��Ŀ����
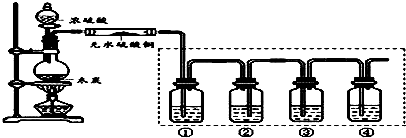
8�� ijС��ͬѧ̽��Ũ���ἰ�䲿�ַ�Ӧ����Ļ�ѧ���ʣ�װ����ͼ��ʾ���г�װ����ȥ����
ijС��ͬѧ̽��Ũ���ἰ�䲿�ַ�Ӧ����Ļ�ѧ���ʣ�װ����ͼ��ʾ���г�װ����ȥ������1���Թܢ��з�Ӧ�Ļ�ѧ����ʽ��Cu+2H2SO4��Ũ��$\frac{\underline{\;\;��\;\;}}{\;}$CuSO4+SO2��+2H2O���÷�Ӧ����Ũ�������ab������ţ���
a������ b��ǿ������ c����ԭ��
��2�����Թܢ��е��Լ�ΪƷ����Һ��ʵ���й۲쵽��������Ʒ����Һ��ɫ��
��3�����Թܢ��е��Լ�Ϊ�ữ��Ba��NO3��2��Һ���۲쵽�а�ɫ�������ɣ�˵���Թܢ������ɵ�������л�ԭ�ԣ�
��4���Թܢ��е��Լ�ΪNaOH��Һ����Ӧ�����ӷ���ʽ��SO2+2OH-�TSO32-+H2O��
���� ��1�����������£�Cu��H2SO4��Ũ����Ӧ����CuSO4��SO2��H2O���������ᷴӦǰ�ϼ۱仯�жϣ�
��2�����������ܺ���ɫ���ʷ�Ӧ������ɫ���ʶ�����Ư���ԣ�
��3����������ͨ�����ữ��Ba��NO3��2��Һ�б������Ӻ��������������������뱵���ӽ�ϳ����ᱵ��ɫ������
��4���������������������������ж����ܱ������������գ�
��� �⣺��1�����������£�Cu��H2SO4��Ũ����Ӧ����CuSO4��SO2��H2O����Ӧ����ʽΪCu+2H2SO4��Ũ��$\frac{\underline{\;\;��\;\;}}{\;}$CuSO4+SO2��+2H2O��2H2SO4���뷴Ӧ��1H2SO4���ϼ�û�䣬1H2SO4��S���ϼ���+6��Ϊ+4�����Ը÷�Ӧ����Ũ����������Ժ�ǿ�����ԣ�
�ʴ�Ϊ��Cu+2H2SO4��Ũ��$\frac{\underline{\;\;��\;\;}}{\;}$CuSO4+SO2��+2H2O��ab��
��2�����������ܺ���ɫ���ʷ�Ӧ������ɫ���ʶ�����Ư���ԣ�����������Ư��Ʒ����Һ��ʹƷ����Һ��ɫ�����Կ�������������Һ��ɫ��ȥ��
�ʴ�Ϊ��Ʒ����Һ��ɫ��
��3����������ͨ�����ữ��Ba��NO3��2��Һ�б������Ӻ��������������������뱵���ӽ�ϳ����ᱵ��ɫ������˵���Թܶ���������л�ԭ�ԣ�
�ʴ�Ϊ����ԭ��
��4���������������������������ж�������ֱ���ſգ��ܺ�NaOH��Ӧ���������������ƣ���Ӧ�����ӷ���ʽ��SO2+2OH-�TSO32-+H2O���ʴ�Ϊ��SO2+2OH-�TSO32-+H2O��
���� ������Ũ�����ͭ��ӦΪ���忼��Ũ�������ʣ���ȷʵ��ԭ���ǽⱾ��ؼ���֪��ÿ��װ�õ����ã�ע�����������Ư�����ָʾ����

| A�� | ������ˮϴ���ձ���������2��3�Σ�ÿ��ϴ�Ӻ����Һ��ע������ƿ�� | |
| B�� | �ѳ�����NaOH�������ʢ����������ˮ���ձ��У��ܽ����������Һת�Ƶ�����ƿ�� | |
| C�� | ��������ƽ�����������ϸ���һ�Ŵ�Сһ����ֽ��Ȼ��NaOH�������ֽ�Ͻ��г��� | |
| D�� | ���Ų�����������ƿ�м�������ˮ��ֱ����Һ��Һ��ǡ����̶����� |
| A�� | ���淴Ӧ���ʶ����� | |
| B�� | ƽ�ⲻ�ƶ� | |
| C�� | �ٴ�ƽ��ʱc��D�� ���� | |
| D�� | C������������ٱ仯˵���ٴδﵽƽ�� |
 ijѧУ����ѧϰ�С����Խ̲���ͭ��Ũ���ᷴӦ�������̽�����ܹ���ͭ��Ӧ����������Ũ���Ƕ��٣��������⣬����������·�������ʵ�飺
ijѧУ����ѧϰ�С����Խ̲���ͭ��Ũ���ᷴӦ�������̽�����ܹ���ͭ��Ӧ����������Ũ���Ƕ��٣��������⣬����������·�������ʵ�飺