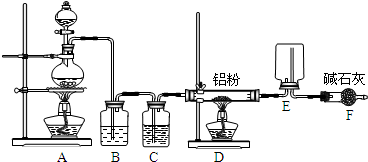
��Ŀ����
ijѧϰС������ͼװ�òⶨ��þ�Ͻ������������������������ԭ��������
(1)A���Լ�Ϊ_____________________________��
(2)ʵ��ǰ���Ƚ���þ�Ͻ���ϡ���н���Ƭ�̣���Ŀ����___________________________��
(3)����������ԣ���ҩƷ��ˮװ��������У�����
��װ�ú�����еIJ������У�
�ټ�¼C��Һ��λ�ã��ڽ�B��ʣ�������ˡ�ϴ�ӡ�������أ��۴�B�в���������������ָ������º�¼C��Һ��λ�ã�����A��B�еμ������Լ����ݼ�������ԡ�����������˳����_________���� ____________������ţ���
��¼C��Һ��λ��ʱ��������ƽ���⣬��Ӧ_________������������ ________________��
(4)B�з�����Ӧ�Ļ�ѧ����ʽΪ:
______________________������������ _______________________��
(5)����ʵ������þ�Ͻ������Ϊa g ������������Ϊb mL ���ѻ���Ϊ��״������B��ʣ���������Ϊc g�����������ԭ������Ϊ_____________�� _______��
(6)��ʵ������У���δϴ�ӹ������õIJ����������������������___________________���ƫ����ƫС����������Ӱ�족����
��1��NaOH��Һ ��2����ȥ��þ�Ͻ���������Ĥ
��3���ݢ٢ܢۢڣ� ʹD��C��Һ����ƽ
��4��2Al��2NaOH��6H2O��2AlO2-��3H2��
��5��![]()
��6��ƫС

 ������ϵ�д�
������ϵ�д� �±�Сѧ��Ԫ�Բ���ϵ�д�
�±�Сѧ��Ԫ�Բ���ϵ�д�