾװ���Ʊ���������������
ʵ��������ͼ��ʾװ���Ʊ��������������� ��
��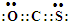 ��
��