��Ŀ����
�յ���һ�ţ�sudan����һ��ż��Ⱦ�ϣ�������ΪʳƷ���Ӽ�ʹ�ã������ɱ�����2-����Ϊ��Ҫԭ���Ʊ��ģ����ǵĽṹ��ʽ��ͼ��ʾ��
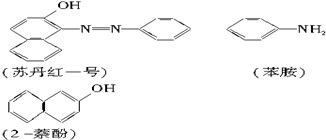
��1���յ���һ�ŵĻ�ѧʽ������ʽ��Ϊ ��
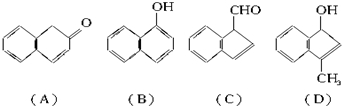
��2�������滯���A������D���У���2-���ӻ�Ϊͬ���칹����У�����ĸ���ţ� ��
��3�����ʵ��������£�2-���Ӿ���Ӧ�ɵõ����㻯����E��C8H6O4����1mol E��������̼�����Ʒ�Ӧ�ɷų�44.8L CO2 ����״������E�Ľṹ��ʽΪ ��
��4������E�������Ҵ���Ũ���������¼��ȣ���������һ����ѧʽ������ʽ��ΪC12H14O4���»�����÷�Ӧ�Ļ�ѧ����ʽ�� ����Ӧ������ ��

��1���յ���һ�ŵĻ�ѧʽ������ʽ��Ϊ
��2�������滯���A������D���У���2-���ӻ�Ϊͬ���칹����У�����ĸ���ţ�

��3�����ʵ��������£�2-���Ӿ���Ӧ�ɵõ����㻯����E��C8H6O4����1mol E��������̼�����Ʒ�Ӧ�ɷų�44.8L CO2 ����״������E�Ľṹ��ʽΪ
��4������E�������Ҵ���Ũ���������¼��ȣ���������һ����ѧʽ������ʽ��ΪC12H14O4���»�����÷�Ӧ�Ļ�ѧ����ʽ��
���㣺�л���Ľṹ������,�л�������еĹ����ż���ṹ
ר�⣺�л���Ļ�ѧ���ʼ��ƶ�
��������1���յ���-�ź���C��H��O��N��Ԫ�أ��ɸ��ݽṹ��ʽ��д����ʽ��
��2������ͬ���칹�����ʽ��ͬ���ṹ��ͬ�����жϣ�
��3��1mol E��������̼�����Ʒ�Ӧ�ɷų�44.8L CO2����2��-COOH��E�Ľṹ��ʽΪ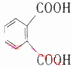 ��
��
��4��E�������Ҵ���Ũ���������¼��ȣ��ɷ���������Ӧ��
��2������ͬ���칹�����ʽ��ͬ���ṹ��ͬ�����жϣ�
��3��1mol E��������̼�����Ʒ�Ӧ�ɷų�44.8L CO2����2��-COOH��E�Ľṹ��ʽΪ
 ��
����4��E�������Ҵ���Ũ���������¼��ȣ��ɷ���������Ӧ��
���
�⣺��1���յ���-�ź���C��H��O��N��Ԫ�أ�����ʽΪC16H12N2O���ʴ�Ϊ��C16H12N2O��
��2��2-���ӵķ���ʽΪC10H8O������ABC�ķ���ʽҲΪC10H8O����2-���ӻ�Ϊͬ���칹�壬D�ķ���ʽΪC10H10O����2-���ӵķ���ʽ��ͬ������ͬ���칹���ϵ��
��3��2-���Ӿ���Ӧ�ɵõ����㻯����E��C8H6O4����1mol E��������̼�����Ʒ�Ӧ�ɷų�44.8L CO2 ����״��������2��-COOH����Ϊ�ڱ������ᣬE�Ľṹ��ʽΪ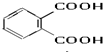 ���ʴ�Ϊ��
���ʴ�Ϊ�� ��
��
��4��E�������Ҵ���Ũ���������¼��ȣ��ɷ���������Ӧ������������Ӧ�Ļ�ѧ����ʽΪ ��
��
�ʴ�Ϊ�� ��������Ӧ��
��������Ӧ��
��2��2-���ӵķ���ʽΪC10H8O������ABC�ķ���ʽҲΪC10H8O����2-���ӻ�Ϊͬ���칹�壬D�ķ���ʽΪC10H10O����2-���ӵķ���ʽ��ͬ������ͬ���칹���ϵ��
��3��2-���Ӿ���Ӧ�ɵõ����㻯����E��C8H6O4����1mol E��������̼�����Ʒ�Ӧ�ɷų�44.8L CO2 ����״��������2��-COOH����Ϊ�ڱ������ᣬE�Ľṹ��ʽΪ
 ���ʴ�Ϊ��
���ʴ�Ϊ�� ��
����4��E�������Ҵ���Ũ���������¼��ȣ��ɷ���������Ӧ������������Ӧ�Ļ�ѧ����ʽΪ
 ��
���ʴ�Ϊ��
 ��������Ӧ��
��������Ӧ��
���������⿼���л���Ľṹ�����ʣ�Ϊ��Ƶ���㣬��Ŀ�Ѷ��еȣ�����ע��ͬ���칹����жϣ�Ϊ������ѵ㣬Ҳ���״��㣮

��ϰ��ϵ�д�
 ������ѧ���̲���ȫ���ϵ�д�
������ѧ���̲���ȫ���ϵ�д� ������ʱ����ҵ����ϵ�д�
������ʱ����ҵ����ϵ�д�
�����Ŀ
��50mL 0.3mol?L-1 NaOH��Һ��ˮϡ�͵�300mL��ϡ�ͺ���Һ��NaOH�����ʵ���Ũ��Ϊ��������
| A��0.03mol?L-1 |
| B��0.05mol?L-1 |
| C��0.5mol?L-1 |
| D��0.04mol?L-1 |
ȡ0.2mol?L-HX��Һ��0.2mol?L-NaOH��Һ�������ϣ����Ի�Ϻ���Һ����ı仯������û����Һ��pH=8��25�棩��������˵�������ϵʽ����ȷ���ǣ�������
| A�������Һ����ˮ�������c��OH-��=1��10-8mol/L |
| B��c��Na+��=c��X-��+c��HX��=0.2mol?L- |
| C��c��Na+��-c��X-��=9.9��10-7mol?L- |
| D��c��OH-��-c��HX��=c��H+��=1��lO-6mol?L- |
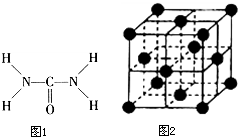 ��ͼ1����֪���صĽṹʽΪ�����ؿ��������л����ʣ���Ҫ�������������ᣬ�����غ���������ѧʽΪ[Fe��H2NCONH2��6]
��ͼ1����֪���صĽṹʽΪ�����ؿ��������л����ʣ���Ҫ�������������ᣬ�����غ���������ѧʽΪ[Fe��H2NCONH2��6]


